chemisty year 10 sem 1
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
40 Terms
element symbols
- represent the names of different elements
- consist of one or two letters
- first letter is always capital, if there is a second letter then it is lowercase
structure of periodic table (colums/rows)
- horizontal rows are called periods, and each one is given a number starting from the top
- vertical columns are called groups, and each one is given a number starting from the left
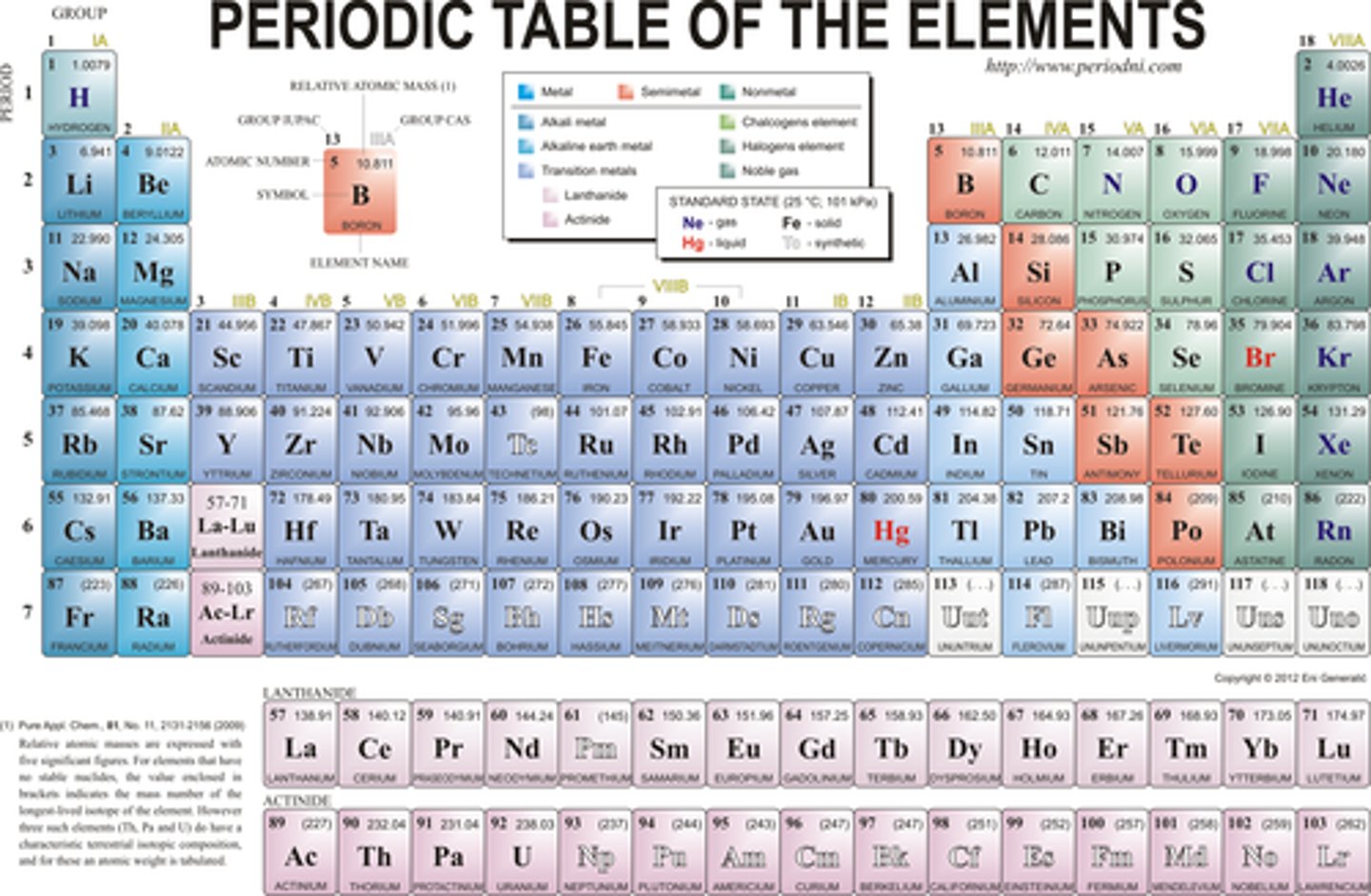
metals, non-metals and metalloids location on periodic table
metals are on the left side of the periodic table
non-metals are on the right side of the periodic table
metalloids are on a staircase shape between the metals and non-metals
metals
form 80%~ of the periodic table
group 1 (alkali metals): low melting points, soft, and very reactive
group 2 (alkaline earth metals): relatively soft and reactive
group 3-12 (transition metals): form coloured compunds, some are magnetic.
- only metals that are not silver in colour are copper (Cu) and gold (Au)
non-metals
while only 18 elements are considered to be non-metals, they make up most of the atomosphere and crust of Earth as well as living organisms
group 17 (halogens): only group to contains gases, a liquid (Bromine), and solids, most reactive group of non-metals
group 18 (noble gases): all gases and have a full valence shell, so are very unreactive
metalloids
- properties of both metals and non-metals
subatomic particles location in atom
protons and neutrons are tightly packed in the nucleus of an atom so they can only vibrate in position
electrons move in regions of space around the nucleus
cannot determine where an electron (we describe the region of space is electron is likely to be in) is and how fast it moving
region of space = electron cloud
properties of sub-atomic particles
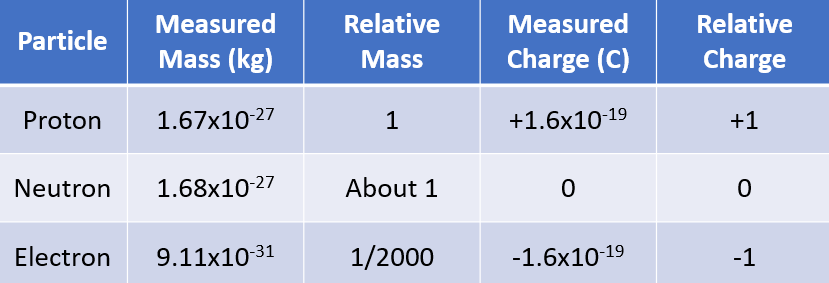
number of protons, electrons, neutrons in atoms
neutral atoms - same number electrons and protons
atoms usually have the same number of neutrons and protons
adding a proton makes a new kind of atom
adding a neutron makes an isotope of that atom, a heavier version of that atom
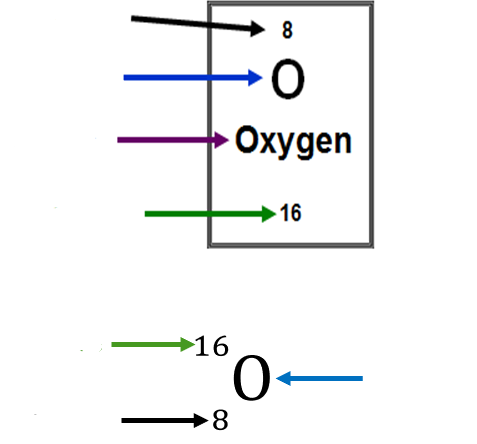
atomic number and atomic mass
atomic number - number of protons in an element and number of electron in a neutral atom of the element
atomic mass - number of protons and neutrons added together in an isotope of an element
mostly not whole numbers on the periodic table
because masses are a weighted average of all the isotopes of that element
the bottom thing is called an atomic symbol
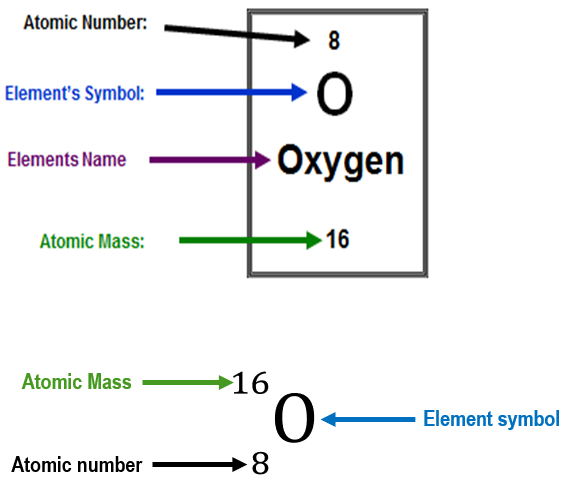
isotopes
atoms with the same number of protons but a different number of neutrons are called isotopes
named by their mass number as they have a different mass (because of the different number of neutrons they contain)
electron configuration
arrangement of electrons around the nucleus
electrons occupy energy levels
first energy level: max 2 electrons
second energy level: max 8 electrons
third energy level: 8 electrons (not max)
fourth energy level: 2 electrons (not max)
an abbreviated way of writing this electron structure would be: 2, 8, 8, 2
this is called its electron configuration.
orbitals
electrons can only be in an energy level if they have the correct amount of energy
energy level can contain sub-levels called orbitals
orbital contains a maximum of two electrons
in energy level diagrams, electrons are drawn in pairs, showing they occupy the same orbital.
Only done if an energy level contains more than 4 electrons
electron configuration and the periodic table
element’s position can tell its electron configuration
period number determines number energy levels
eg: period 2 elements have 2 energy levels and period 6 elements have 6 energy levels
group number determines the number of valence electrons
eg: group 2 elements have 2 valence electrons and group 14 elements have 4 valence electrons
electron configuration + periodic table + valence electrons
valence electrons influence → element’s chemical properties
same group → similar properties → same number of valence electrons
excited electrons
in an atom, electrons are found in specific areas around the nucleus → electron shells
when thermal/electrical energy is added → electrons in the outermost shell jump to a shell further away from the nucleus
when an electron has jumped to a higher electron shell → excited electron
excited electrons eventually drop back to their original shell (extra energy is emitted as light/a photon)
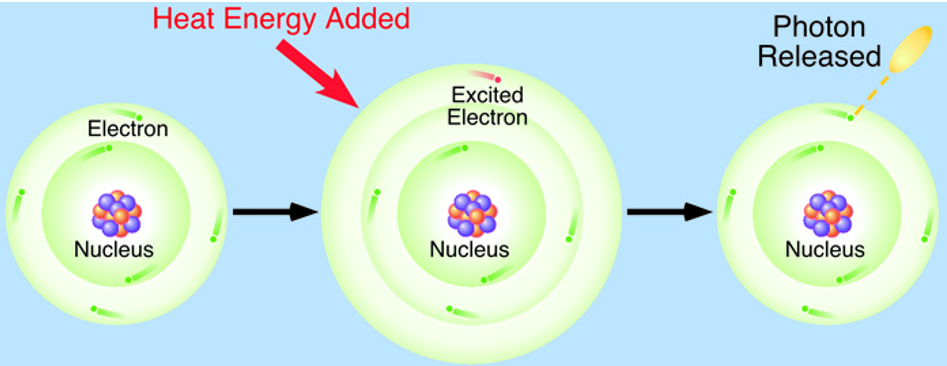
colours of light emitted in excited electrons
colour of light emitted depends on the element
used to identify different elements
eg: compounds with sodium → yellow colour
eg: potassium → pale purple
electrons and chemical reactions (1)
in chemical reaction → valence electrons of atoms interact
electrons are shared/exchanged between atoms during the reaction
at the end of reaction:
atoms either have filled their valence energy levels to 8 (non- metals)
atoms empty their valence energy levels (metals)
eg: fluorine is a non-metal with 7 valence electrons, so it will gain 1 more to have a full valence energy level
hydrogen can either empty it valency energy level or fill it up to 2 depending on the reaction

electrons and chemical reactions (2)
depending on which makes the least change to the atom, they either:
gain electrons to fill their valence energy level to 8
lose electrons to empty their valence energy level
atoms with a full/empty valence energy level are more stable (why noble gases are stable)
ions and valency
atoms which have gained/lost electrons are ions
they are no longer neutral (became charged)
the charge on an ion (eg: 2+, 3-) is called its valency
most atoms can only form one ion but some metals can have different valencies
when naming the ion; the valency is written in Roman numerals in brackets after the element name
Fe2+ → Iron (II)
Fe3+ → Iron (III)
positive ions
when atoms lose electrons
number of electrons lost → amount of positive charge
valency is written at the top right of the symbol
Al3+: an aluminium ion form when an aluminium atom loses 3 valence electrons
Li+: a lithium ion forms when a lithium atom loses a valence electron
Ca2+: a calcium ion forms when a calcium atom loses 2 valence electrons
negative ions
when atoms gain electrons
number of electrons gained → amount of negative charge
valency is written at the top right of the symbol
F-: a fluoride ion forms when a fluorine atom gains 1 valence electron
P3-: a phosphide ion forms when a phosphorus atom gains 3 valence electrons
S2-: a sulfide ion forms when a sulfur atom gains 2 valence electrons
ions and the periodic table
metal elements → positive ions
non-metal elements → negative ions
more than 4 = gains electrons
less than 4 valence electrons = loses electrons
except for metaloids (form covalent bonds) and some metals who lose or gain
(except hydrogen which forms either)

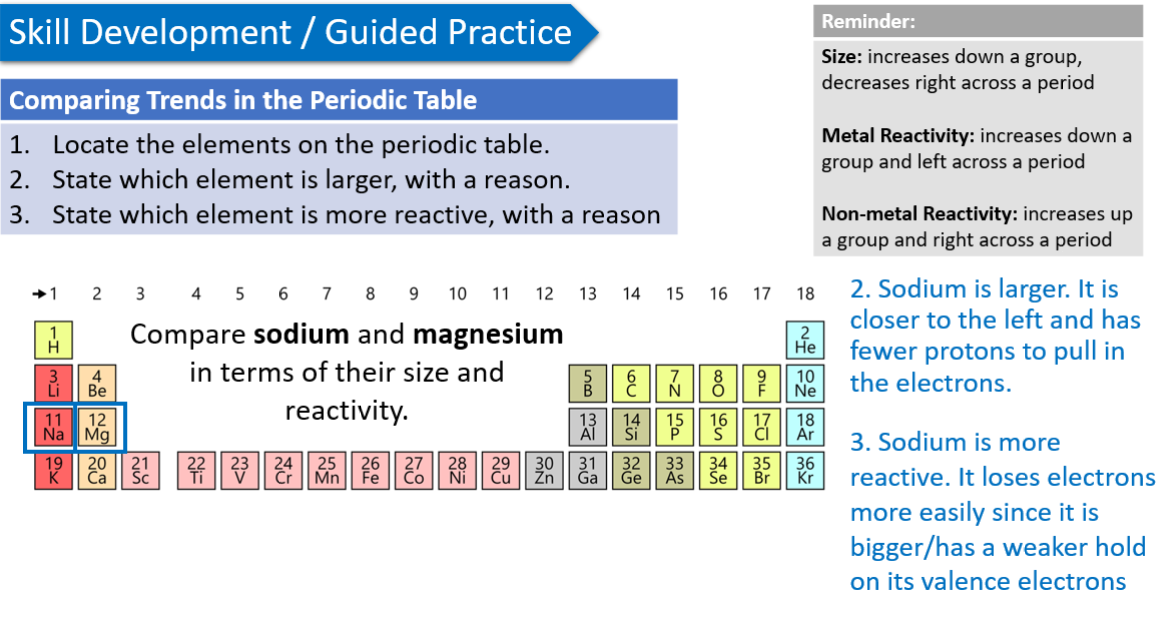
trends in periodic table - atomic size
atom’s size is measured using the radius of the atom
a new energy level is added with each period → atom grows bigger → atomic size increases from top to bottom
negative electrons are pulled in more tightly as the number of positive protons in the nucleus increases → atomic size decreases from left to right
trends in periodic table - reactivity METALS
metal reactivity increases from top to bottom and right to left
larger metal atoms have a weaker hold on their valence electrons → more readily removed → causing chemical reaction to occur
trends in periodic table - reactivity NON-METALS
metal reactivity increases from bottom to top and left to right
smaller non-metal atoms with more electrons in their valence shell → more capable of ripping electrons off other atoms to fill their valence shell

Compare sodium and magnesium in terms of their size and reactivity.
Bonding
In a chemical reaction, the valence electrons of atoms are shared or exchanged
At the end of the reaction, atoms have either filled or emptied their valence shell
Reactions cause atoms to join together, forming chemical bonds
Types of Bonding
Metallic bonding: metals only
Covalent bonding: non-metals only
Ionic bonding: metals and non-metals
Metallic Bonding
metal atoms have a weak hold on their valence electrons
valence elections are free to move through out the metal without being bound to one particular atom
the electrons are “shared” by the metal atoms are called delocalised atoms
eg: copper, iron, zinc (CIZ)
Properties of Metallic Bonds
Metals are good conductors of electricity → electrons can move freely through the metal allowing them to carry an electric current
This is why metals are also good conductors of heat- the electrons carry the energy through the substance
Metals are malleable; can be bend or reshaped. Even if the metal atoms are moved around, the electrons will stay between them and hold them together
Metals have high melting and boiling points → positive metal atoms are attracted to negative electrons → a lot of energy is needed to separate them
Covalent Bonding:
Non-metal atoms bond with other non-metal atoms by sharing their valence electrons
Electrons are shared so that the valence shell for each atom is ful
eg: water, hydrogen, carbon dioxide
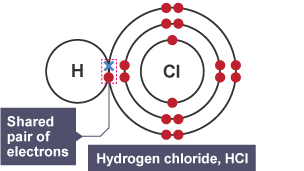
Properties of Covalent Bonding
Covalent substances do not conduct electricity or form ions (even when dissolved) and their electrons are trapped in the bonds between atoms → can’t carry an electric current
Brittle and break easily → bonds within covalent molecules are very strong, the bonds between them are weak and easy to break
Low melting and boiling point for the same reason
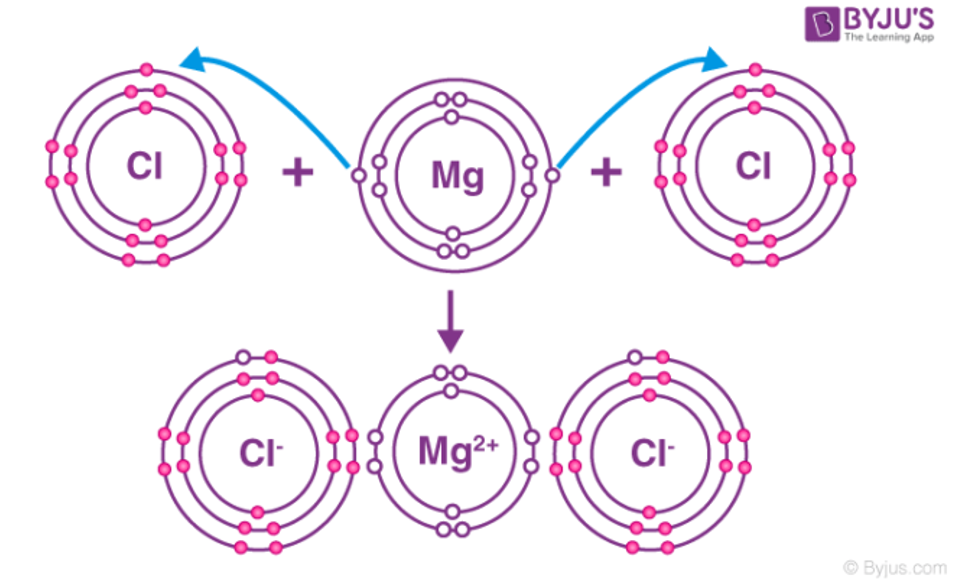
Ionic Bonding
Form when a metal atoms transfers its valence electrons to non-metal atoms
Metal becomes positively charged and non-metal becomes negatively charges
Ionic bond forms from the electrostatic attraction between the oppositely charged ions
Each ion is surrounded by ions of the opposite charge, building up a 3D structure called a lattice
eg: table salt; sodium chloride
Properties of Ionic Bonds
Ionic solids cannot conduct electricity → ions hold each other in place so particles can’t move but when ionic substance is melted/dissolved → ions can move freely and conduct electricity
Ionic compounds are brittle. Large enough force can push ions with the same charge close together → like charges repel → push each other apart
Force of attraction between opposite ions in the lattice is very strong → high melting and boiling points
Forming of Ionic Compounds
Positively charged ions (cations) bond with negatively charged ions (anions) through electrostatic forces of attractions (positive and negative charges attract)
Bond to create a compound with a net (total) charge of zero
sum of neg charges = sum of positive charges
Polyatomic Ions
composed of more than one atom
create a compound with a net (total) charge of zero
more than 1 of a polyatomic ion → brackets around it
before writing the number in subscript
eg: Mg(NO3)2
Naming Ionic Compounds
name of the cation first
name of the anion with the ending changed to “ide“ second
Numbers are ignored.
Naming Ionic Formulae
symbol of cation first and symbol of anion second
If necessary, adjust the number of one or both ions so that the total positive charge equals the total negative charge
Remove charges and use subscripts to indicate the quantity of each ion. 1s are not written.
Electron Dot Diagrams
also called Lewis dot diagrams
Electron dot diagrams show the electrons in the valence energy level.
bc chemical reactions only involve atoms’ valence electrons
can be used to show bonds between elements