3.3- halogenoalkanes
0.0(0)
Studied by 1 personCard Sorting
1/19
Earn XP
Description and Tags
Last updated 9:41 PM on 1/5/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
1
New cards
Define halogenoalkanes
Alkanes with 1/more halogens attached to it
2
New cards
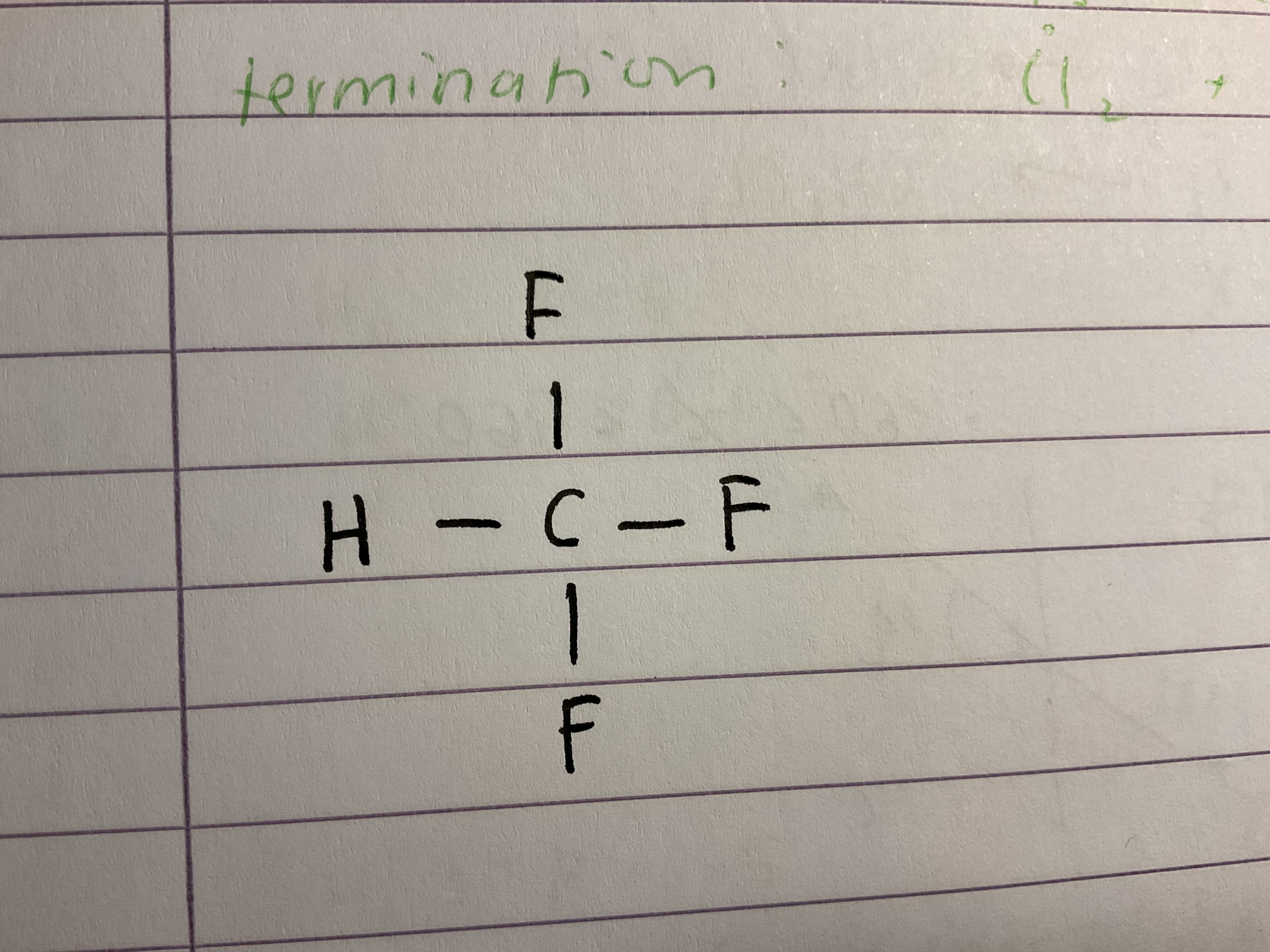
Name this halogenoalkane
Trifluoromethane
3
New cards
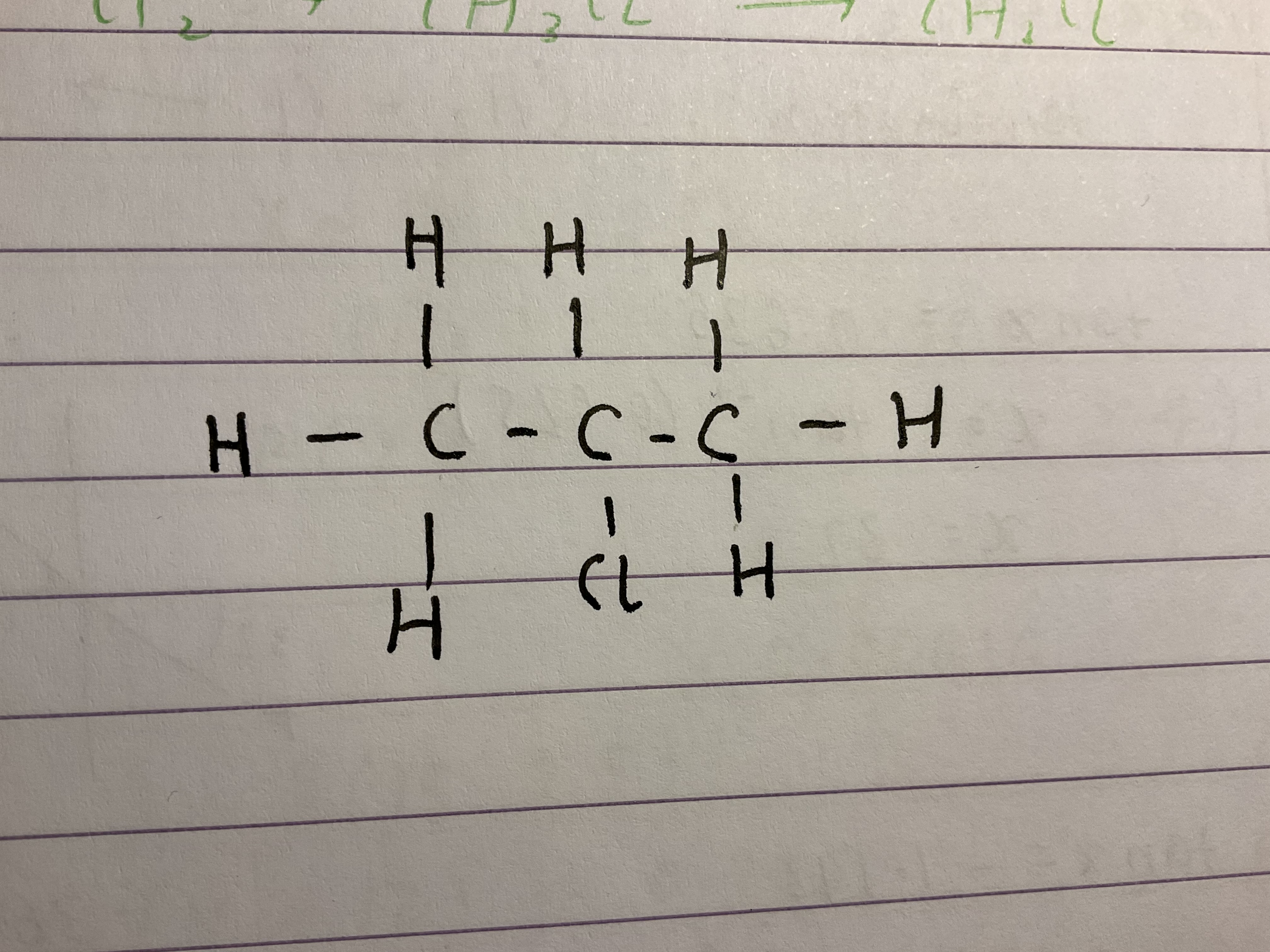
Name this halogenoalkane
2-chloropropane
4
New cards
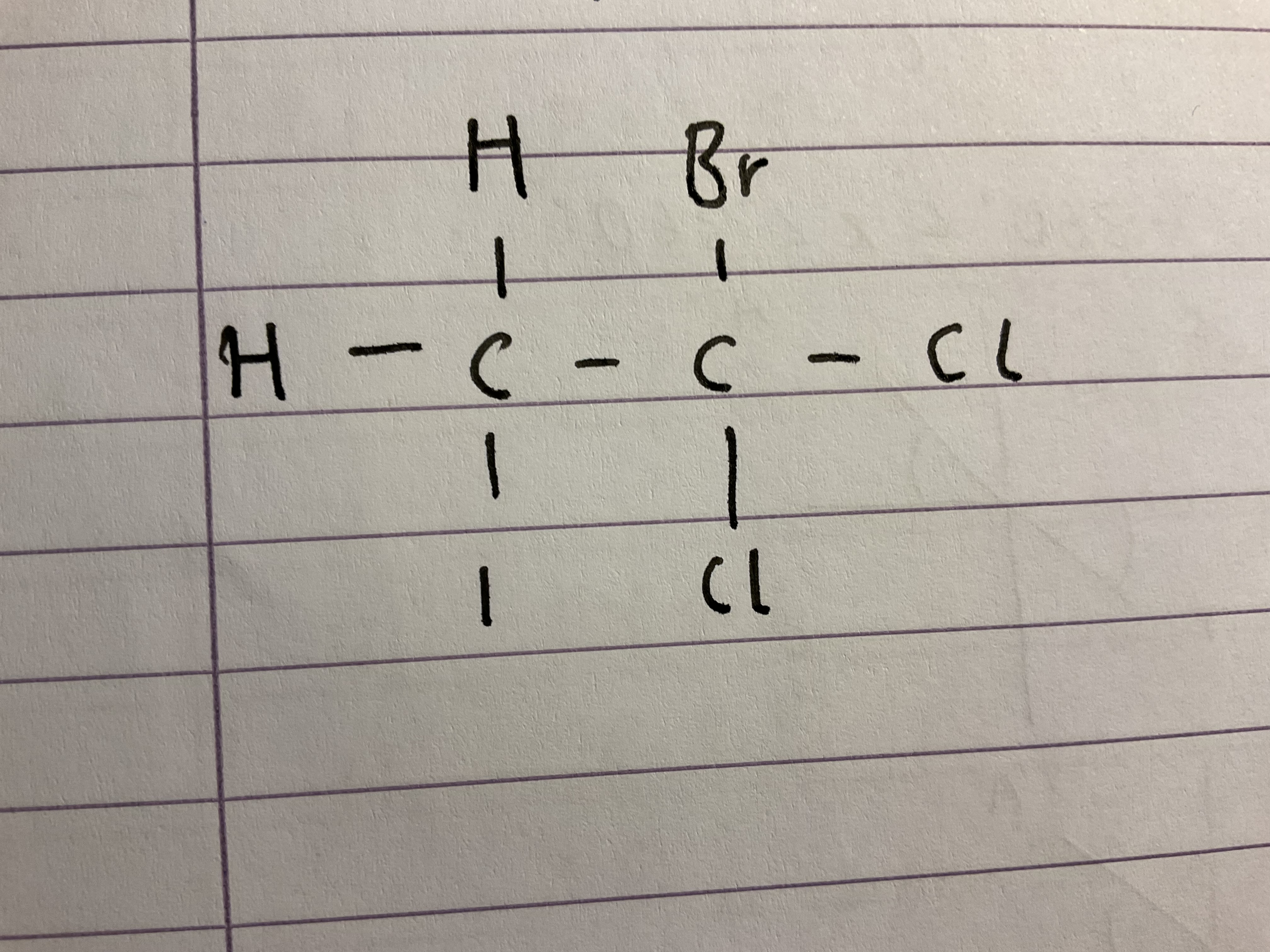
Name this halogenoalkane
1-bromo-1,1-dichloro-3-iodoethane
5
New cards
Describe the boiling point trend of halogenoalkanes
Increases down the group
6
New cards
Why do the boiling points increase down the group
\-down the group electrons increase
\-stronger van de waals between molecules
\-more energy needed to overcome forces
\-stronger van de waals between molecules
\-more energy needed to overcome forces
7
New cards
Define a nucleophile
Substance that is an electron pair donor
8
New cards
Give examples of nucleophiles
\-ammonia
\-hydroxide ions
\-cyanide ions
\-hydroxide ions
\-cyanide ions

9
New cards
How do halogenoalkanes react with hydroxide ions
Via nucleophilic substitution
10
New cards
Conditions for nucleophilic substitution
\-warm aqueous sodium hydroxide
\-carried out under REFLUX
\-carried out under REFLUX
11
New cards
How do halogenoalkanes react with cyanide ions
Halogenoalkanes react with cyanide ions to make nitriles
12
New cards
Conditions for nucleophilic substitution with cyanide ions
\-warm ethanoic potassium cyanide
\-carried out under REFLUX
\-carried out under REFLUX
13
New cards
Describe trend of halogenoalkane activity
Reactivity increases down the group
14
New cards
What determines reactivity
Bond enthalpy
15
New cards
Describe reaction between hydroxide ions and halogenoalkanes
Elimination using ethanol solvent
16
New cards
Conditions for the reaction with hydroxide ions
\-warm ethanol in sodium hydroxide
\-carried out under REFLUX
\-carried out under REFLUX
17
New cards
How does UV radiation break down CFCs
Breaks C-Cl bonds, radicals are formed and catalyse the break down of ozone
18
New cards
Why are C-Cl bonds broken easiest in CFCs
Lowest bond enthalpy
19
New cards
How does CFCs destroy ozone
Breaks down to form chlorine radicals that catalyse the breakdown of ozone
20
New cards
What is an alternative to CFCs
HFCs and hydrocarbons