Stage 2 Chemistry, Unit 1.3- Monitoring the Environment-Volumetric Analysis
1/29
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
30 Terms
Mass =
m (in grams
Volume
V (in litres)
Density (probably dont need this)
p (kg/L)
Avogardo’s number (アボガドロの定数)
NA (6.022×1023) (Mol/L)
Mole
n (Mol) one of avogardo’s number
Molar mass
M (g/mol)
Moles=
n=m/M
Mass concentration =
P= m/v
Molar concentration =
C= n/v
Dilution equation =
C1V1 =C2V2
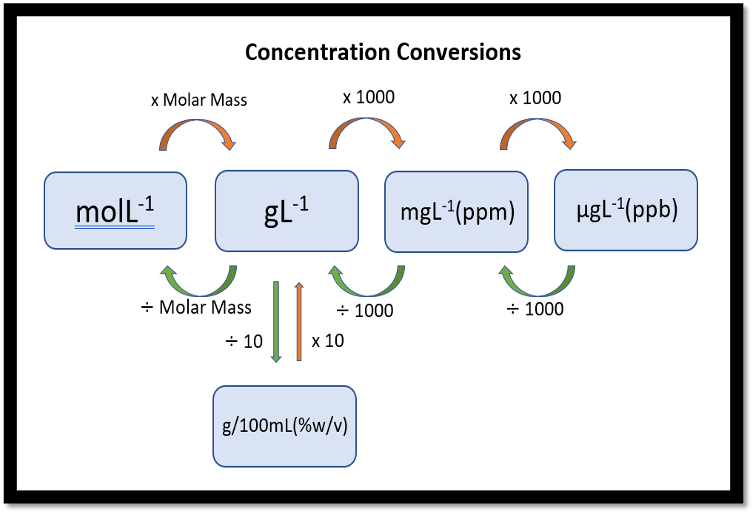
Concentration conversion
Mass-mass Stoichiometry
Use the mass given to convert the chemical into moles. Use the mole ratio to find the moles of the unknown value., when the moles of the unknown chemical is found, find the volume, concentration, or mass
Write fully balanced chemical equation 2. Find the number of moles of known solution (n=c x v) 3. Use mole ratio to find moles 4. Calculate concentration (c = n/v) OR volume (v = n/c) OR Mass (m=M/n)
Limiting reagent
The first reactant that runs out in a reaction. This is determined with comparing the moles of each reactants, and check which is smaller, OR use the mole ratio (coefficients present in the reaction) to determine how much is needed if one is fully used. The mole of the limiting reagent also tells you the mole of the products.
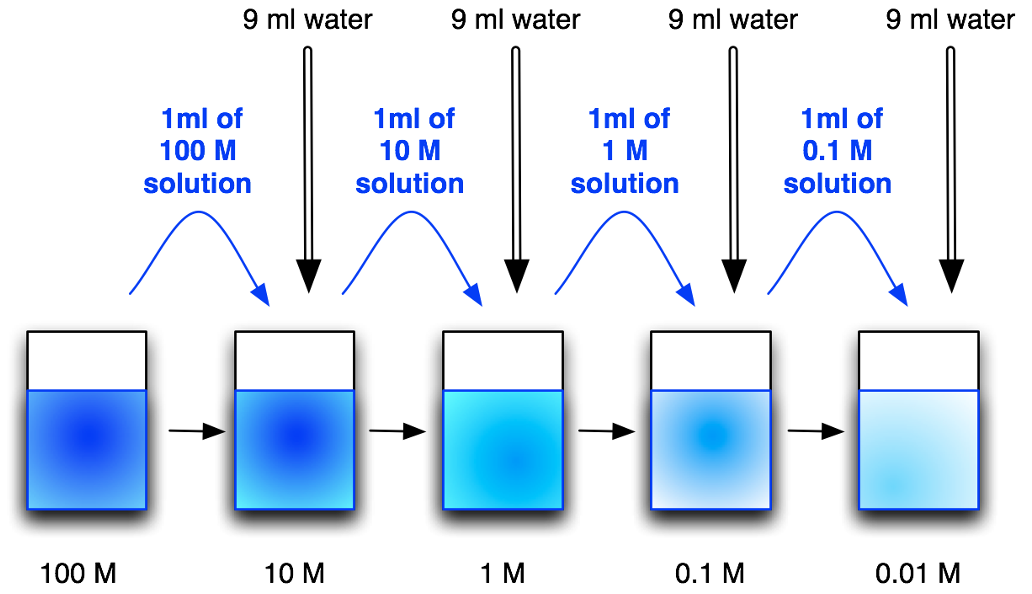
Serial dilutions
are done to generate a series of solutions from a single standard solution, to create calibration graphs.
Standard solution
Clean all glassware
Weight out the mass of solute needed for the solution
Transfer the mass of solute to a clean beaker (use solvent to remove excess solute from the surface)
Dissolve the solute in appropriate amount of solvent
Transfer the solution from the beaker to the volumetric flask
funnel used to ensure all of the solution is transferred.
Add solvent to the volumetric flask through the use of a Burette until the solution is parrallel with the graduation mark at eye level. (bottom of meniscus)
invert several times until substance is fully dissolved.
Accuracy
About the validity of the results given. How close are your answers to the expected answer.
Precision
Is about consistency between the numbers, and the reliability and reproducibility of the data
Systematic errors
(error in the system or procedure)
Poor accuracy,
definite causes,
reproducible (high precision)
Random Error
Poor precision, no specific causes,
not reproducible (could be accurate but fluctuating results
Direct titration
a solution of an unknown concentration is determined by applying the mole ratio of a known concentration via a neutralisation
Titration steps.
Using the given concentration and volume, find moles. use mole ration to multiply or divide the unknown by the ratio. use the new mole to find the concentration (C=N/v
What is a reagent
a chemical added to start or test a chemical reaction, or determine the presence of a specific chemical substance.
What is an Analyte?
the unknown in a titration reaction
Back titrations
1) A chemical with unknown concentration (analyte) (A) is reacted to a known amount of reagent (B). A+B →
2) However, after this reaction, there is an excess of B leftover. This undergoes a second reaction with another reagent (C). (B+C)
3)The amount of C used to neutralise B’s excess is used to determine how much was of B was leftover from reaction 1 (from neutralising A.) (mole ratio)
4) As the amount of B was added originally is still known, the volume used to neutralised A can be determined
Volume of B to neutralise A (= original volume- excess)
5) The volume of B used for neutralisation is then changed to moles, and via the mole ratio determines the moles of A present.
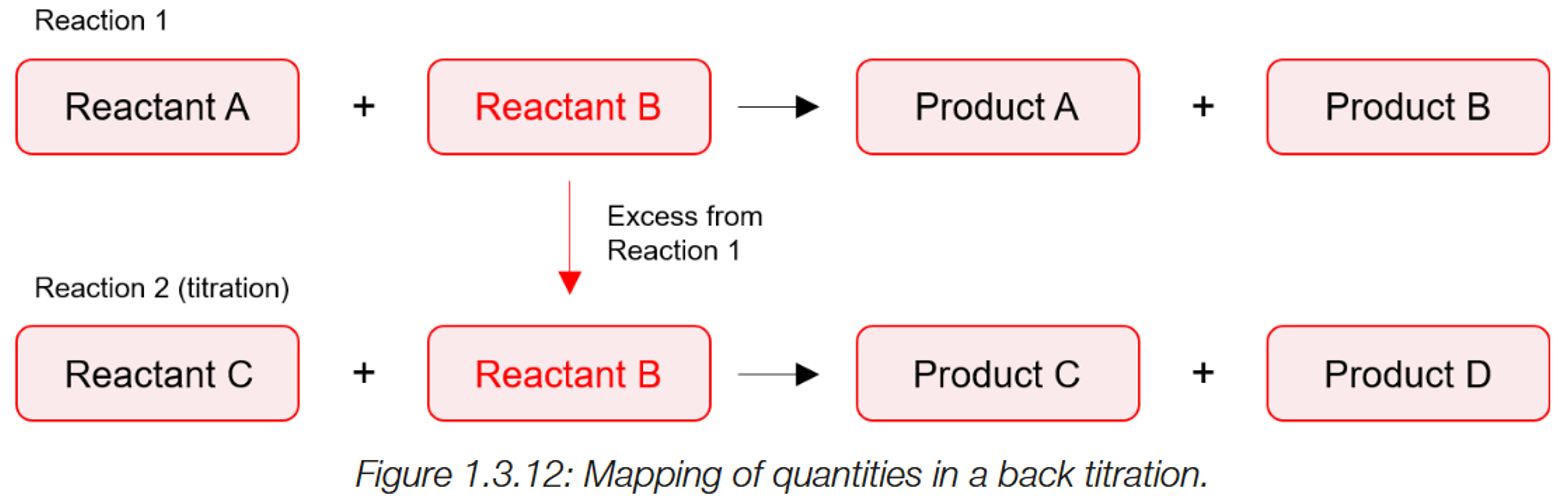
Why are back titrations and indirect titrations done?
The unknown could be volatile and dissolve into the atmosphere, the unknown could also be a salt, or reacts too slow
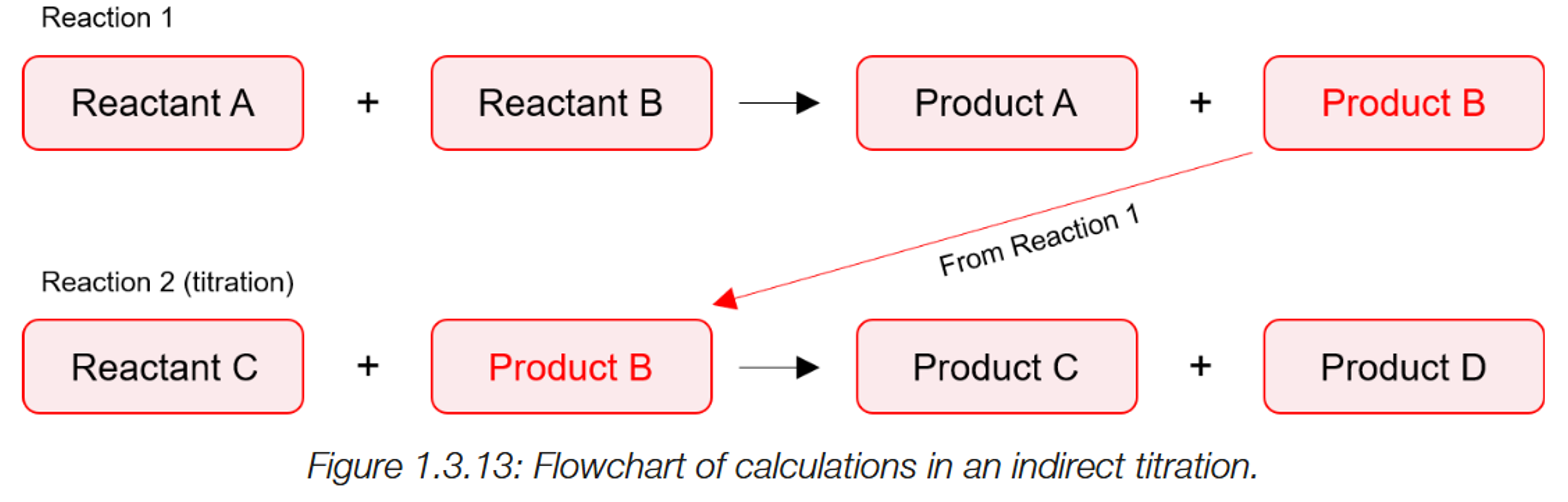
Indirect titrations.
A initial reaction takes place, with two unknowns to make a product (C) (A+B→C).
This product is used in a second reaction. (C+D), where the product is neutralised by chemical D.
The moles of chemical D used in the reaction is found as the concentration is known and the volume used is found.
Using the mole ratio, the moles of C can be found, allowing for reactants A and B from the initial reaction can also be found via the mole ratio.
G/mol→ G/L
xmolar mass
g/L→ ppm
x1000
ppm(mg)→ ppb(µg)
x1000
g/l→ g/100ml
x10