CH 223: Orgo 1 Review, Radical Bromination. Aromaticity, Conjugated Dienes, EWGs/EDGs
1/104
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
105 Terms
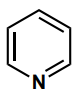
Determine the hybridization of the nitrogen atom.
sp2
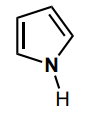
Determine the hybridization of the nitrogen atom.
sp2
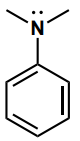
Determine the hybridization of the nitrogen atom.
sp2
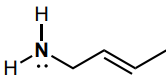
Determine the hybridization of the nitrogen atom.
sp3
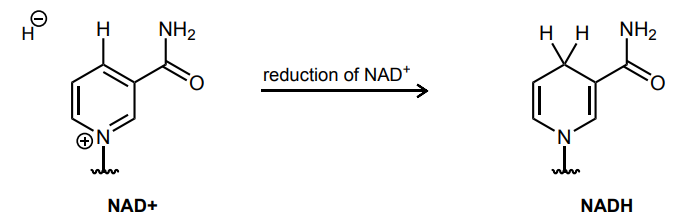
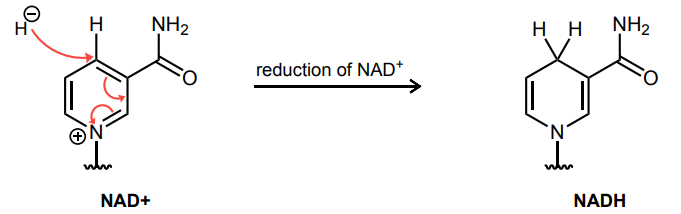
Draw the curved-arrows needed to accomplish the reduction of NAD+ to NADH.
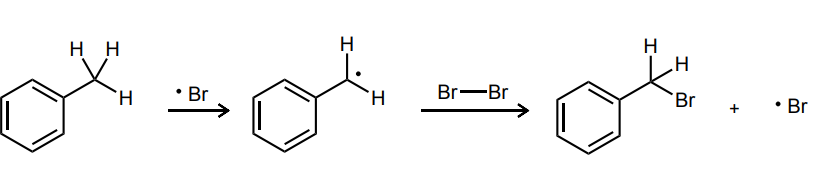
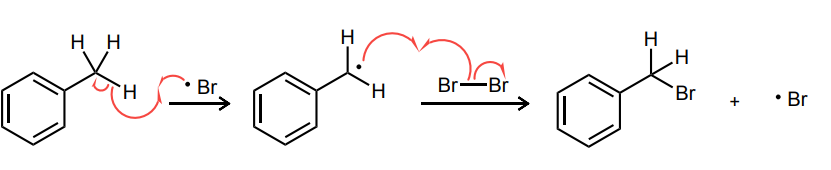
Provide the curved-arrow mechanism for the following CH bromination reaction.

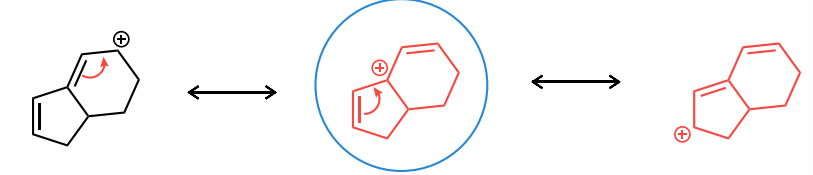
Draw the two resonance structures for the cation shown and the curved arrows needed to convert between each structure. Then, circle the resonance structure that is the major contributor.
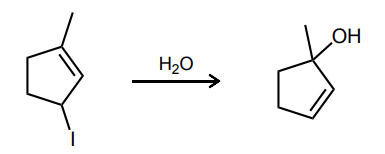
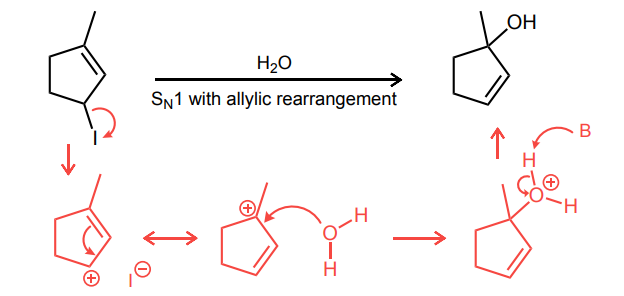
Provide the curved-arrow mechanism for the reaction below.
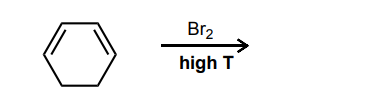
Provide the curved-arrow mechanism for the reaction below.
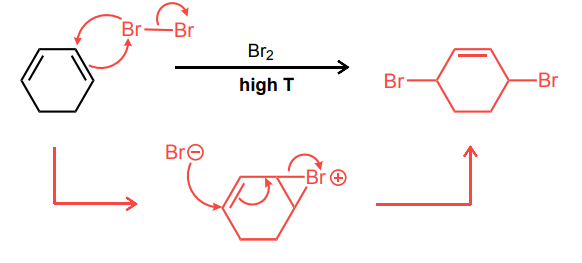
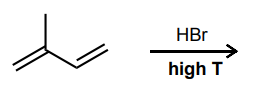
Provide the curved-arrow mechanism for the reaction below.
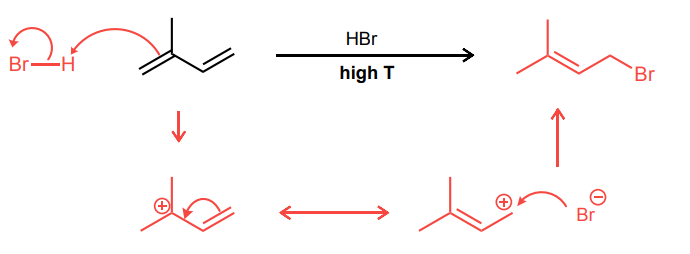
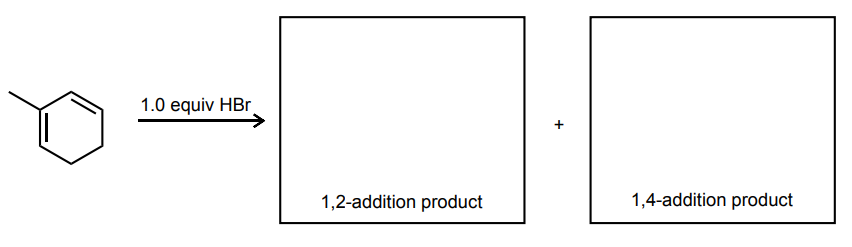
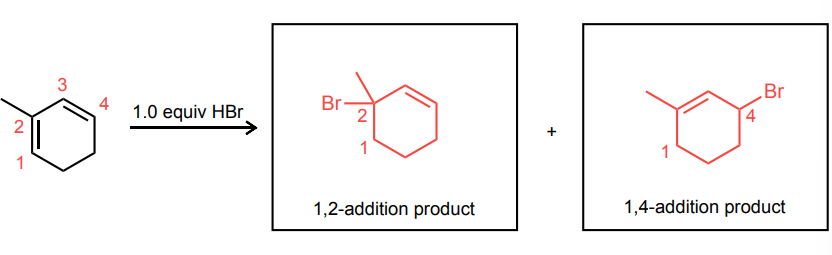
Draw the 1,2- and 1,4-addition products for the reaction below:
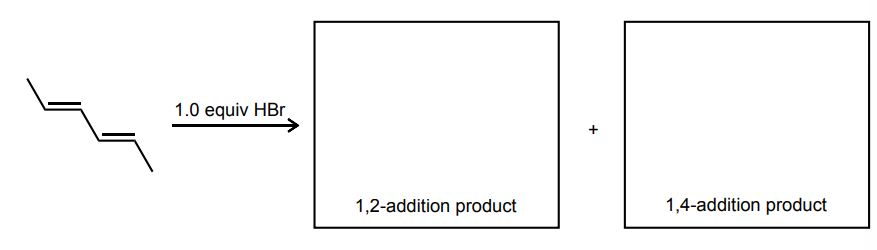
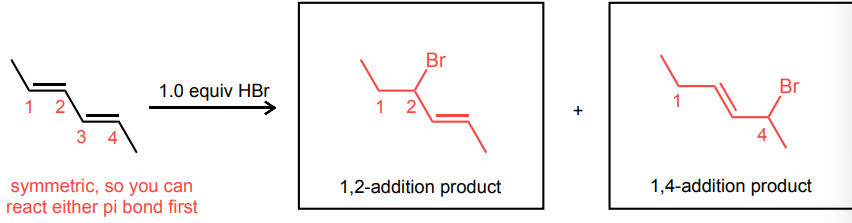
Draw the 1,2- and 1,4-addition products for the reaction below:
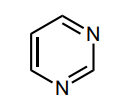
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
aromatic
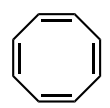
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
anti-aromatic
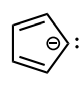
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
aromatic
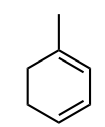
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
not aromatic
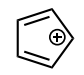
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
anti-aromatic
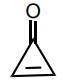
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
aromatic
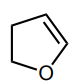
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
not aromatic
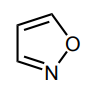
Classify the molecule as aromatic, anti-aromatic, or not aromatic.
aromatic
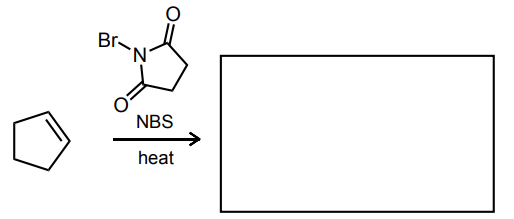
Provide the missing starting material, reagent(s), or major product of the reaction below.
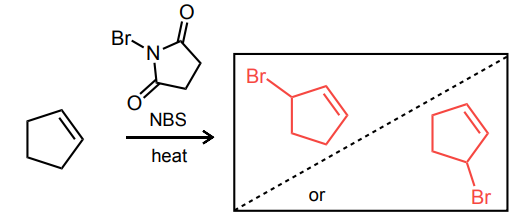
Provide the missing starting material, reagent(s), or major product of the reaction below.
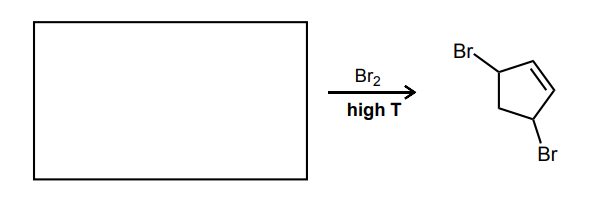
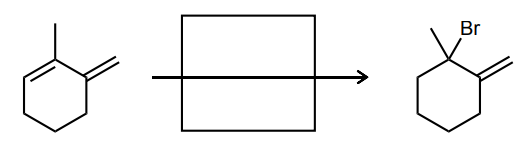
Provide the missing starting material, reagent(s), or major product of the reaction below.
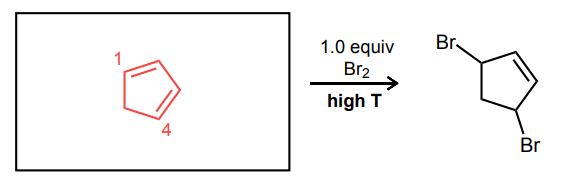
Provide the missing starting material, reagent(s), or major product of the reaction below.
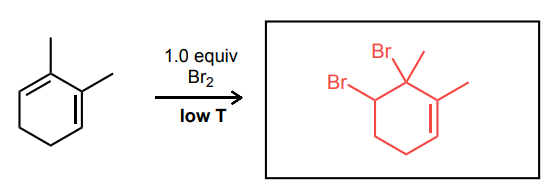
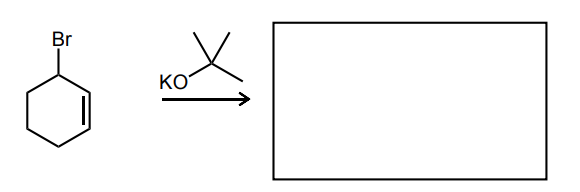
Provide the missing starting material, reagent(s), or major product of the reaction below.
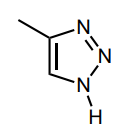
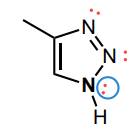
Draw any implied lone pairs and circle the single nitrogen, N, atom in triazole 1 whose lone pair is involved in the aromatic system.
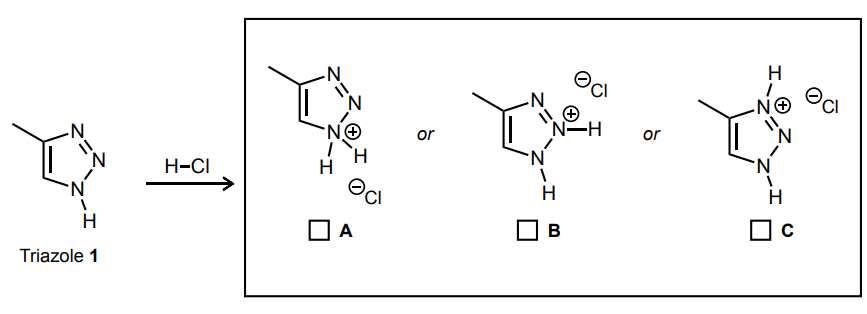
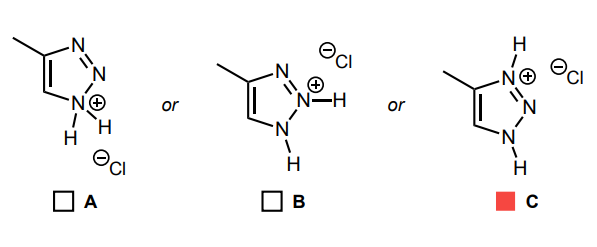
Select the single product that is formed in the reaction below
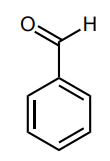
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EWG
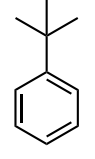
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EDG
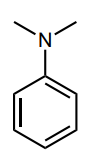
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EDG
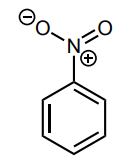
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EWG
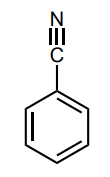
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EWG
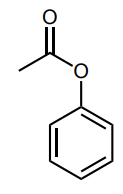
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EDG
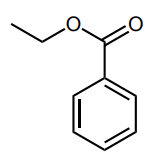
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EWG
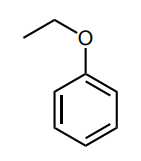
Assess whether the group is an electron-donating group (EDG) or electron-withdrawing group (EWG) to the benzene ring.
EDG
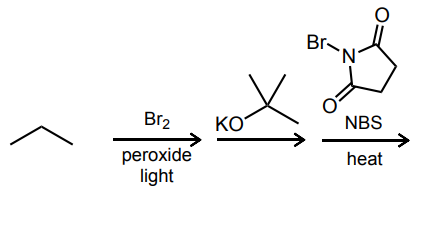
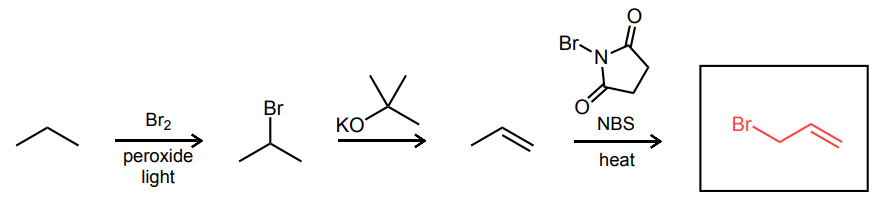
Predict the final product of each reaction sequence.
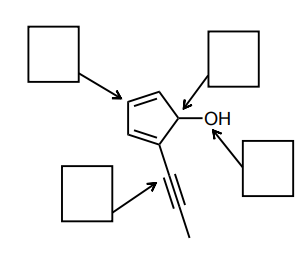
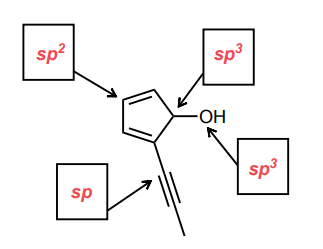
Assign the hybridization of each of the indicated atoms below.
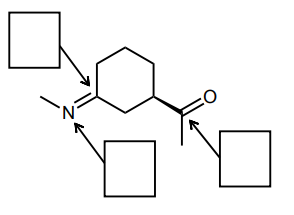
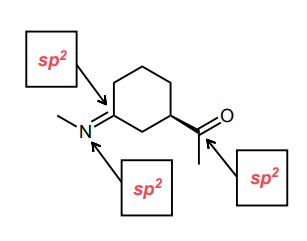
Assign the hybridization of each of the indicated atoms below.
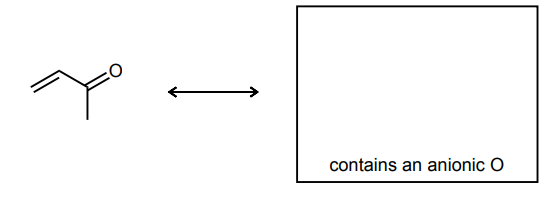
Draw the resonance structure of the species below and provide the curved-arrows needed to convert the structure shown to the new resonance structure.
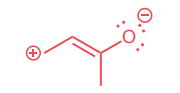
Draw the resonance structure of the species below and provide the curved-arrows needed to convert the structure shown to the new resonance structure.
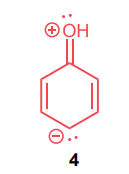
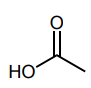
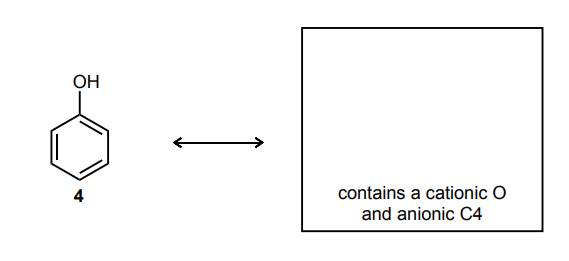
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Brønsted acid
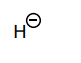
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Brønsted base
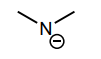
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Brønsted base
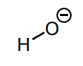
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Brønsted base, Nucleophile
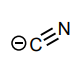
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Nucleophile
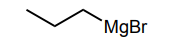
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Nucleophile
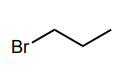
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Electrophile
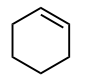
Classify each compound/species below with the most appropriate terms. (electrophile, nucleophile, Brønsted acid, Brønsted base)
Nucleophile, Brønsted base
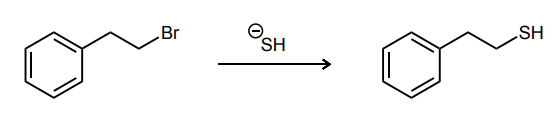
Draw the curved-arrow mechanism for the reaction shown below. Be sure to show any necessary intermediates and non-zero formal charges.
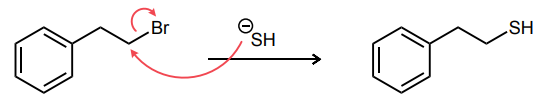
Draw the curved-arrow mechanism for the reaction shown below. Be sure to show any necessary intermediates and non-zero formal charges.
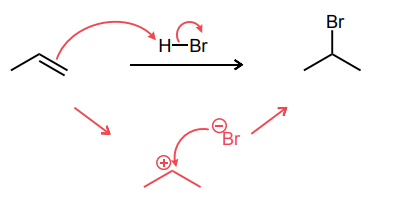
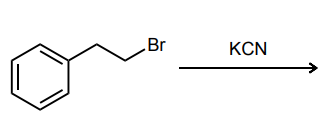
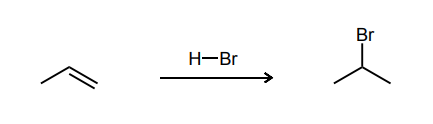
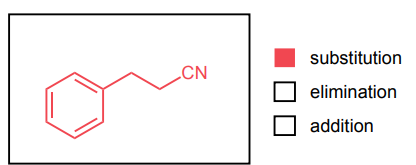
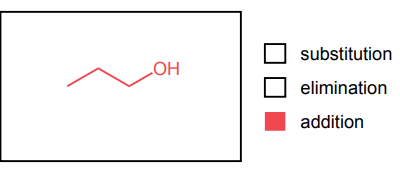
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
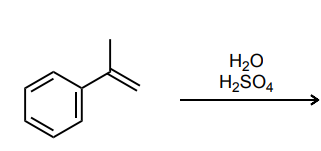
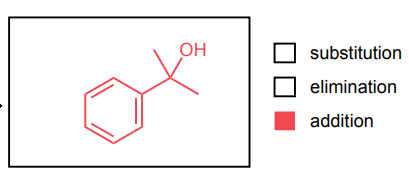
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
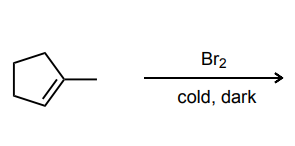
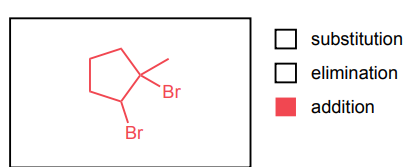
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
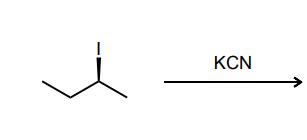
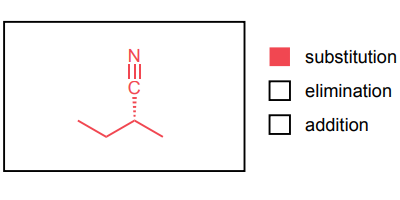
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
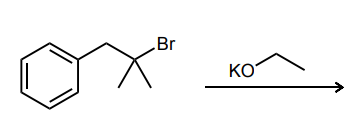
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
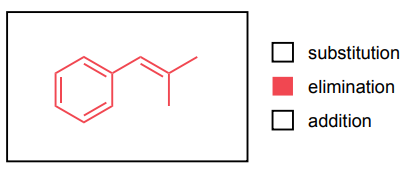
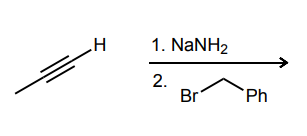
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
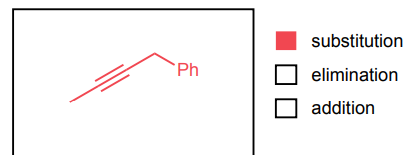
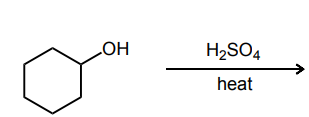
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
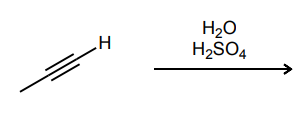
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
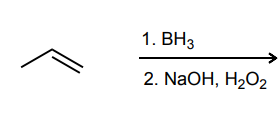
Provide the major product of each of the following reactions. Then classify each reaction as either a substitution reaction, an elimination reaction, or an addition reaction.
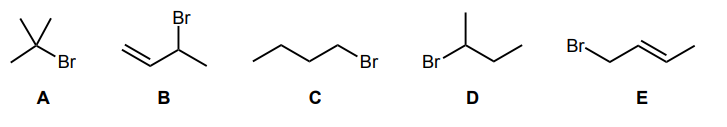
Rank the following electrophiles in order of increasing reactivity towards an SN2 reaction with KCN.
A, D, C, B, E
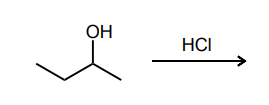
Draw the product(s) of the SN1 reaction below.
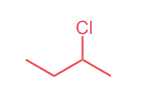
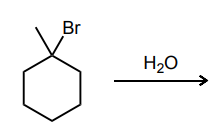
Draw the product(s) of the SN1 reaction below.
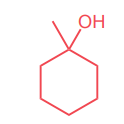
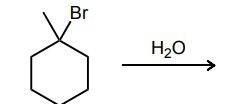
Draw the curved-arrow mechanism of the SN1 reaction below:
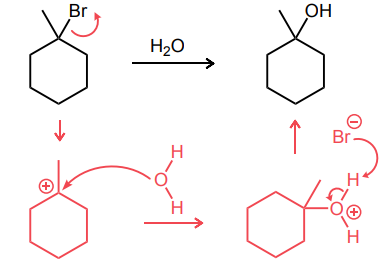
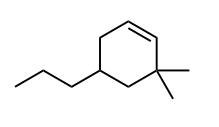
Draw in the C—H bond that is most likely to broken to form a carbon radical.
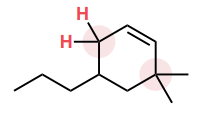
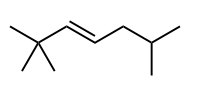
Draw in the C—H bond that is most likely to broken to form a carbon radical.
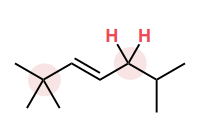
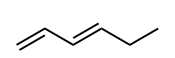
Draw in the C—H bond that is most likely to broken to form a carbon radical.
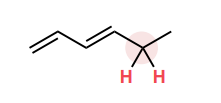
Which c-h bond is most likely to be broken to form a carbon radical?
The most stable c-h bond
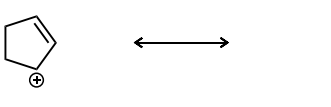
Draw the second resonance structure and show the curved arrows needed. Then, evaluate if the resonance structures are equivalent or not.
Equivalent
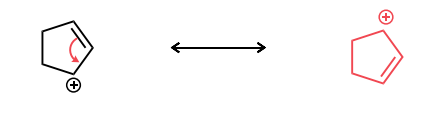
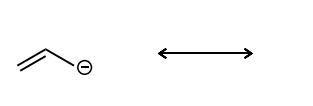
Draw the second resonance structure and show the curved arrows needed. Then, evaluate if the resonance structures are equivalent or not.
Equivalent
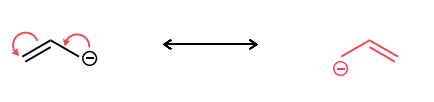
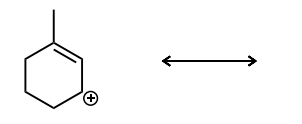
Draw the second resonance structure and show the curved arrows needed. Then, evaluate if the resonance structures are equivalent or not.
Not equivalent
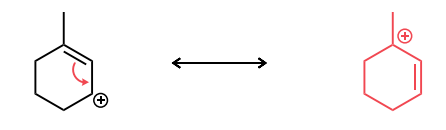
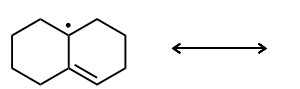
Draw the second resonance structure and show the curved arrows needed. Then, evaluate if the resonance structures are equivalent or not.
Not equivalent
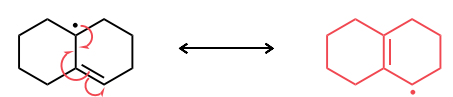
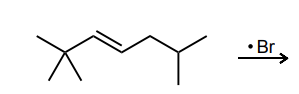
Draw the most stable radical that could be formed via an allylic C—H abstraction. Then, draw the resonance structure obtained via an allylic rearrangement of this radical species.
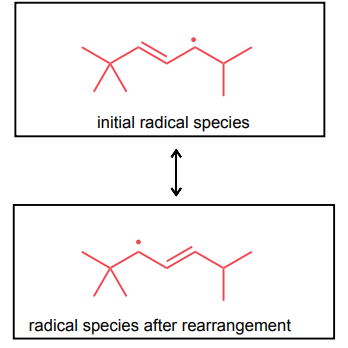
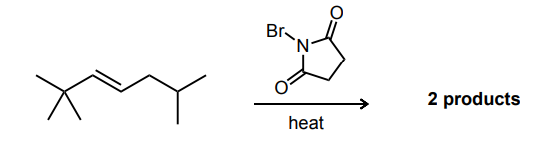
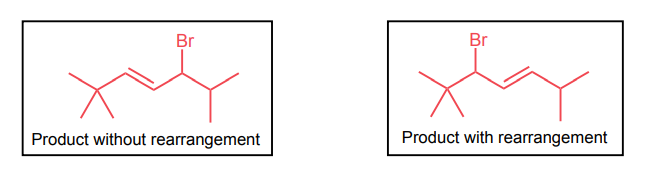
Draw the two brominated products obtained from this reaction.

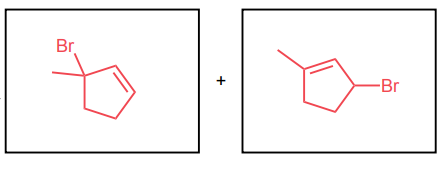
Draw the two potential products of each reaction below considering allylic rearrangements.
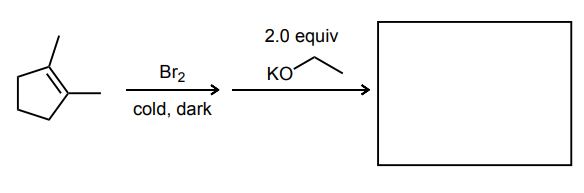
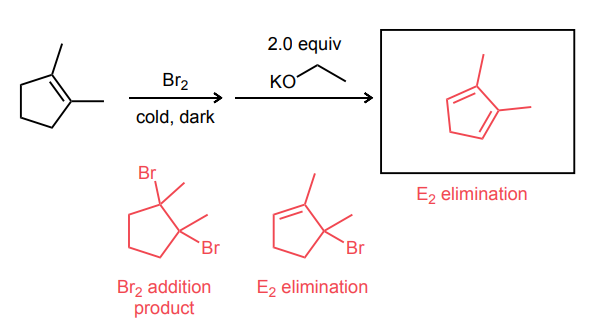
Provide the major product of the following guided multi-step synthesis reaction.
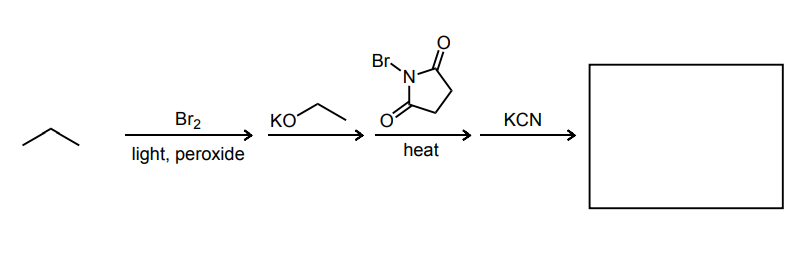
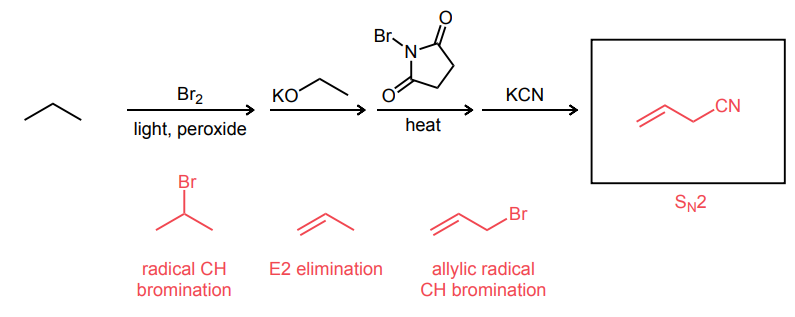
Provide the major product of the following guided multi-step synthesis reaction.
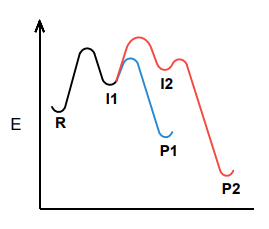
Which product, P1 or P2, is the kinetic product? Which is the thermodynamic product?
P1 is the kinetic PDT and P2 is the thermodynamic PDT
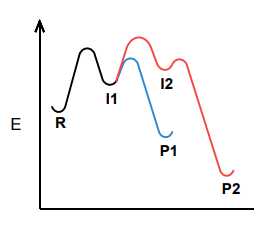
Which product, P1 or P2, would be formed faster?
P1
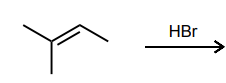
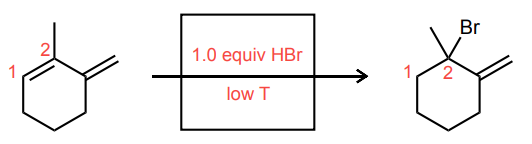
Draw the two possible carbocations that would be generated upon protonation with HBr. Then, identify the more stable carbocation, which is the major carbocation that forms in the reaction.
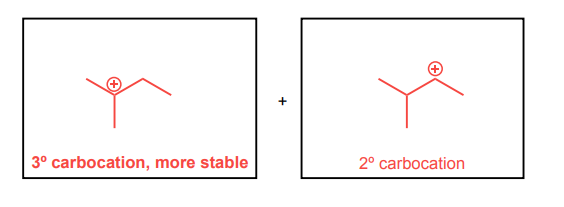
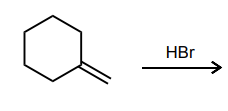
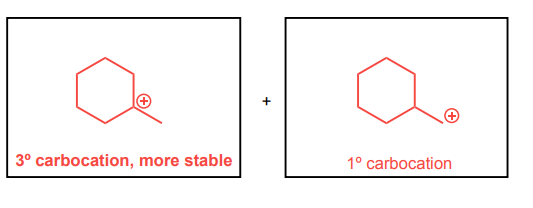
Draw the two possible carbocations that would be generated upon protonation with HBr. Then, identify the more stable carbocation, which is the major carbocation that forms in the reaction.
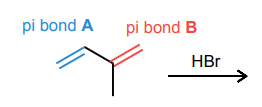
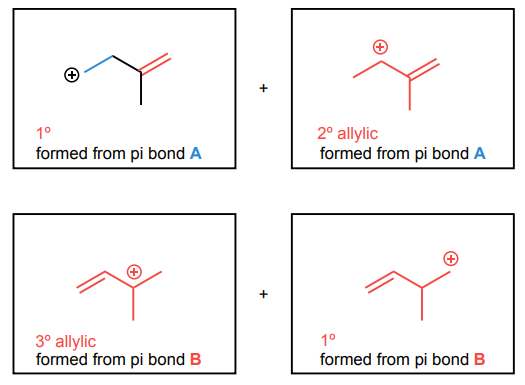
For the diene below, draw the four potential carbocations that could form via a protonation of each double bond in the molecule below.
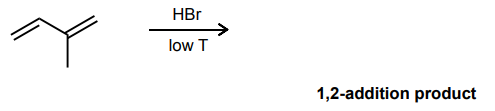
Draw the product and curved-arrow mechanism for the 1,2- addition below.
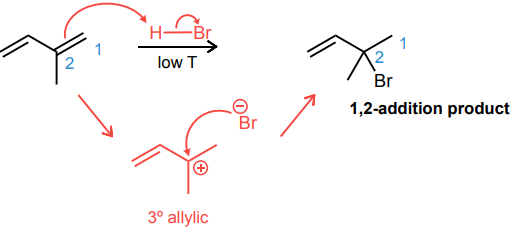
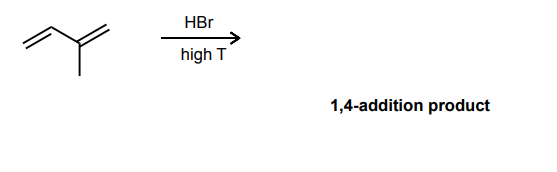
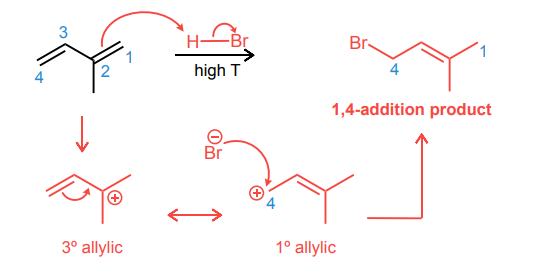
Draw the product and curved-arrow mechanism for the 1,4- addition below.

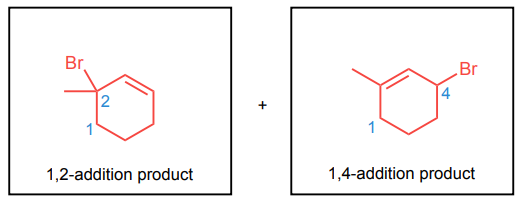
Draw the 1,2- and 1,4-addition products for the reactions below:
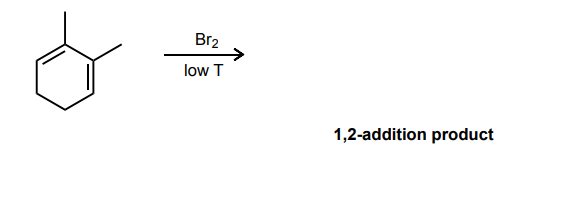
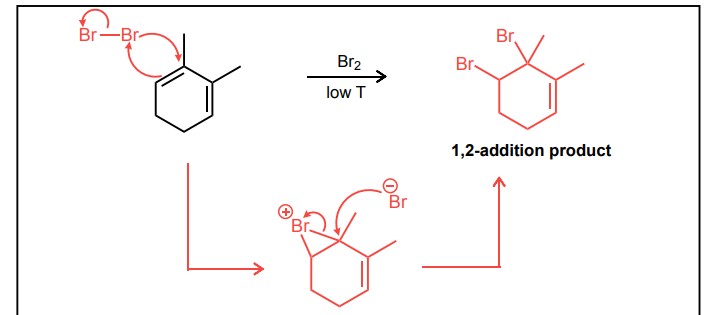
Draw the mechanism for the 1,2-bromination reaction of the conjugated diene below.
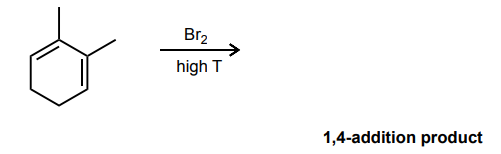
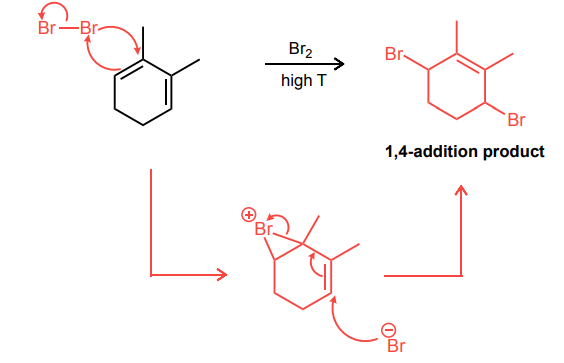
Draw the mechanism for the 1,4-bromination reaction of the conjugated diene below.
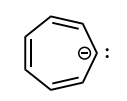
Identify whether the species is aromantic, anti-aromatic, or not aromatic. For any aromatic species, determine the number of pi electrons and the n value for Huckel’s rule. For non-aromatic or anti-aromatic species, select which requirement for aromaticity is not met: cyclic, planar, and/or Huckel’s # of pi electrons.
anti-aromatic: cyclic, planar, 8 pi electrons
Identify whether the species is aromantic, anti-aromatic, or not aromatic. For any aromatic species, determine the number of pi electrons and the n value for Huckel’s rule. For non-aromatic or anti-aromatic species, select which requirement for aromaticity is not met: cyclic, planar, and/or Huckel’s # of pi electrons.
not aromatic: cyclic, not planar
Identify whether the species is aromantic, anti-aromatic, or not aromatic. For any aromatic species, determine the number of pi electrons and the n value for Huckel’s rule. For non-aromatic or anti-aromatic species, select which requirement for aromaticity is not met: cyclic, planar, and/or Huckel’s # of pi electrons.
anti-aromatic: cyclic, planar, 4 pi electrons
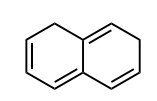
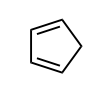
Identify whether the species is aromantic, anti-aromatic, or not aromatic. For any aromatic species, determine the number of pi electrons and the n value for Huckel’s rule. For non-aromatic or anti-aromatic species, select which requirement for aromaticity is not met: cyclic, planar, and/or Huckel’s # of pi electrons.
not aromatic: cyclic, not planar
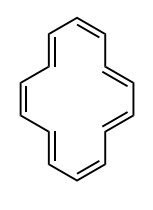
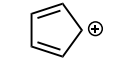
Identify whether the species is aromantic, anti-aromatic, or not aromatic. For any aromatic species, determine the number of pi electrons and the n value for Huckel’s rule. For non-aromatic or anti-aromatic species, select which requirement for aromaticity is not met: cyclic, planar, and/or Huckel’s # of pi electrons.
aromatic: cyclic, planar, 14 pi electrons
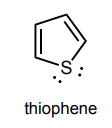
Circle any lone pairs that are involved in the aromatic system
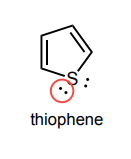
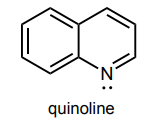
Circle any lone pairs that are involved in the aromatic system
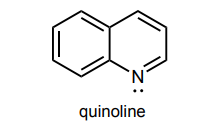
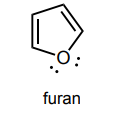
Circle any lone pairs that are involved in the aromatic system
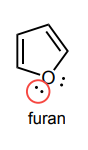
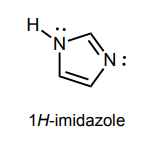
Circle any lone pairs that are involved in the aromatic system
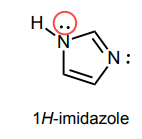
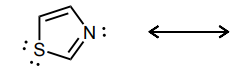
Draw atleast one resonance structure that shows the delocalization of a lone pair involved in the aromatic system
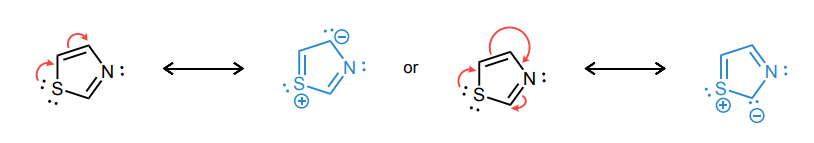
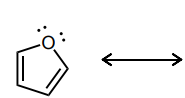
Draw atleast one resonance structure that shows the delocalization of a lone pair involved in the aromatic system

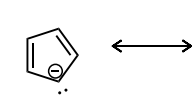
Draw atleast one resonance structure that shows the delocalization of a lone pair involved in the aromatic system
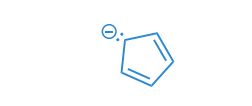
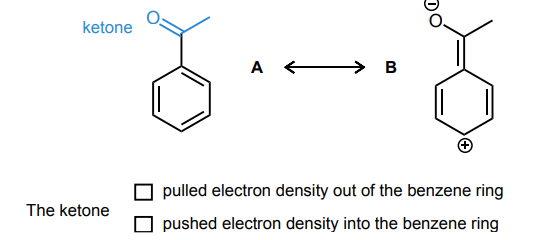
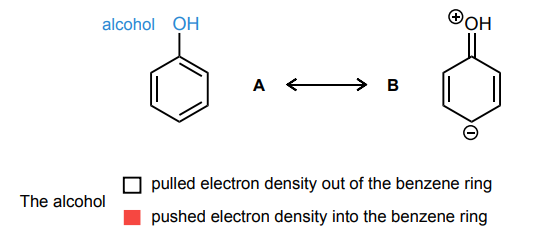
Draw the curved-arrows needed to convert resonance structure A into resonance structure B. Then, assess whether electrons have been pulled out from the benzene ring or if electrons have been pushed into the benzene ring
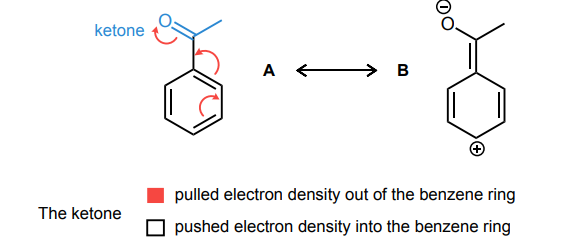
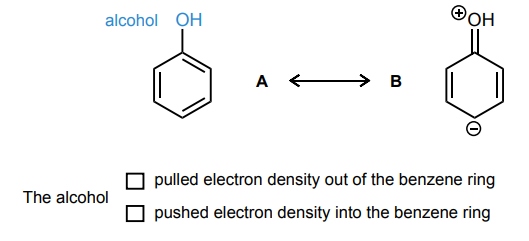
Draw the curved-arrows needed to convert resonance structure A into resonance structure B. Then, assess whether electrons have been pulled out from the benzene ring or if electrons have been pushed into the benzene ring