UNIT 4
1/180
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
181 Terms
Achiral
Doesn’t have a chiral centre
Chiral
Has a chiral centre
Chiral centre
Atom in a molecule bonded to 4 different atoms or groups
Enantiomer
Non superimposable mirror
image forms of each other that's rotate the plane of polarized light in opposite directions
Isomer
Say molecular formula different arrangement of atoms
Optical activity
Occurs in molecules that possess chiral centres
these molecules can rotate the plain and polarized light
Stereoisomerism
Occurs in isomers with the same molecular formula but different arrangements of atoms in space
Structural isomer
Compounds with the same molecular formula but different arrangements of atoms
Racemic mixture/ racemate
An equimolar mixture of both enantiomers that produce no overall rotation of plane polarized light
Stereoisomerism
E-Z isomer
shown in alkenes
E- same atoms opposite each other ( trans)
Z- next to each other (cis)
the pi bond restricts rotation about the double bond
If a compound contains 2 or more chiral centres
it is possible to have stereoisomers that are not Mirror image forms of each other [called diastereoisomers]
optical activity mechanism
Light consists of waves that are vibrating in all planes
if light is passed through a polarising filter, the light that emerges is vibrating in one plane only [called plane polarised light]
if a solution of an enantiomer is placed in a beam of plane polarised light, the beam is rotated
The extent to which the plane of polarised light is rotated depends on:
Particular enantiomer
concentration of the enantiomer In solution
Length of the tube containing the solution through which the light passes
frequency of the light source used and the temperature also needed to be considered
enantiomer may rotates clockwise or anticlockwise
equal amounts of each in solution produces no overall rotation since rotation effects cancel each other out
the mixture results from external compensation as the effect is caused by two different compounds.
The equimolar mixture is called a racemic mixture.
Ibuprofen
contains Kyle Centre and is sold as a racemic mixture
since biological systems usually respond to one in the required way rather than the other, there is a danger that the unwanted form could have serious side-effects
Separation of the two enantiomer is called resolution would be difficult and expensive
When ibuprofen is taken, one of the enantiomer is much more biologically active than the other, but the less active form is converted by an enzyme in the body into the other, more active enantiomer
Benzene
Relative mr- 78
C6H6
kekules suggestion on benzene
6 membered ring
containing alternating single and double C-C bond
Why kekules suggestion was incorrect
addition reaction
It should react as an alkene and easily undergo addition reactions (eg reaction with aqueous bromine where bromine is decolorized) However it doesn't react this way
to explain this, he suggested benzene had two forms in which one form changed to the other so quickly that an approaching molecule would it have time to react by addition ( double a single bonds alternate)
Why kekules suggestion was incorrect
bond length
if Benzene had two forms, they would be two different bond lengths between carbon atoms in the molecule
X-ray crystallography showed each C-C bond was the same length at 0.140 nm
this distance between C-C double bond at 0.135 nm and a carbon carbon single bond at 0.147 nm
Benzene Enthalpy
When cyclohexane is hydrogenated the energy released is 120 kJ mol-1
If benzene existed as Kekules structure and all three double bonds are hydrogenated than the entropy change would be through -360 kJmol-1
but when benzene is fully hydrogenated, the entropy change is -208 kJmol-1
152 difference, (resonance energy) suggests benzene is more stable than kekules structure and suggests it is incorrect
Bonding in benzene and ethene
remaining
orbital
benzene is a planer molecule and the angles between three adjacent carbon atoms are 120°.
Ethan C2H4, Each carbon atom is bonded to 2 hydrogen atoms and a carbon atom by sigma bonds
remaining outer P electrons of each carbon atom overlap above and below the plane of a molecule giving a localised pi orbital
delocalised bonding of benzene
fourth outer shell electron of c
each carbon atom is bonded to 2 other carbon atoms and a hydrogen atom by sigma bonds
Fourth outer shell electron of C is in the 2P orbital above and below the plane of the C atoms. These orbitals overlap to give a delocalised electron structure above and below the plane of C atoms.
if benzene underwent addition reaction
process with disrupt the staple delocalised electron system and the resulting product would be less stable
needs higher temperatures and a nickel or platinum catalyst
will also react with chlorine in addition to give hexachlorocyclohexane C6H6CL6. radical reaction that needs bright sunlight to be effective
Mechanism of electrophilic substitution
ring of
benzene has a delocalised ring of electrons above and below the plane of the carbon atom. This area of high electron density makes it susceptible to attack by an electrophile.
electrophile
an electron deficient species that can accept a loan pair of electrons
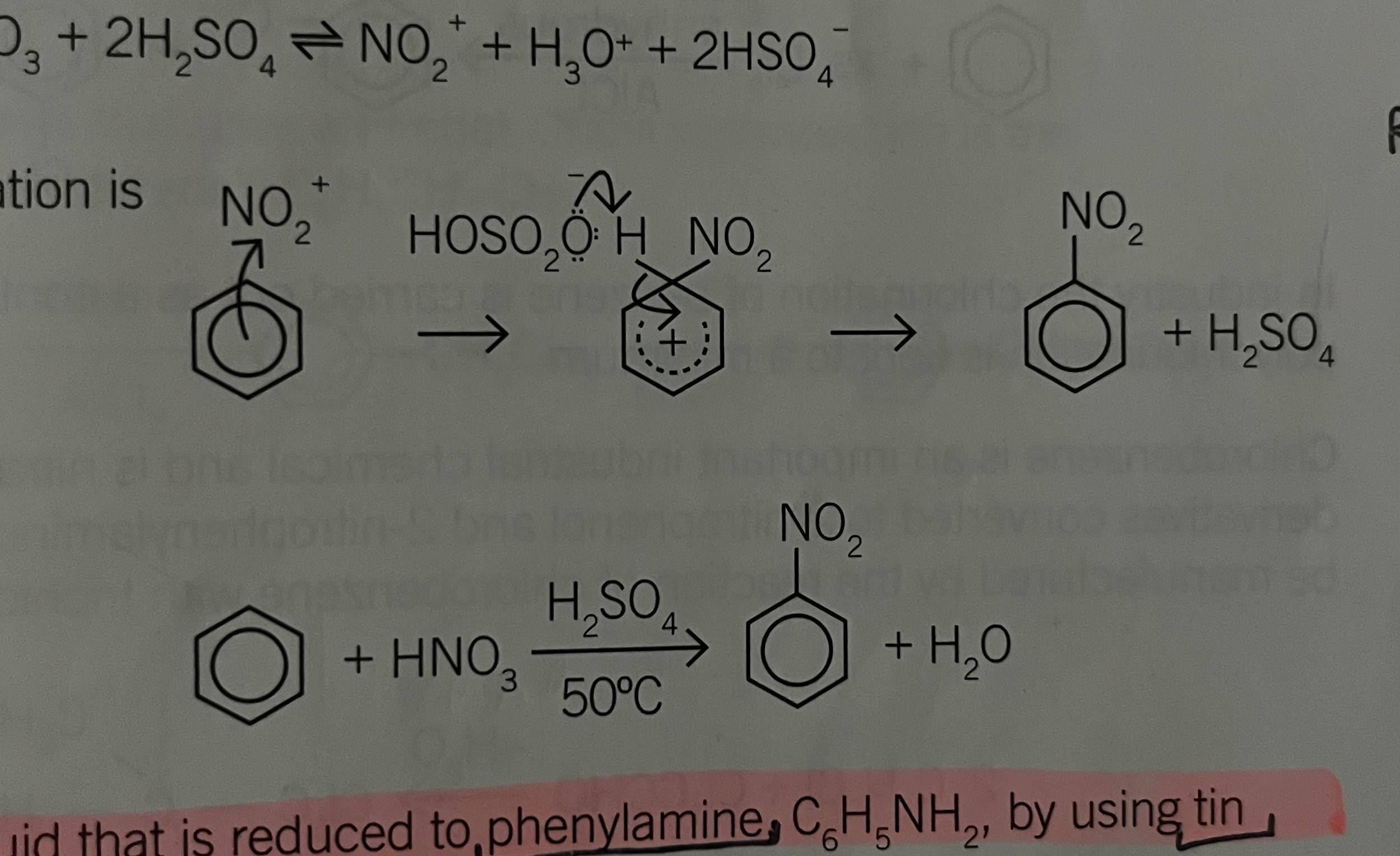
Nitration
HNO3 + 2H2SO4 = NO2+ + H3O- + 2HSO4-
nitrobenzene is yellow liquid that is reduced to phenyl amine C6H5NH2 by using tin metal and hydrochloric acid
If the temperature of nitration exceeds 50°C, other branches of nitro groups are produced
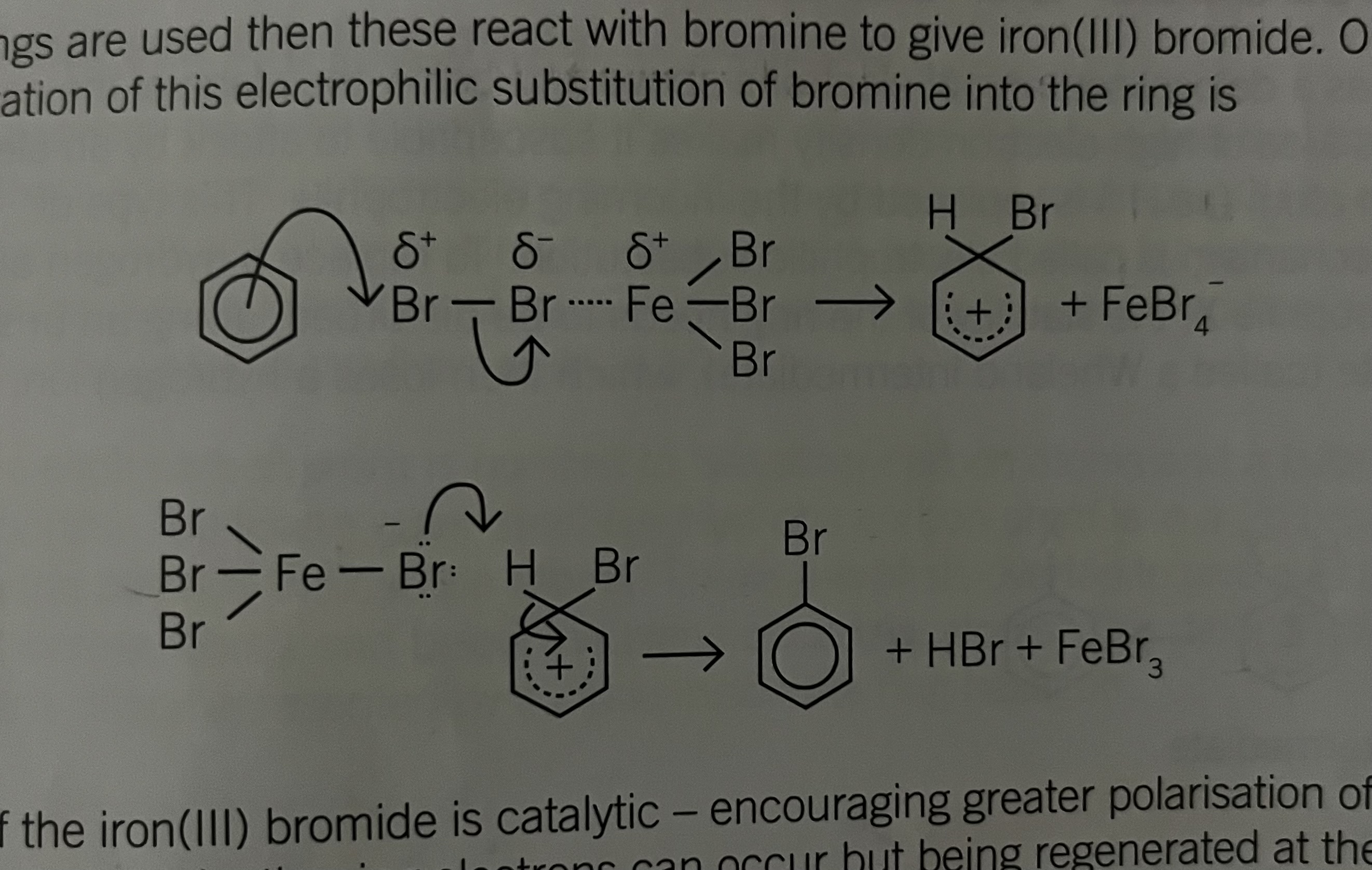
Halogenation- Bromine
bromine and benzene do not react together unless a catalyst is used eg. Fe/FeBr3/AlBr3
Pi Electron System is not sufficiently nucleophilic to polarise the bromine In the presence of iron bromide, the colonisation of bromine molecule is more pronounced
Br-Br is polarised, delta + and - by the pi electron system
polarised by the eletron deficient iron in catalyst

Halogentaion bromine mechanism
The role of the iron bromide is catalytic encouraging greater polarisation of the Br-Br bond so that attack by the ring electrons can occur but being regenerated at the end of the reaction
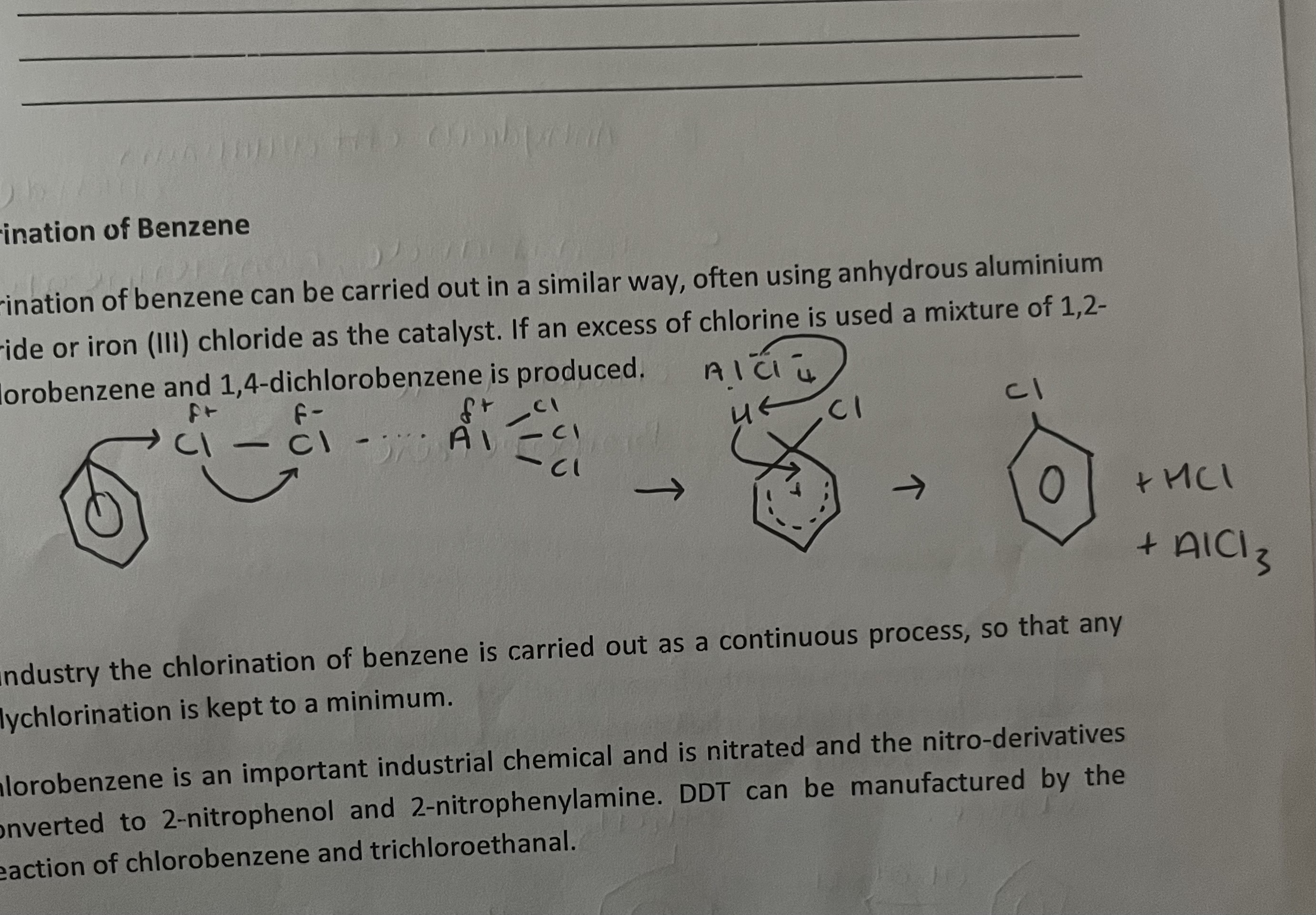
chlorination of benzene
anhydrous AlCl3/ FeCl3 as the catalyst
in industry- used as a continuous process, so any polychlorination is kept to a minimum
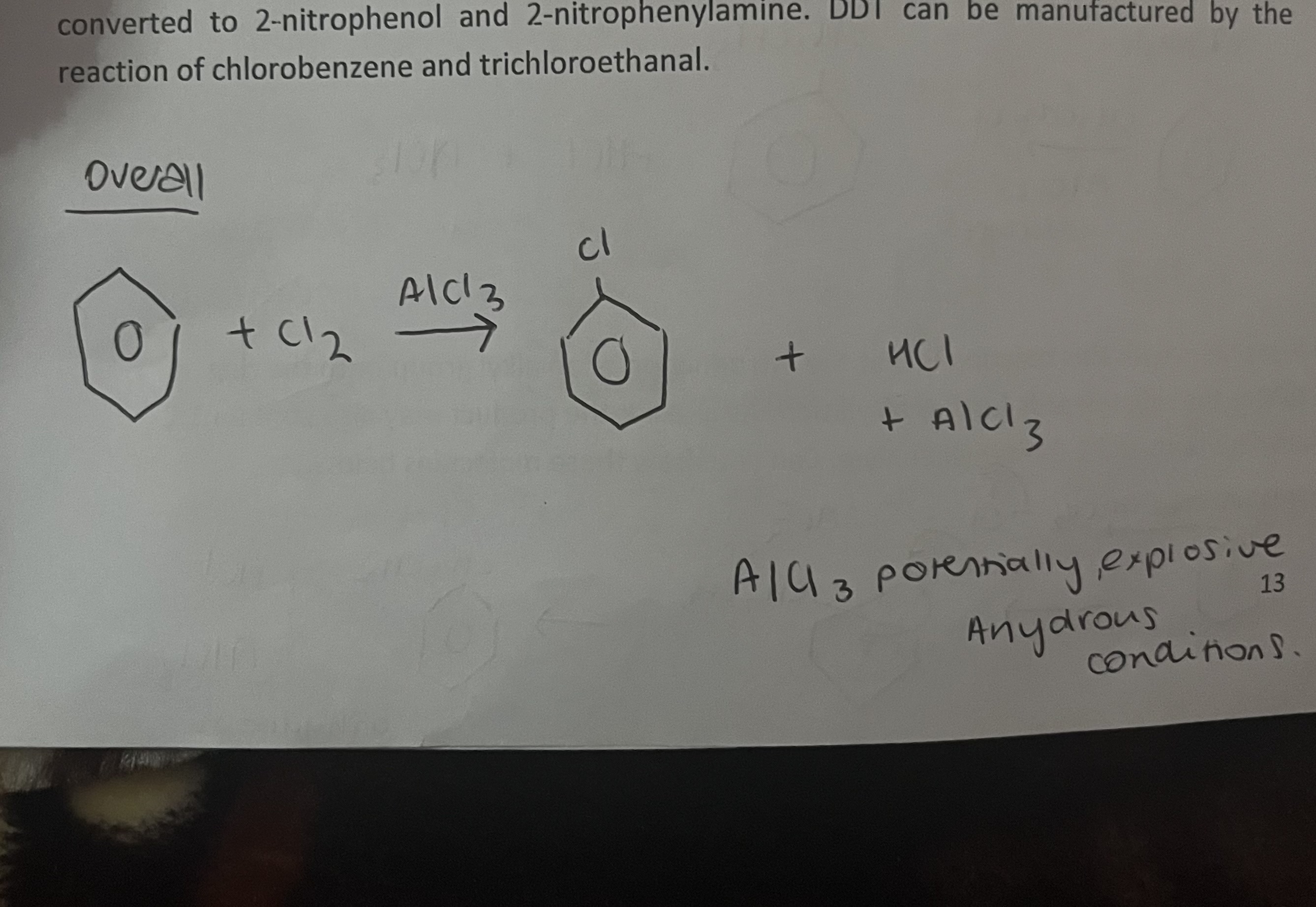
chloro benzene- important industrial chemical
is nitrated and nitro- derivatives converted to 2-nitrophenol and 2-nitrophenylamine. Insectide DDT can be manufactured by the reaction of chlorobenzene with tricbloroethanl
DDT
insecticide
problems related to toxicity
problem with bio accumulation, persistence in the environment and presence in the food chain
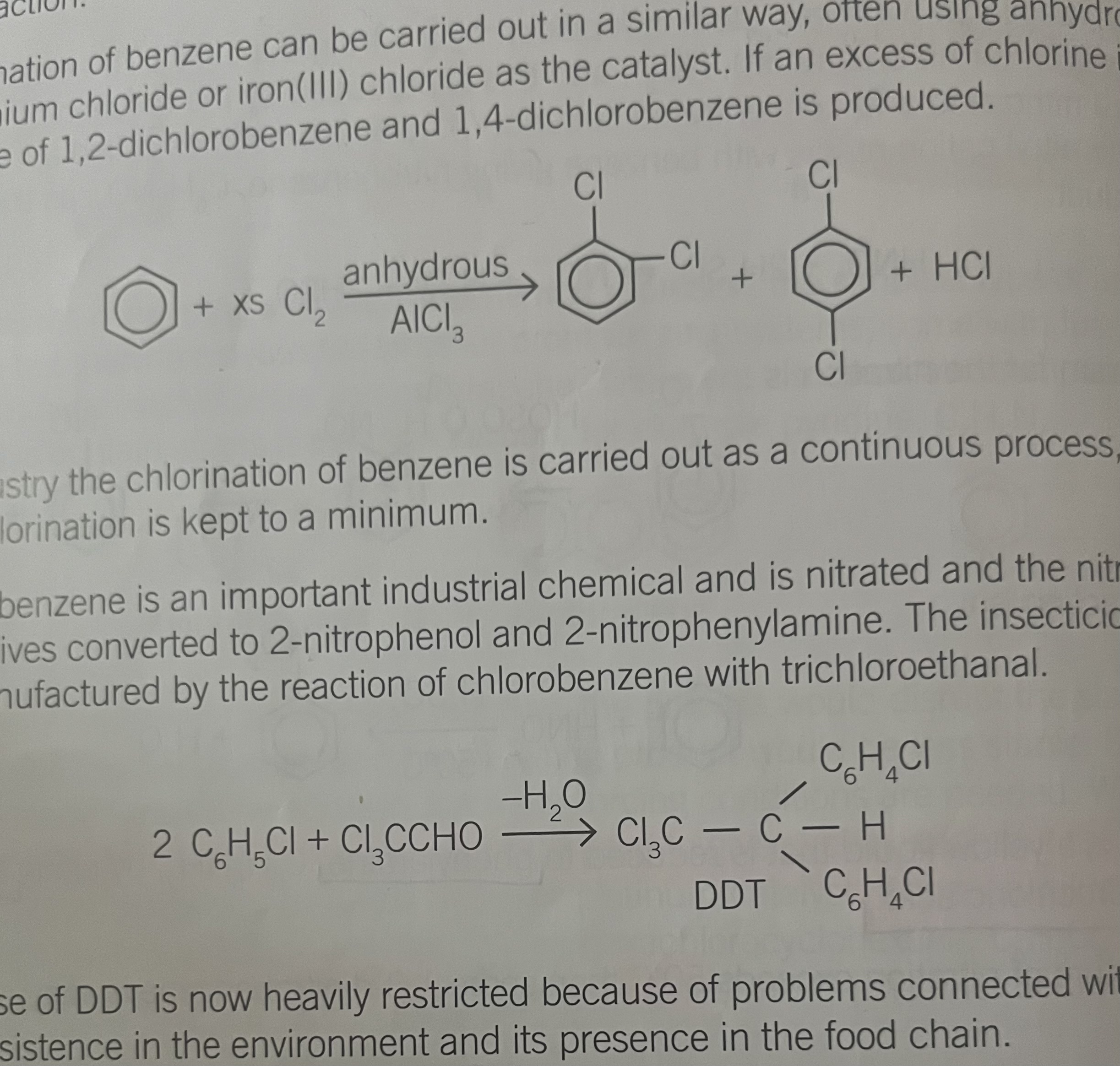
Friedel- crafts alkylation
method to produce new C-C bonds, giving alykl derivatives of benzene eg. Methylbenzene
anhydrous AlCl3 as a catylst, HCl as product
Friedel- crafts alkylation
mechanism
Produces new carbon carbon double bond, giving alkyl derivatives of benzene
anhydrous aluminium chloride as a catalyst
1-chlorohexane with aq NaOH on heating under reflux
gives hexan-1-ol
CH2(CH2)4CH2Cl + NaOH —> CH3(CH2)4CH2OH + NaCl
Mechanism:
Benzene tend to react by electrophillic substitution, has little tendency to react with nucleophiles, as these would be replelled by the stable Pi system of electrons
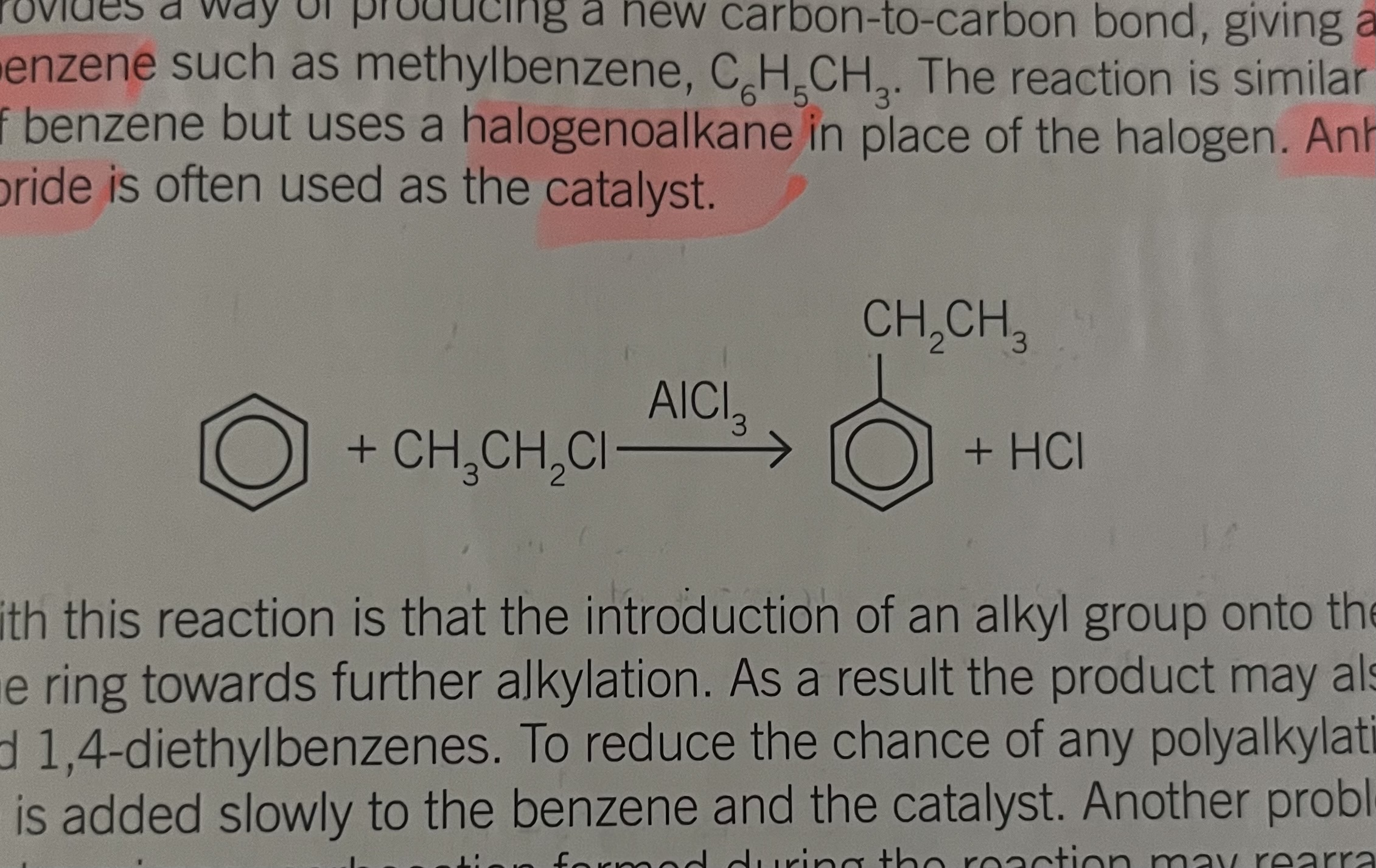
strength in C-Cl bonds in chlorobenzene and chloroalkane
the stronger and shorter bond betweenC-Cl in chlrobenzene results from a non bonding p electrons pair on chlorine overlapping with he ring pi system of electrons.
The resulting bond needs much more energy to be broken and is stronger than an aliphahtic C-Cl bond
Forcing conditions are therefore needed to produce phenol from chlorbenzene
producing phenol equation
this reaction is not an environmentally viable method of producing phenol commercially. In industry phenol Talk is generally made from 1-methyl ethylbenzene This method has the advantage of Propanone is a useful co product of the reaction.
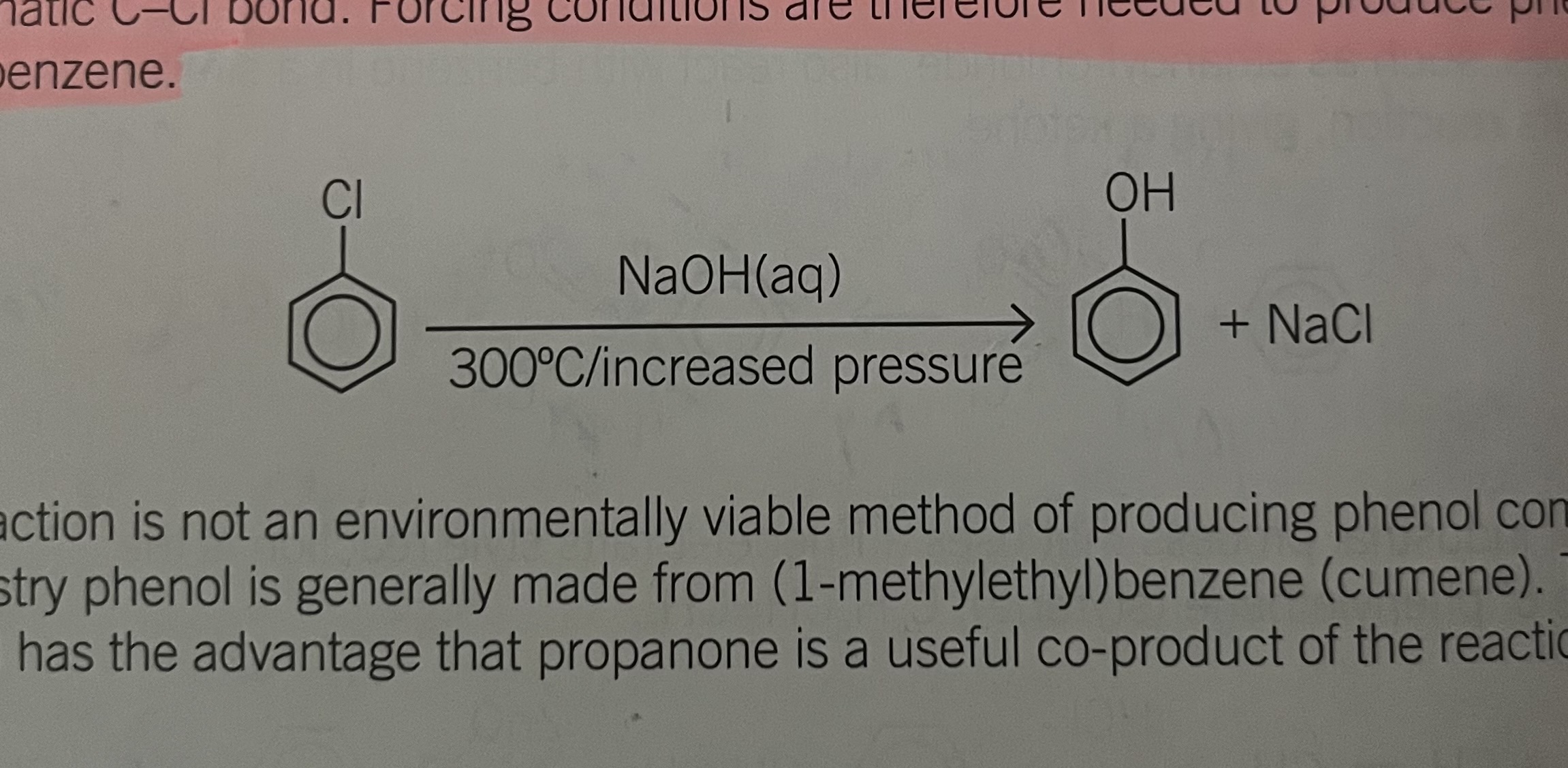
problems with alkylation
introduction of an alkyl group onto the ring activates the ring towards alkylation
as a result the product may contain 1,2- and 1, 4-diethyl benzene. to reduce polyakylation, the halogen alkane is added slowly to the benzene and the catalyst
another problem is that a primary carbocation formed during the reaction may rearrange to a secondary
reaction of 1- chloropropane
with benzene gives mainly 1-methyl ethyl benzene rather than 1- propylbenzene as the organic product
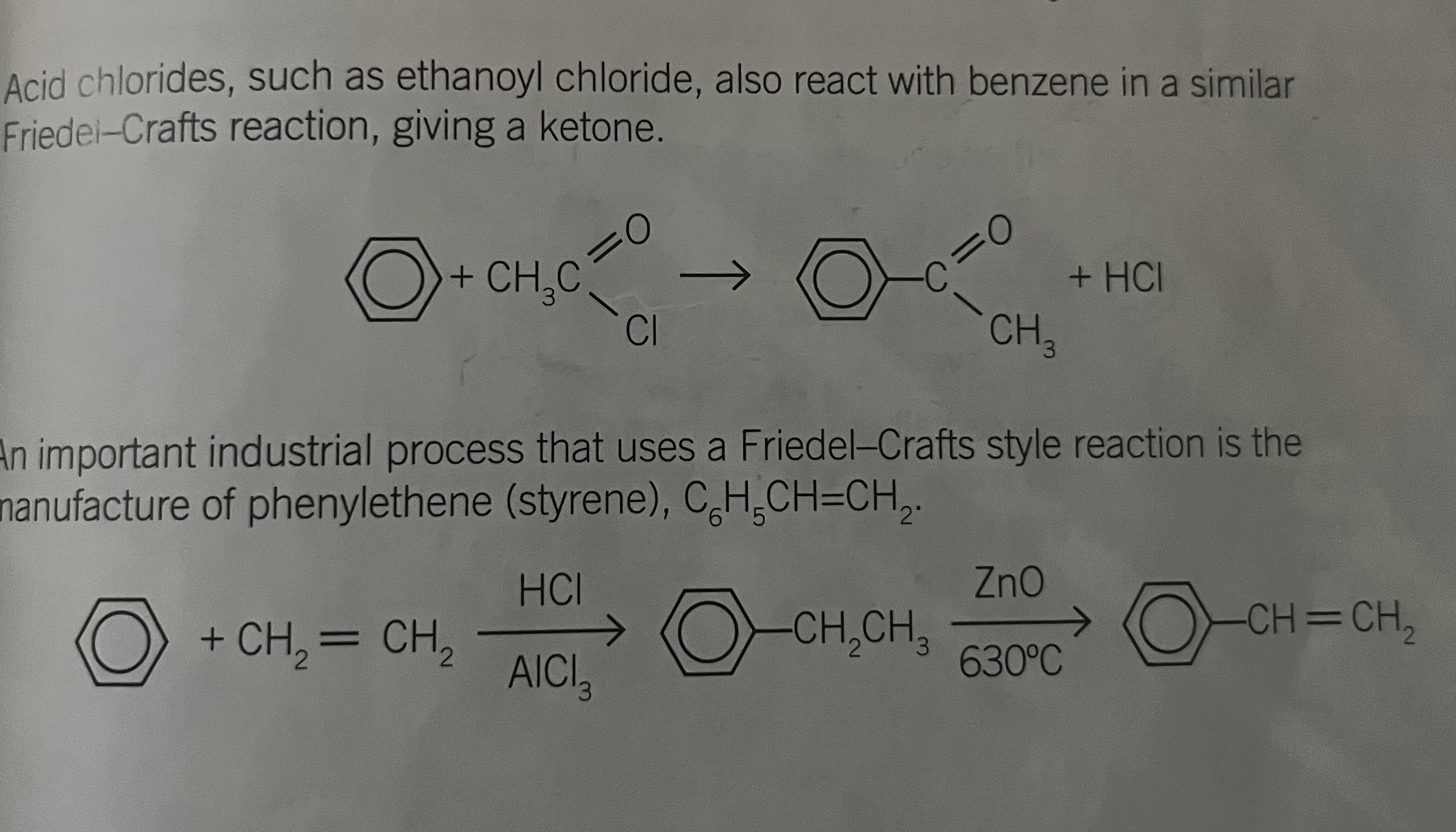
acids chlorides with benzene - give a ketone
manufacture of phenyl ethene
EG ethanoyl chloride with benzene
important industrial process used Friedel crafts sty!e reaction
Nucleophiles
ions or compounds possessing a lone pair of electrons that can seek out a relatively positives site
donates a pair
Eg. -OH, -CN, NH2
Primary, secondary and teritary alcohols
P- Only one alkyl group attached to the carbon hydroxide group
S- 2
T- 3
Electronegativity
Ability of an atom in a chemical bond to polarise electron density towards itself
Difference in boiling points between alcohols and alkanes
set up between
In alcohol there are hydrogen bonds set up between the slightly positive hydrogen and lone pairs on oxygen in other molecules
in alkanes the only intermolecular forces are vander waals dispersion forces hydrogen bonds are much stronger and take more energy to separate alcohol molecules
Forming primary and secondary alcohols from halagenoalkanes
refluxing
Carried out by refluxing together the halogenoalkane and an aqueous solution of an alkali (Na or KOH)
during reflux the liquid is evaporated, cooled and condensed to continue the reaction
separated at the end of the reaction by fractional distillation from diff boiling points
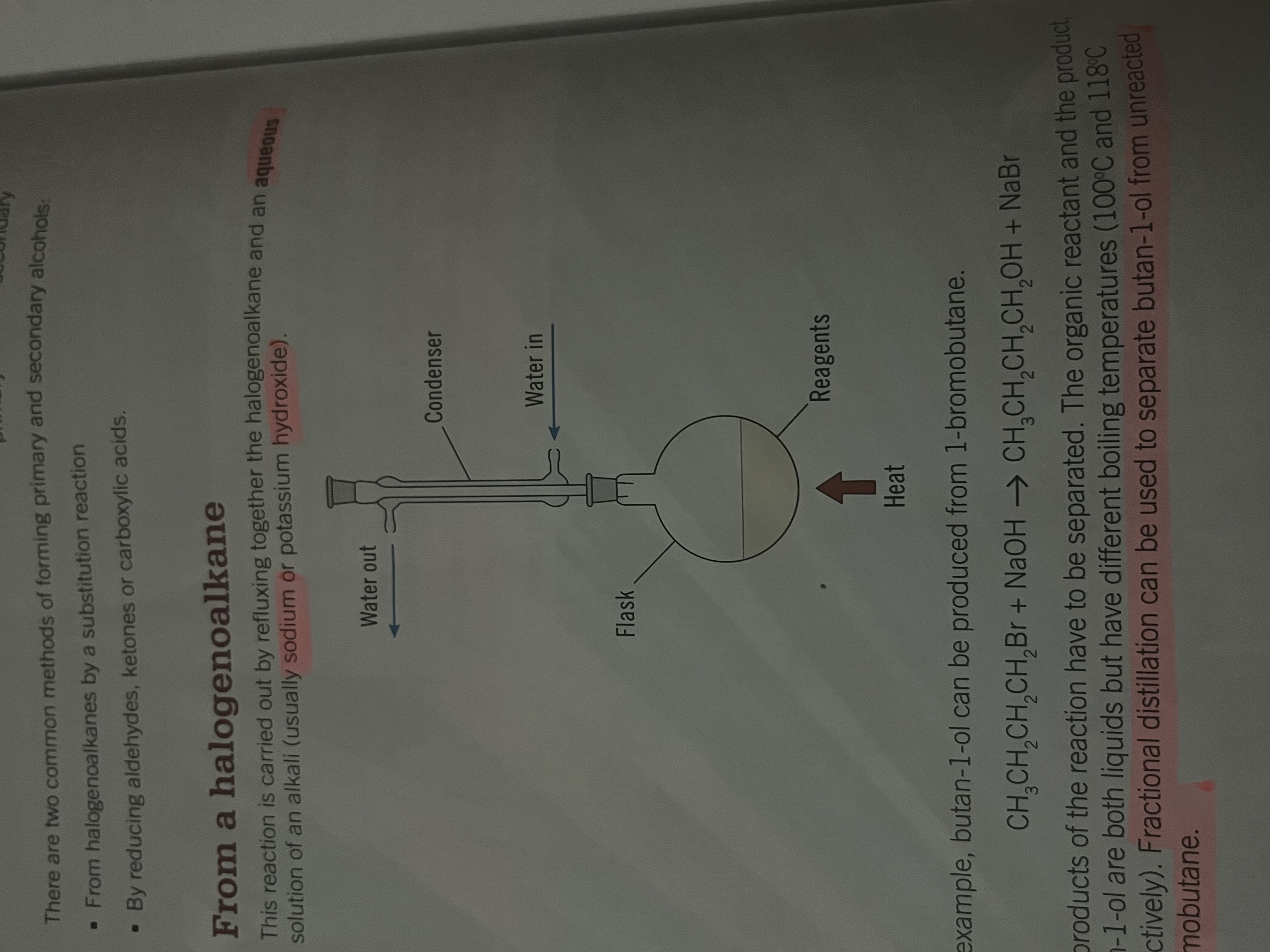
Forming a primary alcohol Eg. Butan-1-ol from 1-bromobutane
substitution
CH3CH2CH2CH2Br + NaOH → CH3CH2CH2CH2OH + NaBr
nucleophilic substitution where the hydroxide ion acts as the nucleophile and attacks the relatively positive carbon atom on the C-Br bond

secondary alcohol formation example
Rate of hydrolysis for alcohols from halogenoalkanes
C—I > C—Br > C—Cl
yield of primary alcohols is usually higher than the yield of secondary alcohols
when producing secondary alcohols a alkene can be formed as a side product
greater yields of alkenes are produced if high concentrations of the alkali are used or if an ethanolic solution of the alkali
Formation of alcohols from the reduction of a carbonyl compound
aldehydes and ketones
Both aldehydes and ketones can be reduced to primary and secondary alcohols by using an aqueous solution of sodium tetrahydridoboarate (NaBH4)
use [H] to represent a reducing agent
![<p>Both aldehydes and ketones can be reduced to primary and secondary alcohols by using an aqueous solution of sodium tetrahydridoboarate (NaBH4)</p><p>use [H] to represent a reducing agent</p>](https://knowt-user-attachments.s3.amazonaws.com/9ed5fb03-1dea-4e74-9885-b7093c9be509.jpg)
Reducing carboxylic acids
dissolved in
Sodium tetrahydridoboarate is not powerful enough to reduce carboxylic acids and the stronger reducing agent lithiumtetrahydridoaluminate (LiAlH4)
Dissolved in ethoxyethane

Reactions with the reducing agents
excess
Sodium tetrahydridor borate is a much safer reaction as an excess of lithium tetrahydridaluminate is difficult to dispose and the solvent ethoxyethane is very flammable
To obtain a good yield of alcohol
the organic solvent
An access of the reducing agent is used
the excess reducing agent can be removed by adding a dilute acid
the organic products needs to be separated from the aqueous mixture
carried out using a separating funnel if the alcohol produced is immisciple with water or separated by adding an organic solvent such as ethoxyethane by the technique of solvent extraction
Reaction of primary and secondary alcohols with hydrogen halides
Halogenoalkanes are produced by reacting the alcohols with hydrogen halides
the reactions are generally slow and reversible often giving poor yields

Chlorination
Passes hydrogen chloride gas into the alcohol in the presence of anhydrous zinc chloride, acting as a catalyst

Chlorination using phosphorus chloride (V)
Room temperature
one problem with this reaction is that phosphorus oxide trichloride (POCl3) is a liquid and needs to be removed from the reaction mixture. if the halogenoalkane has a similar boiling temperature to POCl3 then separation becomes difficult

Chlorination using sulphur (VI) oxide dichloride SOCl2
An advantage to this method is that the co products sulphur oxide and hydrogen chloride are both gaseous and are easily lost from the reaction mixture, ensuring easier separation
Bromination
‘in situ’ reaction: potassium bromide and 50% sulfuric (VI) acid is heated
the sulfuric acid protonates the alcohol and this then reacts with the bromide ions from the potassium bromide
CH3CH2CH2CH2OH + KBr + H2SO4 → CH3CH2CH2CH2Br + KHSO4 + H2O
Iodination
warm d
Warm dump red phosphorus and iodine together to form phosphorus iodide (III) PI3
2P + 3I2 → 2Pl3
3CH3CH2CH2OH + PI3→ 3CH3CH2CH2I + H3PO3
Reaction with carboxylic acids
Primary and secondary alcohols react with carboxylic acids to give esters
Alcohol + COOH → ester + H2O
the reaction is reversible and eventually the mixture will reach a position of equilibrium
To increase the yield of Esters
distilled
Little concentrated sulfuric (VI) acid is added to the mixture of the alcohol and the carboxylic acid, the mixture is heated under reflux
the products can then be distilled and the Ester collected at its boiling temperature
the distillate generally consists of the Ester and water together with a little unreacted alcohol and carboxylic acid. Many esters are immiscible with water and the distillate often consists of two layers
Separating funnel used to extract the Ester
dried
Then shaken with sodium hydrogen carbonate solution to remove any remaining carboxylic acid
Ester is then dried with anhydrous calcium chloride which reacts with any remaining alcohols
it can then be redistilled to give a pure product
Reactions with ethanoyl chloride
Alcohol reacts rapidly with ethanyol chloride giving an ester. During this reaction misty fumes of hydrogen chloride are seen.
Gives a better yield of an Ester than using a carboxylic acid as the reaction is not reversible
however the cost of acid chloride means this is not a cost efficient process in industry

Reactivity of phenols compared to alcohols
lone pair overlap
extended delo
Reactivity is very different
One of the lone pairs of oxygen atoms overlap with the delocalized Pi system to form a more extended delocalized system.
As a result the C—O bonds in phenols is shorter and stronger than in alcohols
making C—O bond fission in phenols harder than co-bond in alcohols
the extended delocalization creates a higher electron density in the Ring and makes the ring structure more susceptible to attack by electrophiles
Acidity of phenol
Phenol is a very weak acid and the position of the equilibrium lies to the left
phenol can lose a hydrogen ion because the phenoxide ion formed is stabilized to some extent. The negative charge on the oxygen atom is delocalized around the ring
The more stable the ion the more likely it is to form
Why is phenol only a weak acid
one of the lone
One of the lone pairs on the oxygen atom overlap with the delocalized electrons on the benzene ring
this overlap leads to a delocalization which extends from then ring out over the oxygen atom resulting in the negative charge no longer entirely localised on the oxygen, but is spread out around the whole ion
spreading the charge around makes the ion more stable
oxygen is the most electronegative element in the ion and the delocalized electron will be drawn towards it. This means that they will be still a lot of charge around the oxygen
which will tend to attract the hydrogen ions back again
Phenol with the sodium hydroxide
gives a
Gives a colourless solution containing sodium phenoxide- showing it must be acidic
hydrogen ion has been removed by the strongly basic hydroxide ion in the solution
this shows phenol reacting as an acid losing a proton to the aqueous hydroxide ion
The presence of substituents on the benzene ring affects the acidity of phenol

phenol tion with sodium carbonate/ sodium hydrogen carbonate
Phenol is not strong enough to react to produce carbon dioxide. No bubbles are produced
shows it's only a weak acid
Phenol with metallic sodium
slow
Slow reaction because phenol is such a weak acid
phenol is warmed in a dry tube until it is molten and a small piece of sodium is added there is some fizzing as hydrogen gases given off
mixture is left in the tube will contain sodium phenoxide
Directing effect of the -OH group
Has more activating effect on some positions around the ring than others
incoming groups will go into these positions much faster
has a 2,4,6 directing effect, incoming groups will tend to go into the 2, 4 or 6 position
Phenol with bromine
presence activate
Presence of an OH group bonded directly to a benzene ring will activate the ring to attack by electrophiles causing directing effect
when female reacts with bromine the increased electron density in the ring polarises the bromine molecules
Aqueous bromine reacts with phenol to produce a white precipitate of 2,4,6- tribromophenol
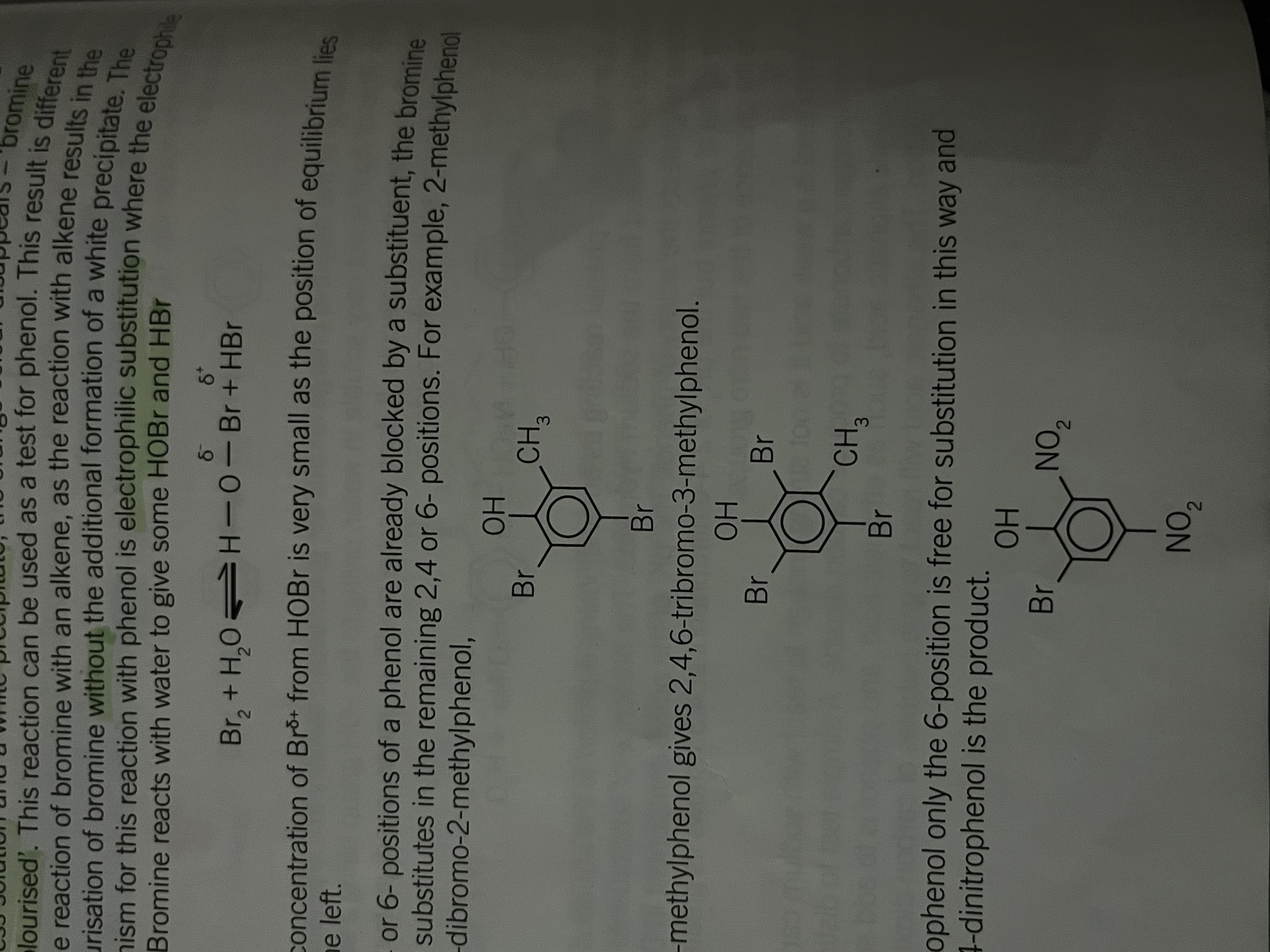
Colour change in the bromine test
Aqueous bromine= orange
products= colourless solution and a white precipitate
this means the orange colour disappears since the bromine is decolorized
How is reaction with bromine with phenol different to an alkene
In an alkene reaction, it will result in the decolorization of bromine without the additional formation of a white precipitate
mechanism for this reaction is electrophilic substitution. Electrophile is Br+
Bromine reaction with water
gives HOBr and HBr
Br2 + H2O = H-O-Br + HBr
concentration of Br+ from HOBr is very small as a position of the equilibrium lies well to the left
Reaction of Phenols and ethanoyl chloride
react as
Alcohols and phenols can react as nucleophiles by the use of their oxygen lone pairs
the delocalisation of an electron pair from the oxygen atom in a phenol means it is more difficult for a phenol to react as a nucleophile
as a result carboxylic acids are not suitable reagents to make an ester with phenol
Phenol and ethanoyl chloride
Slow at room temp
Phenol and a pyridine ( a base)
Can be added to speed up the reaction
the pyrdine react with the hydrogen chloride co-product to give pyridinium chloride (C5H5NH+Cl-)
Phenol and ethanoic anhydride
Ethanoyl chloride is an expensive reagent and ethanoic anhydride is used in preference
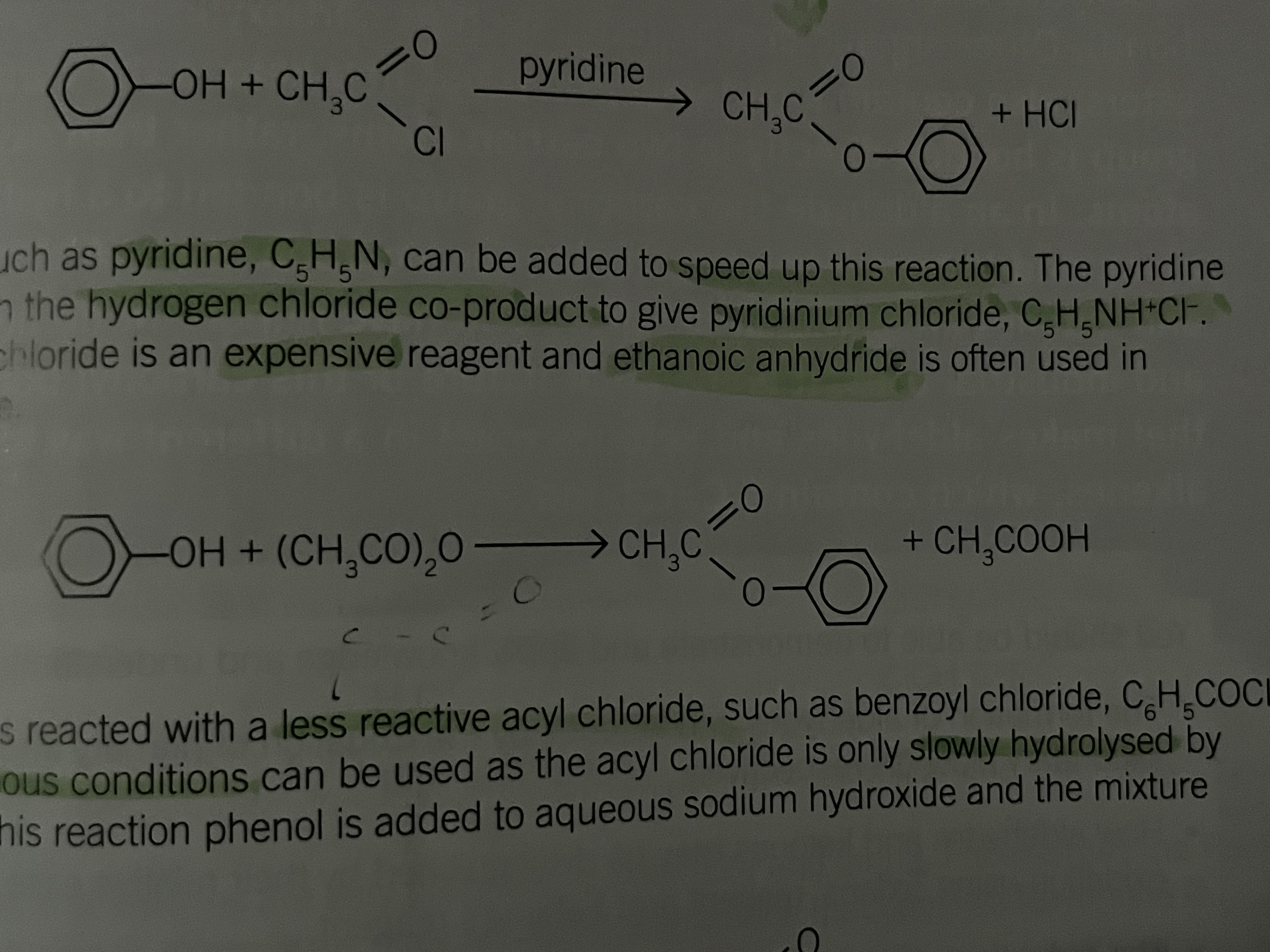
Phenol and acyl chloride
Less reactive
aqueous conditions can be used as the acyl chloride is only slowly hydrolysed by water
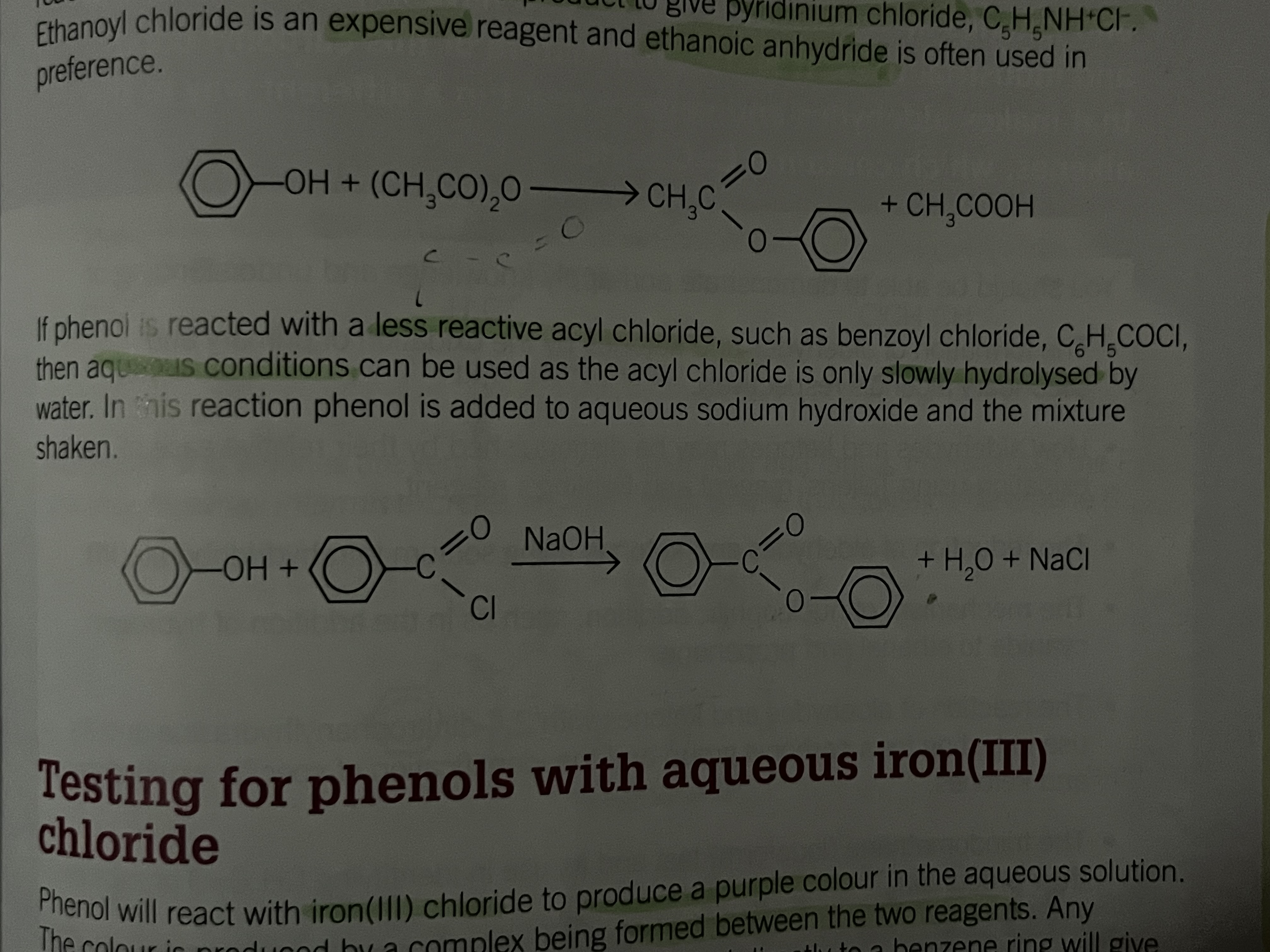
Test for phenols
Phenol will react with iron (lll) chloride to produce a purple color in the aqueous solution
the colour is produced by a complex being formed between the two reagents
group in aldehydes and ketones
Both have a polar C=O group
this occurs because oxygen in this bond has a greater electronegativity, leaving the carbon atom slightly electron deficient
the double bond comprises a ó bond and a pi bond above and below the plane of the sigma bond caused by p-p orbitals overlapping
Aldehyde
Will have at least one hydrogen atom
C-R-H=O
Ketone
Has a carbonyl carbon atom directly bonded to two other carbon atoms
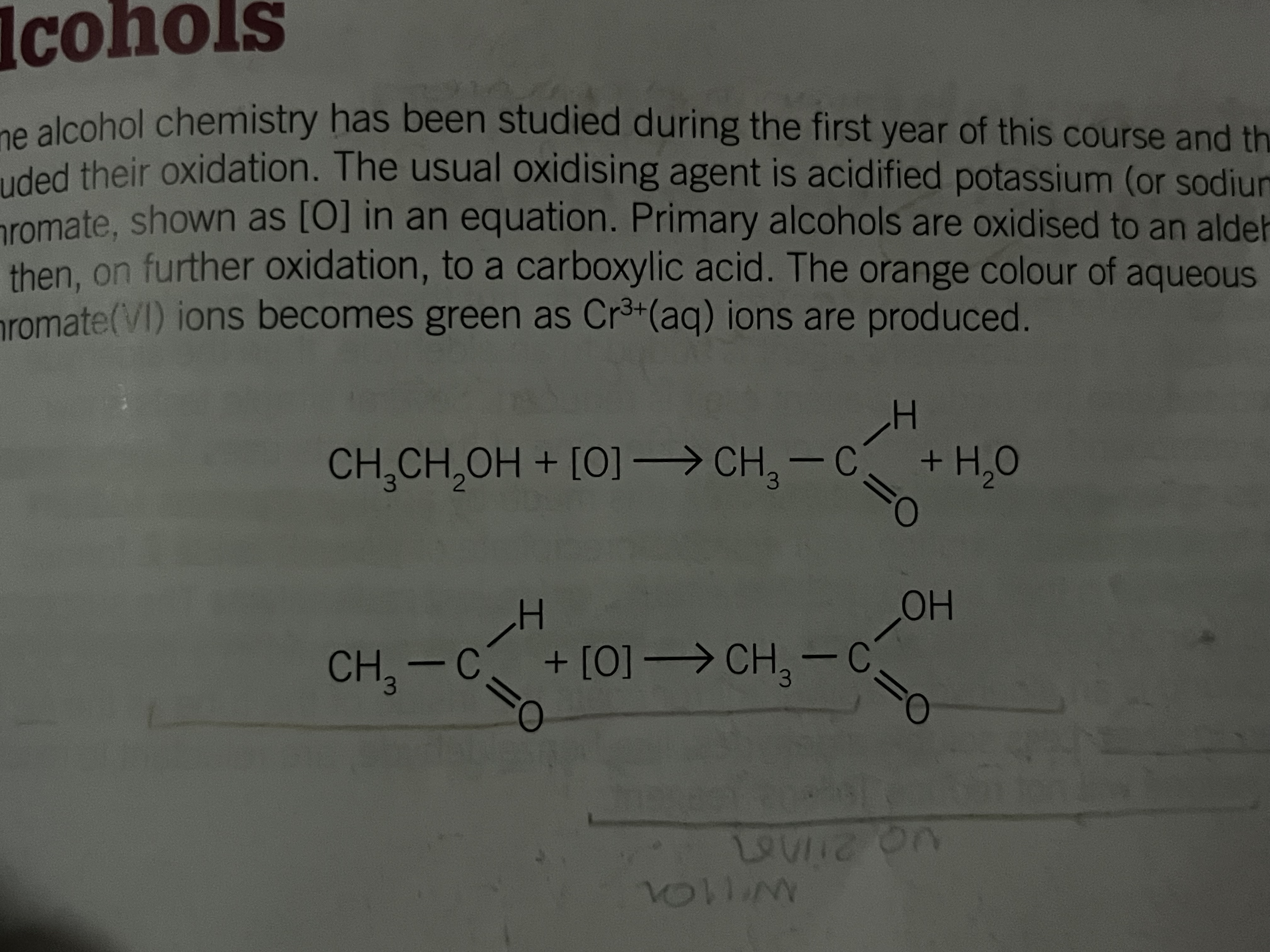
Formation by the oxidation of primary alcohols
Primary alcohols are oxidized to aldehydes, then further to carboxylic acids
the oxidising agent is acidified potassium (or sodium) dichromate K2Cr2O7H+
colour change from orange to green
Formation by the oxidation of secondary alcohols
Under normal conditions a ketone cannot be further oxidized by this method
acidified potassium manganate can also be used as the oxidizing agent
purple/pink colour of the manganate ion is lost and a colourless solution containing aqueous manganese ions remain

Distinguishing between aldehydes and ketones
Tollens agent
Ammoniacal silver nitrate)
made by adding aqueous sodium hydroxide to silver nitrate solution until a brown precipitates of silver (l) oxide is formed
aqueous ammonia is then added until the silver oxide just redissolves
aldehyde is then added to this reagent and the tube is gently warmed in a beaker of water
Result of Tollens reagent
If the compound is an aldehyde, a silver mirror coats the inside of the tube as the Ag+ ions are reduced to Silver
less soluble aldehydes are reluctant to react in this way
ketone doesn't form a silver mirror
Distinguishing between aldehydes and ketones
Fehling's reagent
tartate
Reacts to an aldehyde but not a ketone
also shows the presence of an aldehyde group in reducing sugars (eg. Glucose)
Fehling A is an aqueous solution of copper (ll) sulfate
Fehling B is an aqueous solution of potassium sodium tartrate (made alkaline with sodium hydroxide)
on mixing the two Solutions a deep blue solution containing complex copper (ll) ion is produced
Result of Fehlings solution
If an aldehyde is added to the deep blue solution and mixture is warmed in a water bath, then the aldehyde reduces the complex copper (ll) ions and the deep blue solution is replaced with an orange red precipitate of copper (l) oxide
Decomposition of Fehlings solution
Solution decomposes easily and the two solutions are mixed when needed.
Benedict's reagent is a similar reagent containing a complex copper ll ion and is similarly reduced to Copper (l) oxide
Advantage to Benedict's reagent
Stable and is safer to use as it is less alkaline
the test using the Solutions is effective in identifying ALIPHATIC aldehydes but doesn't react with aromatic aldehydes
The reduction of aldehydes and ketones
Aldehydes and ketones are reduced to primary and secondary alcohols respectively
reducing agents are sodium tetrahydridoborate (NaBH4) or lithiumtetrahydridoaluminate (LiAlH4)
NaBH4 is preferred as it is safer and can be used in acquies conditions
Nucleophilic addition reactions of aldehydes and ketones
Both contain a polar carbonyl bond C=O
the relatively electron of deficient carbon atom can be attacked by a nucleophile (Eg. CN)
Identifying aldehydes and ketones using
2,4-dinitrophenylhydrazine
2,4 DNP is used because a react with an aldehyde or ketone to produce an orange/yellow or red crystalline solid which can be filtered off and purified
it's it's dissolved in an acid to give a solution (Brady's reagent)
Melton temperature of purified product
( 2,4 dinitrophenylhydrazone) is taken and compared with a table of known values to identify the aldehyde or ketone present
called “making a derivative/ derivatisation”
Mechanism of this reaction
Takes place by nucleophilic addition-elimination (sometimes called condensation reaction)
this happens because the nitrogen atom has a lone pair of electrons attached to it, and so act as a nucleophile, attacking the slightly positive carbon atom of the C=O in the aldehyde or ketone
followed by the elimination of a water molecule
derivatives are called hydrazones
Iodoform reaction/ triiodomethane
Triiodomethane CHI3 is a yellow solid that has some uses as an antiseptic
The yellow solid formed helps to identify a methyl carbonyl group (CH3CO)
A positive result from the iodoform
Methanal would be the only aldehyde to give a positive result
methyl ketones would be the other type of compound to give a positive result
Contents and reaction of iodoform
The organic compound is warmed with a color solution of iodine in aqueous sodium hydroxide (alkaline iodine).
or aqueous mixture of potassium iodide and sodium chlorate (l), NaCOl
Test can also be used on methyl alcohols
The OH group in alcohols must be attached to a carbon atom with a hydrogen and a methyl group attached
therefore this will work for ethanol and secondary alcohols
Acidity of carboxylic acids
extent of ionisation
They are weak acids and the extent of the ionization in aqueous Solutions is very small
they are acidic because of the hydrogen in the COOH group
carboxylic acids > phenols > water/alcohols

Test for relative difference in acidity
Can be shown by their reaction with sodium hydrogen carbonate solution
only carboxylic acids are strong enough to produce colorless bubbles of carbon dioxide gas