Valence Bond Theory + Molecular Orbitals
0.0(0)
Card Sorting
1/15
Earn XP
Description and Tags
Last updated 8:10 PM on 10/17/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
1
New cards
Valence Bond Theory
a merger of Quantum Mechanics and Lewis's Model for shared electron pairs
2
New cards
Sigma Bond
bond where electron density is the highest between 2 atoms along "bond axis"
3
New cards
Hybridization
atomic orbitals can be mixed to generate new, hybrid atomic orbitals in different geometries
4
New cards
Pi Bond
bond where the electron density is highest above and below the "bond axis"
5
New cards
Chirality
not superimposable with its mirror image
6
New cards
Isomers
same chemical formulas but different structural formulas
7
New cards
Aromaticity
a property of cyclic molecular structures with pi bonds in resonance that gives increased stability. (Benzene, alternating single + double bonds)
8
New cards
Molecular Orbitals
these form when atomic orbitals combine. If 2 atomic orbitals combine, 2 molecular orbitals result.
9
New cards
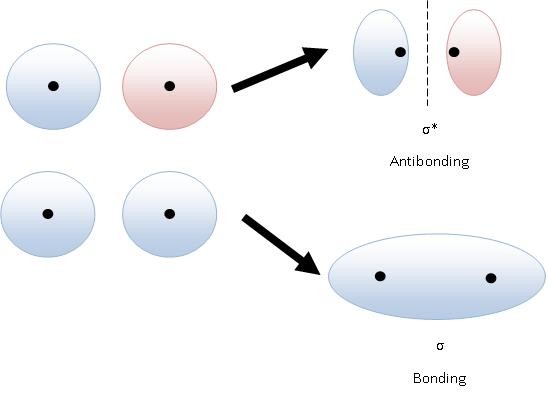
Bonding Orbitals
Lower energy than the atomic orbitals that made it. Increased electron density between nuclei. Help hold atoms together.
10
New cards
Anti-Bonding Orbitals
Higher energy than the atomic orbitals that made it. Decreased electron density between nuclei (nodes), destabilize molecules.
11
New cards
Bond Order Calculation
1/2(bonding electrons - anti bonding electrons)
12
New cards
Diamagnetic
no unpaired electrons, doesn't interact with a magnetic field (nitrogen)
13
New cards
Paramagnetic
has unpaired electrons, does interact with a magnetic field (oxygen)
14
New cards
HOMO
highest occupied energy level
15
New cards
LUMO
lowest unoccupied energy level
16
New cards
HOMO-LUMO GAP
energy required to excite an electron from HOMO to LUMO. Basis of solar energy.