Coordination Compounds PART TWO
1/113
Earn XP
Description and Tags
Bonding Theories, Isomerism. I've skipped colour and organometallic complexes because I really can't lol. Check out my other flashcards too! Answer mode: Answer with Definition
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
114 Terms
What does Werner’s Theory postulate?
Each central metal ion/atom in a coordination sphere shows two types of valences: primary valency and secondary valency.
What does the primary valency of a central metal atom/ion represent?
the oxidation state of that metal atom/ion
What does the secondary valency of a central metal atom/ion represent
the coordination number of that metal atom/ion.
Is the primary valency of a central metal atom/ion directional or non-directional?
non-directional what the fuck even is this btw
Is the secondary valency of a central metal atom/ion directional or non-direction
directional
Which valency of the central metal atom/ion is ionisable? (primary/secondary valency)
primary
Which valency of the central metal atom/ion is NOT ionisable? (primary/secondary valency)
secondary
What is the coordination number of Fe2+?
6
What is the coordination number of Fe3+?
6
What is the coordination number of Co3+?
6
What is the coordination number of Pt4+?
6
What is the coordination number of Cr3+?
6
What is the coordination number of Ru2+?
6
What is the coordination number of Os3+?
6
What is the coordination number of Pt2+?
4
What is the coordination number of Ni2+?
4
What is the coordination number of Cu2+?
4
What is the coordination number of Zn2+?
4
What is the coordination number of Au3+?
4
What is the coordination number of Hg2+?
4
What is the coordination number of Ag1+?
2
What is the coordination number of Mo3+?
8
What sort of molecules can satisfy a central metal atom’s primary valency?
(anions, cations, neutral molecules)
only anions
What sort of molecules can satisfy a central metal atom’s secondary valency?
(anions, cations, neutral molecules)
anions and neutral molecules
If a central metal atom has a coordination number of 2, what is its hybridisation?
sp
linear
If a central metal atom has a coordination number of 4, what are its possible hybridisations?
sp3 (tetrahedral)
dsp2 (square planar)
If a central metal atom has a coordination number of 6, what are its possible hybridisations?
sp3d2 (octahedral)
d2sp3 (octahedral)
If a central metal atom has a coordination number of h, what is its hybridisation?
dsp3 (trigonal bipyramidal)
What are inner orbital complexes / low spin complexes?
complexes where the central metal atom has its inner (n−1)d orbital participating in hybridisation
What are outer orbital complexes / high spin complexes?
complexes where the central metal atom has its outer nd orbital participating in hybridisation
What are Weak Field Ligands?
Ligands that cannot force electrons to pair up in the central metal atoms/ions.
What are the three main groups for donor atoms of Weak Field Ligands?
Sulphur
Halogens
Oxygen
What are the three main groups for donor atoms of Strong Field Ligands?
Nitrogen
Carbon
Phosphorus
What is the formula for calculating the magnetic moment of a complex?
n(n+2)
where n is the number of unpaired electrons
What is the hybridisation of [Fe(CN)6]3+?
d2sp3
What is the hybridisation of [FeF6]3−?
sp3d2
What is the hybridisation of [Ni(CN)4]2−?
dsp2
What is the hybridisation of [NiCl4]2−?
sp3
What is the hybridisation of [Ni(CO)4]?
(special case)
sp3
this is because the s orbital must be empty always for hybridisation.
What is the hybridisation of [Cu(NH3)4]2+?
(special case)
dsp2
this is because dsp2 is more stable than sp3 with one unpaired electron.
What does the Effective Atomic Number of a metal ion denote?
It tells us about the number of electrons in a central metal ion after making coordinate bonds with ligands.
What is EAN’s rule?
Complexes having an Effective Atomic Number equal to the atomic number of the next noble gas are found to be extra stable.
What is the formula for calculating Effective Atomic Number of a central metal ion/atom?
EAN=Z−OS+2CN
EAN=atomic number of metal−oxidation state of metal+the number of electrons provided by ligands through coordinate bonds.
What is the main postulate of Crystal Field Theory?
When a ligand approaches the central metal ion, the degenerate d orbitals of the metal ion undergo splitting into two sets (eg and t2g).
What is crystal field splitting energy?
The energy difference between the two split sets of d orbitals under Crystal Field Theory.
How does the Crystal Field strength of a ligand affect Δ i.e. the Crystal Field Splitting Energy of a metal atom/ion’s d orbitals?
Δ∝crystal field strength of ligand.
What does the dxy orbital look like?
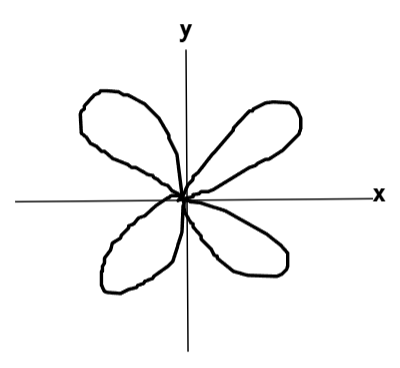
What does the dyz orbital look like?
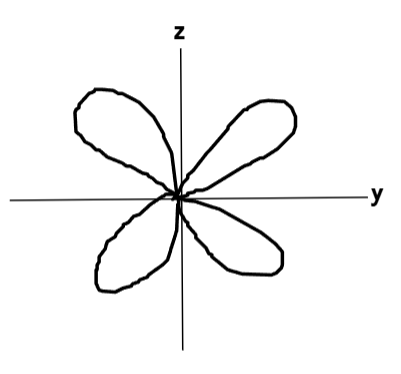
What does the dzx orbital look like?
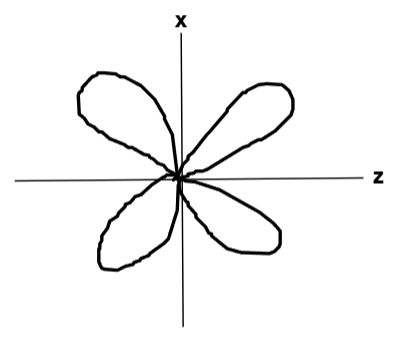
What does the dx2−y2 orbital look like?
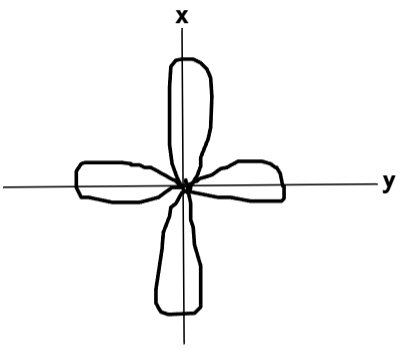
What does the dz2 orbital look like?
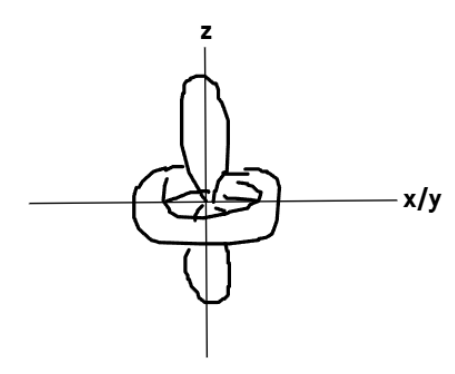
Which d orbitals are axial orbitals?
dx2−y2 and dz2
Which d orbitals are non-axial orbitals?
dxy, dyz and dzx
What are axial orbitals called?
(t2g / eg)
eg
What are non-axial orbitals called?
(t2g / eg)
t2g
How does the diagram of the splitting of d orbitals look in the presence of SFLs and WFLs in an octahedral complex?
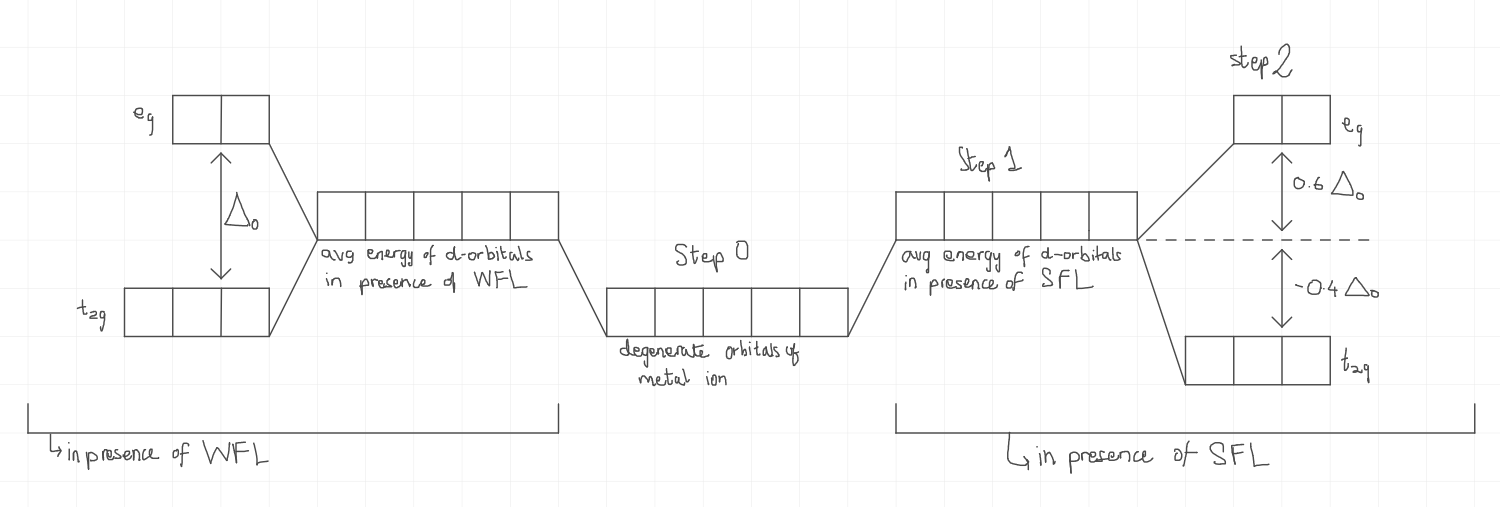
What is pairing energy?
The energy required to force two unpaired electrons into one orbital is called pairing energy.
In the presence of Strong Field Ligands, which one is greater; Crystal Field Splitting Energy or Pairing Energy?
Crystal Field Splitting Energy
In the presence of Weak Field Ligands, which one is greater; Crystal Field Splitting Energy or Pairing Energy?
Pairing Energy
What is Crystal Field Stabilisation Energy?
It is the energy by which complex compounds get stable due to the splitting of d orbitals.
What is the formula for calculating Crystal Field Stabilisation Energy in an octahedral complex?
CFSE=(0.6×Δo×neg)+(−.04×Δo×nt2g)+np
Where:
Δo is the crystal field splitting energy in an octahedral complex
neg is the number of electrons in the eg orbitals
nt2g is the number of electrons in the t2g orbitals
n is the number of pairing done by SFLs (∴ in WFLs this value is 0)
p is the pairing energy
The low spin complex of a d6 cation in an octahedral complex will have what Crystal Field Stabilization Energy? (in terms of Δo and p)
CFSE=(0.6×Δo×0)+(−0.4×Δo×6)+3p
CFSE=−2.4Δo+3p
In an octahedral complex, which orbitals have more energy, eg or t2g?
eg
In a tetrahedral complex, which orbitals have more energy, eg or t2g?
t2g
How does the diagram of the splitting of d orbitals look in the presence of SFLs and WFLs in a tetrahedral complex?
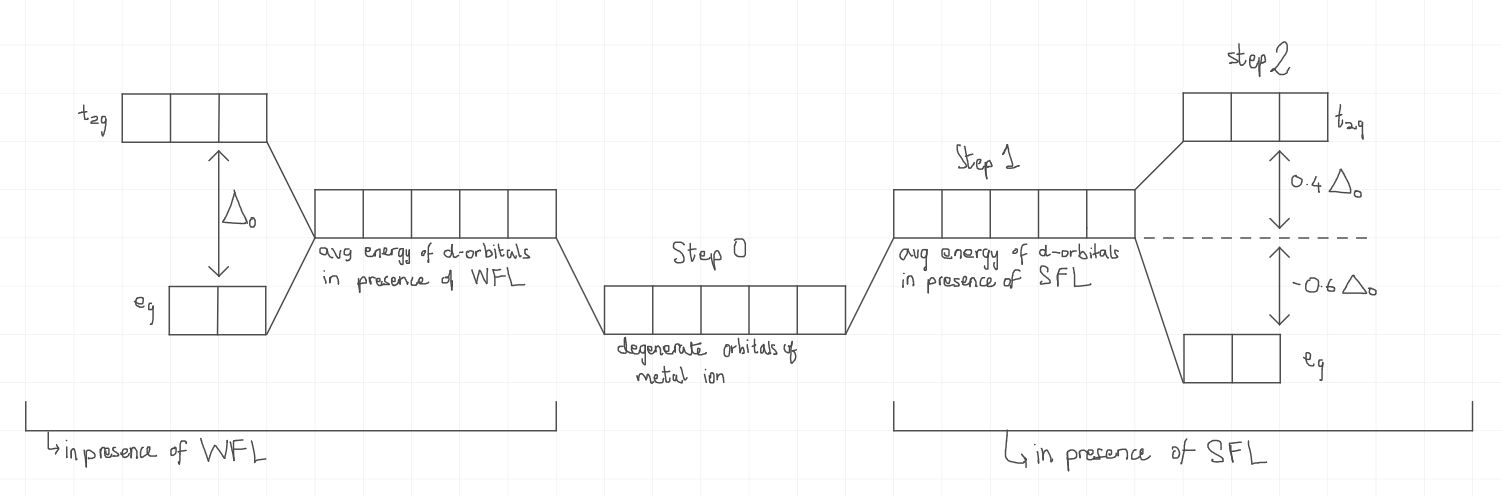
What is the formula for calculating Crystal Field Stabilisation Energy in an octahedral complex?
CFSE=(0.4×Δt×neg)+(−0.6×Δt×nt2g)+np
Where:
Δt is the crystal field splitting energy in an tetrahedral complex
neg is the number of electrons in the eg orbitals
nt2g is the number of electrons in the t2g orbitals
n is the number of pairing done by SFLs (∴ in WFLs this value is 0)
p is the pairing energy
What is the relationship between Δo and Δt?
Δo is the crystal field splitting energy in an octahedral complex
Δt is the crystal field splitting energy in an tetrahedral complex
Δo=49Δt
Δo is greater
List the spectrochemical series as best as you can.
(good luck 💀)
Weak Field Ligands:
I− < Br− < S2− < SCN− < Cl− < NO3− < N3− < F− < OH− < C2O42− < H2O
Strong Field Ligands:
NCS− < CH3CN < py < NH3 < en < bipy < phen < NO2− < PPh3 < CN− < CO
How does the oxidation state of the central metal ion affect the stability of the complex?
the greater the oxidation state, the more stable the complex
For a group complexes with the same oxidation number (of metal ion), same ligands, and same coordination number, how would you determine which one is more stable?
Greater the Zeff, more stable the complex
How does chelation affect the stability of a complex?
Chelating ligands are more stable than non-chelating ligands.
Which ligands make the complex more stable; SFLs or WFLs?
SFLs
How does the number of rings in a complex affect the stability of the complex?
the more the number of rings, the more stable the complex.
What is Ionisation isomerism?
Coordination compounds that have the same composition but different ions in solution.
What sort of isomerism do these two show?
[Co(NH3)5Cl]Br
[Co(NH3)5Br]Cl
Ionisation isomerism
What is hydrate/solvate isomerism?
The coordination compounds that have the same composition but a different number of H2O molecules present as ligands?
What sort of isomerism do these two show?
[Cr(H2O)5Cl]Cl2⋅H2O
[Cr(H2O)4Cl2]Cl⋅2H2O
hydrate/solvate isomers
What sort of isomers give different confirmative tests?
ionisation isomers
What is coordination isomerism?
Isomerism caused by the interchange of ligands and metal ions between two complex ions of the same complex
(yeah both the anion and cation in the compound are complex)
What sort of isomerism do these two show?
[Pt(NH3)3Cl][CuCl3(NH3)]
[Pt(NH3)4][CuCl4]
coordination isomerism
What is linkage isomerism?
The coordination compounds that have the same composition but differ in mode of attachment of ligand to metal ion.
(i.e. ambidentate ligands must be present)
What sort of isomerism do these two show?
[Co(NH3)5NO2]Cl2
[Co(NH3)5B(ONO)]Cl2
linkage isomerism
What are stereoisomers?
Isomers that have the same formula, same composition, but differ in the orientation of ligands around the metal atom.
What are geometrical isomers?
Isomers of compounds where ligands occupy different positions around the central metal ion.
What are cis-isomers?
Isomers where two identical ligands are coordinated to the central metal ion from the same side.
What are trans-isomers?
Isomers where two identical ligands are coordinated to the central metal ion from opposite sides.
What is the characteristic of optically active isomers?
Isomers are due to lack of center/plane of symmetry
What is the characteristic of optically inactive isomers?
Isomers are due to existence of center/plane of symmetry
What sort of geometry must a 4-coordinated complex have for it to definitely not show geometrical isomerism?
tetrahedral geometry
Under what condition can 4-coordinated tetrahedral complexes show optical isomerism?
If all 4 ligands are different, it can show optical isomerism.
Depict the cis and trans isomers of a complex with the general formula [Ma2b2].
(a and b are both monodentate)

Depict the cis and trans isomers of a complex with the general formula [Ma2bc].
(a and b and c are both monodentate)

Can a complex with the general formula [Ma4] show geometrical isomerism?
no
Can a complex with the general formula [Ma2b2] show geometrical isomerism?
yes
Can a complex with the general formula [Ma2bc] show geometrical isomerism?
yes
Can a complex with the general formula [Ma3b] show geometrical isomerism?
no
Depict the geometrical isomers of a complex with the general formula [Mabcd].
(a and b and c and d are both monodentate)
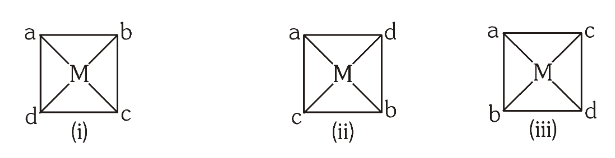
Can a complex with the general formula [M(AA)bc] show geometrical isomerism?
(AA is bidentate with the same donor atom, b and c are monodentate)
no
Can a complex with the general formula [M(AB)cd] show geometrical isomerism?
(AB is bidentate with different donor atoms, c and d are monodentate)
yes
Can a complex with the general formula [M(AA)2] show geometrical isomerism?
(AA is bidentate with the same donor atom)
no