the reactivity series/ reactivity of metals
what is the reactivity series
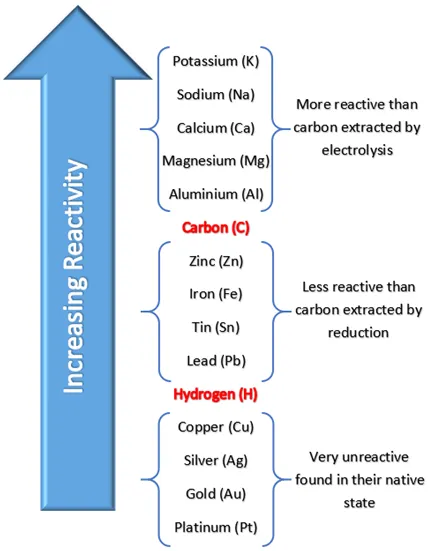
a series of elements going from the highest reactivity to the lowest
reactivity series mnemonic
Please Stop Calling Me A Careless Zebra Instead Try Learning How Copper Saves Gold
1/17
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
18 Terms
what is the reactivity series
a series of elements going from the highest reactivity to the lowest

reactivity series mnemonic
Please Stop Calling Me A Careless Zebra Instead Try Learning How Copper Saves Gold
unreactive metals examples
gold,copper, silver
MASH
metal + acid = salt + hydrogen
copper + sulphuric acid = copper sulphate + hydrogen
alkaline solution
a solution that has a ph more the 7
atom economy
a percentage of the amount of starting materials become useful products
(RFM of desired product from equation/sum of RFM of all reactants in the equation) x 100
displacement reaction
a reaction in which a substance of a higher reactivity replaces one with a lower reactivity in a compound
limiting reactant
a substance that is totally consumed when a reaction is complete, limiting the amount of product formed
oxidation
gain of oxygen
loss of electrons
oxidising agent
a substance that is capable of oxidising another substance
percentage yield
a percentage of the amount of product actually obtained compared to the theoretical amount that is possible to obtain
(mass of product actually made/maximum theoretical mass of product) x 100
why would the mass of the product formed not be the same as the theoretical mass?
- the reaction may not go to completion
- some of the product may be lost
- there may be a spillage somewhere
redox reaction
a reaction in where reduction and oxidation take place
reducing agent
a substance that is capable of reducing another substance
reduction
the gain of electrons or the loss of oxygen
lithium in water
fizzes
produces hydrogen
sodium in water
moves around and fizzes rapidly
produces hydrogen
makes an orange flame
potassium in water
moves around and fizzes vigorously
produces hydrogen
burns with a lilac flame