week 2 - nomenclature, atomic orbitals
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
52 Terms
1
meth
2
eth
3
prop
4
but
5
pent
6
hex
7
hept
8
oct
9
non
IUPAC strongness order
carboxylic acid
esters
amides
aldehydes
ketones
alcohols
thiols
amines
alkenes
alkanes
carboxylic acid suffix
oic acid
esters suffix
oate
amides suffix
amide
aldehydes suffix
al
ketones suffix
one
ketones prefix
oxo
alcohols suffix
ol
alcohols prefix
hydroxy
thiols suffix
thiol
thiols prefix
mercapto
amines prefix
amine
amines prefix
amino
alkenes suffix
ene
alkenes prefix
enyl
alkanes suffix
ane
alkanes prefix
yl
what kind of wave are electrons found in
standing waves
more nodes…
higher in energy
when does a phase change happen
every time it goes through a node
quanta mechanics
allows e-’s to share the same space as long as they have opposite spin
bond is always accompanied by a…
anti-bond
how does a bond break
put e-’s into the anti-bond orbital causing the corresponding bond to break
C-H subtraction/addition
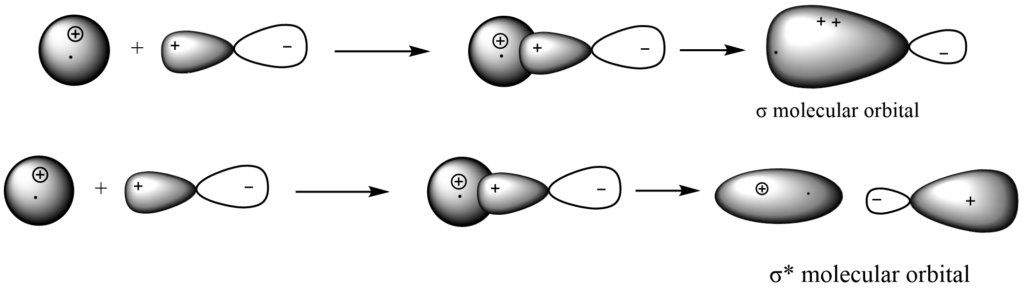
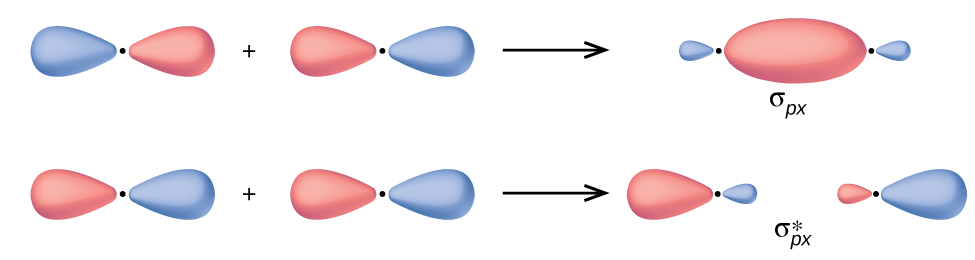
C-C addition/subtraction
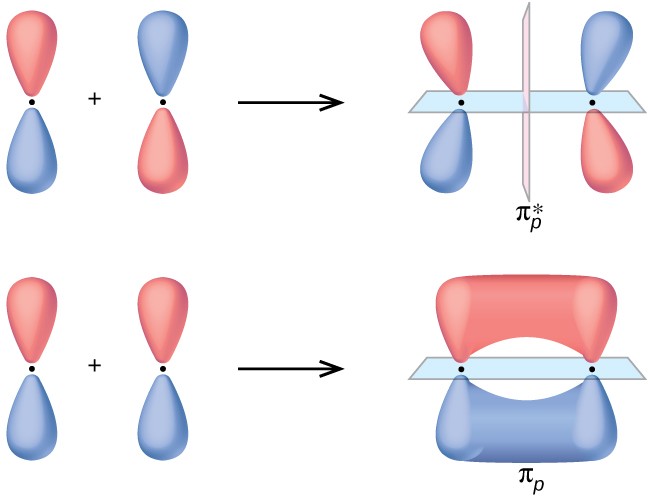
C-C pie subtraction/addition
homo
highest occupied molecular orbital
lumo
lowest unoccupied molecular orbital
higher energy…
less stable
lower in energy if…
on same period as the molecule
O-H
usually higher because oxygen is more electronegative than H
C-C
lowest electron level because most stable
in a carbonyl what is the formal charge of carbon
positive
(oxygen - negative)
what does homo attack
the partial plus charge of the other molecule in the limo bond that’s being broken
rule in finding homo
nb
pie
^what to look for first
rule to finding lumo
pie star (especially c-o)
sigma star (especially o-h)
all atoms involved in resonance…
will be sp2 hybridized
atoms shape if sp2
2D
atoms shape if sp3
3D
4 bonds; sp3
tetrahedral
3 bonds; sp2
trigonal planar
bolded line
towards us
dashed line
away from us