Biology Unit 1 (Measurements)
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
28 Terms
What is the metric base unit?
meter (m) - Length
gram (g) - mass
liter (l) - volume
Kelvin (K) and Celsius (C) - temperature
tera (T)
10^12
giga (G)
10^9
mega (M)
10^6
kilo (k)
10³
hecto (h)
10²
deka (da)
10^1
deci (d)
10^-1
centi ( c )
10^-2
milli (m)
10^-3
micro (µ)
10^-6
nano (n)
10^-9
pico (p)
10^-12
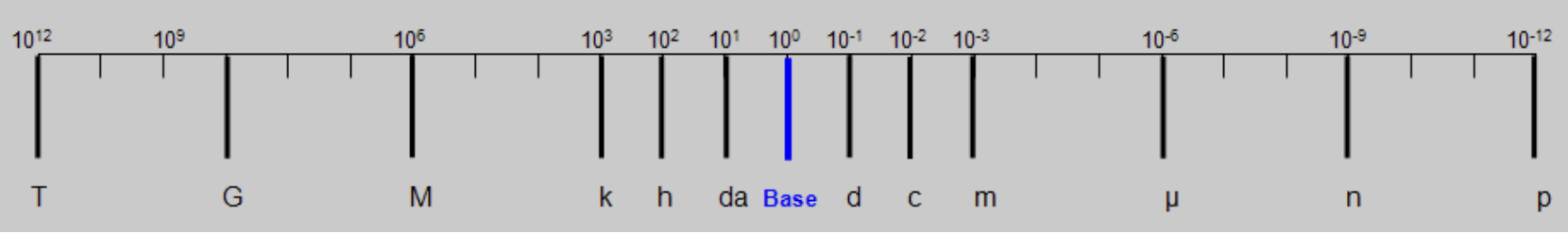
How to convert measurement.
The base represents gram, meter, and liter. Depending on what you want it to be converted to, you either have to move your decimals to the right or left.
decimeter (dm) and meter (m)
1 decimeter (1 dm) = 10 centimeters (10 cm) = 100 millimeters (100 mm)
How much does 1 ml = g
1 ml = 1 g = 1 cc

What is this?
Pipette Filling Device


What is this?
Graduated Cylinder

What is this?
Beaker

What is this?
Erlenmeyer Flask

What is this?
Bathroom Scale

What is this?
Triple Beam Balance

What is this?
Electronic Scale

What is this?
Thermometer
How to convert C to F?
1.8°C + 32
How to convert F to C?
(°F – 32)/1.8
How to do Scientific Notation?
Every time you increase the exponent by 1, you are multiplying by 10. If you decrease the power by 1, you are dividing by 10. For example 123,000 is 1.23 x 105.
Since 105 = 100,000 then ….1.23 x 10,0000 = 123,000. Another example is 0.000123 is 1.23 x 10-4 . Since 10-4 = 0.0001 then… 1.23 x .0001 = 0.000123
How to do Significant Figures?
Zeros within a number are always significant. Both 1203 and 10.02 contain four significant figures.
Zeros that do nothing but set the decimal point are not significant. The number 120,000 has two significant figures. The number 0.000123 has three significant figures.
Trailing zeros that are not needed to hold the decimal point are significant. For example, 1.00 has three significant figures.