enthalpy changes and calorimetry
1/14
Earn XP
Description and Tags
not including equations
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
define energetics:
energy changes during chemical reactions
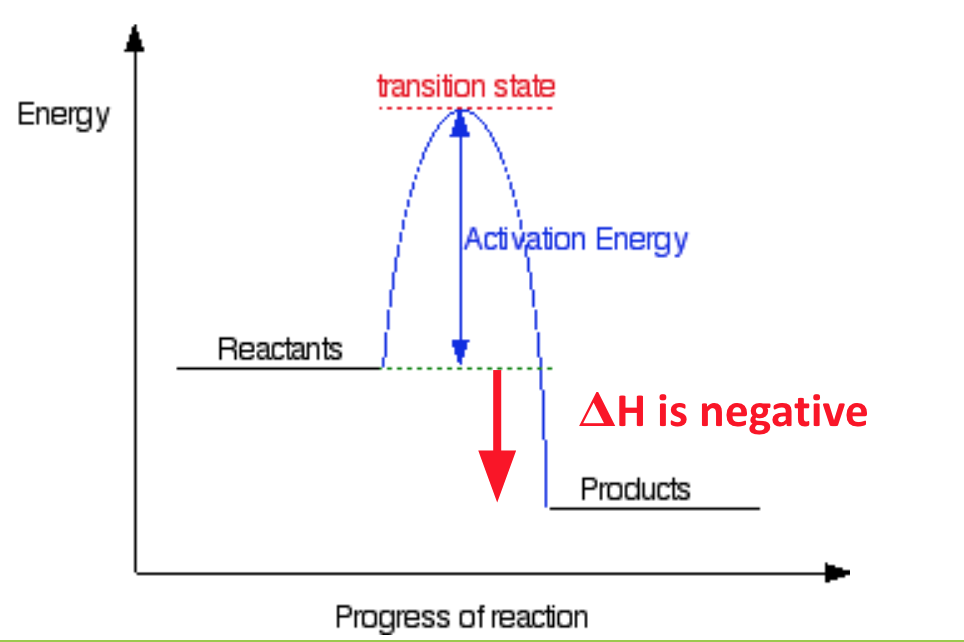
what is an exothermic reaction? describe the diagram for an exothermic reaction
a reaction in which heat energy is given off
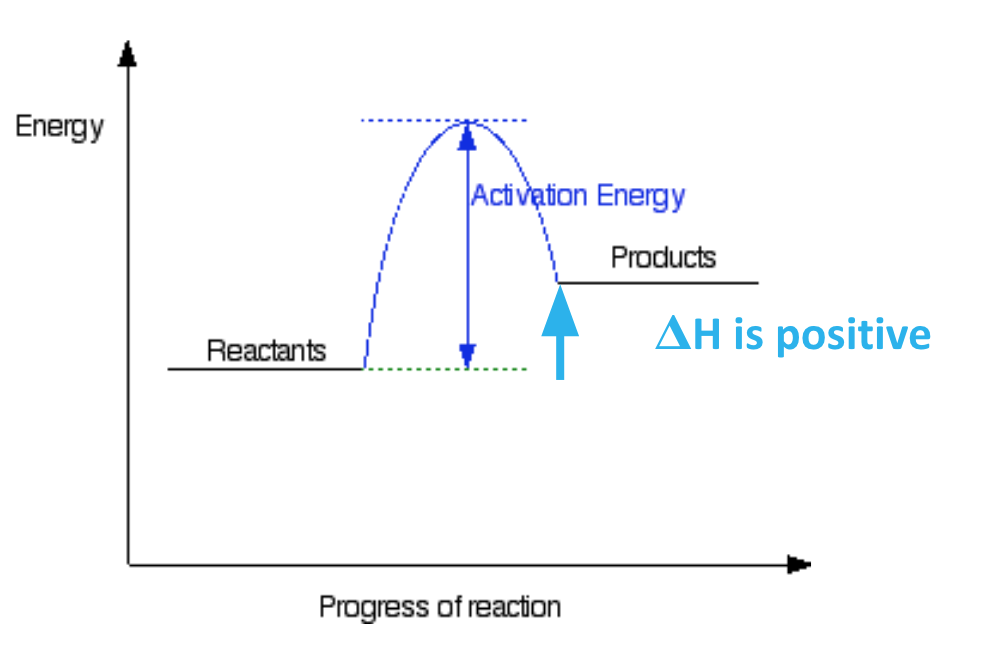
what is an endothermic reaction? describe the diagram for an endothermic reaction and state whether ΔH is +ve or -ve:
a reaction in which heat energy is taken in
what is enthalpy? what symbol is it represented by?
a measure of the heat content of a substance
represented by the symbol H
what are the units for enthalpy?
kJ mol-1
what is enthalpy change? what symbol(s) is it represented by?
heat energy change at constant pressure
represented by ΔH
what are standard conditions for a reaction?
100 kPa
stated temp (usually 298K)
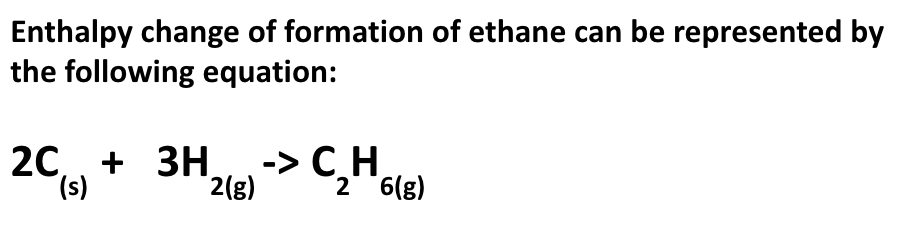
what is the standard enthalpy change of formation of a compound? how is it represented?
enthalpy change which occurs when:
1 mole of the compound is formed from its elements
under standard conditions and w/ everything in its standard state
represented by ∆f H Ɵ
what is the standard enthalpy change of combustion of a compound? how is it represented?
the enthalpy change which occurs when ONE MOLE of the compound is BURNED COMPLETELY in oxygen under STANDARD CONDITIONS and w/ everything in its STANDARD STATE
represented by ΔHƟc
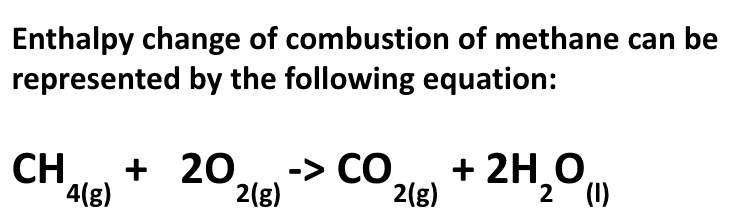
what is the specific heat capacity equation?
q = mcΔT
what is the equation for ΔH?
ΔH = q/moles
what is calorimetry?
the process of measuring the amount of heat energy released/absorbed during a chemical reaction
suggest why the enthalpy change for the thermal decomposition of solid silver nitrate (III) is difficult to determine experimentally (1)
the silver nitrate must be heated so we cannot measure the temp change caused by the reaction


the table in the image shows values, obtained by different methods, for the enthalpy of combustion of a liquid hydrocarbon - suggest reasons for the differences between the values obtained by each of Methods 2 and 3, and the value obtained by Method 1 in the table (5)
any 5 from:
value from calorimetry less exo than method 1
because of heat loss
because of incomplete combustion
because some liquid hydrocarbon could have evaporated
value from bond enthalpies less exo than method 1
mean bond enthalpies values use enthalpies taken across a range of compounds
value from bond enthalpy data ignores E changes in vaporisation of the fuel or condensing the water

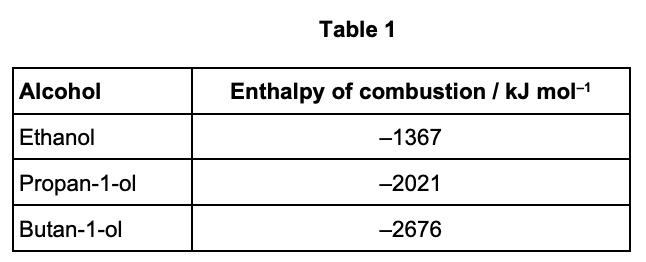
Table 1 shows the enthalpies of combustion of some alcohols.
the enthalpy of combustion calculated from calorimetry is -2060 kJ mol-1 - explain how this suggests the alcohol is butan-1-ol (2)
value measured not accurate due to heat loss/incomplete combustion
result from experiment must be less exo than true value ∴ cannot be propan-1-ol/others
