12.1 / 12.2 - Physical States and Phase Changes
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
What are intramolecular / bonding forces?
forces found within a molecule, responsible for chemical behavior
What are intermolecular / nonbonding forces?
forces found between molecules, responsible for the physical behavior
what are the two equations for phase change questions
q = mc(deltaT)
q = deltaH * n
When to use
q = mc(deltaT)
when no phase change is occuring, only temp is changing.
when to use
q = deltaH * n
when a phase change in occuring, temp is not changing
what is vapor pressure
in a closed container, when the gas in the container puts pressure on the liquid which make its difficult for the liquid to vaporize
when temp increases, vapor pressure ___.
increases
the weakerthe intermolecular forcers, the more difficult/easier it is for particles to enter the vapor phase and the higher/lower the p.
easier, higher
what is the clausius clapeyron equation you use when only 1 temperature is provided in the question
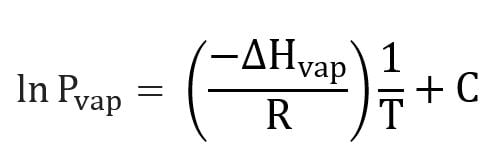
what is the clausius clapeyron equation you use when only 2 temperatures OR 2 pressures are provided in the question

the temperatures for the clausius clapeyron equations must be converted to ___.
Kelvin
what is a critical point
where liquid and gas do not exist as distinct phases