Practicals related to 3.5 Energy transfers in and between organisms
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Describe how pigments from a leaf of a plant can be isolated with paper chromatography
1. Crush leaves with solvent to extract pigments
2. Draw a pencil line on filter / chromatography paper, 1 cm above bottom
3. Add a drop of extract to line (point of origin)
4. Stand paper in boiling tube of (organic) solvent below point of origin
5. Add lid and leave to run (solvent moves up, carrying dissolved pigments)
6. Remove before solvent reaches top and mark solvent front with pencil
Explain why the origin should be drawn in pencil rather than ink.
● Ink is soluble in solvent
● So ink would mix with pigments / line would move
Explain why the point of origin should be above the level of the solvent.
● Pigments are soluble in solvent
● So would run off paper / spots dissolve into solvent
Explain why a pigment may not move up the chromatography paper in one solvent.
● May be soluble in one solvent but insoluble in another
Describe how pigments can be identified
● Rf value = distance moved by spot / distance moved by solvent front
● Compare Rf value to published value
Explain why the solvent front should be markedquickly once chromatography paper is removed.
● Once solvent evaporates, solvent front not visible
Explain why the centre of each pigment spot should be measured.
● Standardises readings as pigment is spread out
● So allows comparisons to be made
Explain why the obtained Rf values were similar, but not identical, to the published values.
● Different solvent / paper / running conditions may
affect Rf value
Explain why Rf values are used and not the distances moved by pigment spots.
● Solvent / pigment moves different distances
● Rf value is constant for same pigment / can be compared
Describe the role of the enzyme dehydrogenase in photosynthesis
● Catalyses the reduction of NADP in the light-dependent reaction
○ NADP accepts (gains) electrons from photoionisation of chlorophyll / photolysis of water
Describe how rate of dehydrogenase activity in extracts of chloroplasts can
be measured
1. Extract chloroplasts from a leaf sample using the method in ‘2.1.3 Methods of studying cells’
2. Set up test tubes as follows:
A. Control 1 - set volume of DCPIP (redox indicator dye, electron acceptor), water and
chloroplasts in isolation medium, covered in foil to block light
B. Control 2 - set volume of DCPIP, water and isolation medium without chloroplasts
C. Standard - set volume of water and chloroplasts in isolation medium, without DCPIP
D. Experiment - set volume of DCPIP, water and chloroplasts in isolation medium
3. Shine light on test tubes and time how long to it takes for DCPIP to turn from blue (oxidised) to
colourless (reduced) in tube D (tube A and B should show no change)
○ Compare to a colour standard (tube C) to identify end point
4. Rate of dehydrogenase activity (s
-1
) = 1 / time taken
Give examples of variables that could be controlled.
● Source of chloroplasts
● Volume of chloroplast suspension
● Volume / concentration of DCPIP
Explain the purpose of control 1 (tube A).
● Shows light is required for DCPIP to decolourise
● Shows that chloroplasts alone do not cause DCPIP to decolourise
Explain why DCPIP in control 1 stays blue.
● No light so no photoionisation of chlorophyll
● So no electrons released to reduce DCPIP
Explain the purpose of control 2 (tube B).
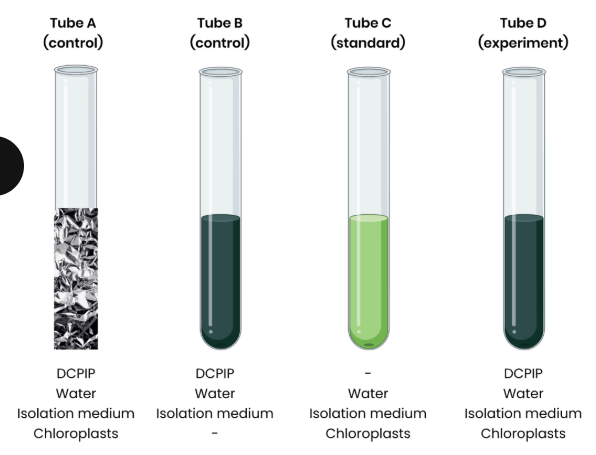
● Shows chloroplasts are required for DCPIP to decolourise
● Shows that light alone does not cause DCPIP to decolourise
Explain why DCPIP changes from blue to colourless.
● DCPIP is a redox indicator / DCPIP gets reduced by electrons
● From photoionisation of chlorophyll
Suggest a limitation with themethod and how the experiment could be modified to overcome this.
● End point (colour change) is subjective
● Use a colorimeter
● Measure light absorbance of sample at set time intervals
● Zero colorimeter using the colour standard
Describe how a respirometer can be used to measure the rate of aerobic respiration (by measuring oxygen uptake)
1. Add set mass of single-celled organism eg. yeast to set volume / conc. of substrate eg. glucose
2. Add a buffer to keep pH constant
3. Add a set volume / conc. of a chemical that absorbs CO2 eg. sodium hydroxide
4. Place in water bath at a set temperature and allow to equilibrate
5. Measure distance moved by coloured liquid in a set time
Explain why the liquid moves.
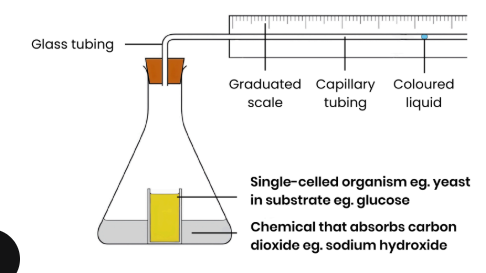
● Organisms aerobically respire so take in O2
● CO2 given out but absorbed by sodium hydroxide solution
● So volume of gas and pressure in container decrease
● Fluid in tube moves down pressure gradient towards organism
Explain why the respirometer apparatus is left open for 10 minutes. (1)
● Allow apparatus to equilibrate
● Allow for overall pressure expansion/change throughout
● Allow respiration rate of organisms to stabilise
Explain why the apparatus must be airtight.
● Prevent air entering or leaving
● Would change volume and pressure, affecting movement of liquid
Describe a more accurate way to measure volume of gas.
● Use a gas syringe
Suggest a suitable control experiment and explain why it is necessary.
● No organisms OR use inert objects OR use dead organisms
AND all other conditions / apparatus / equipment the same.
● To show that (respiring) organisms are causing liquid to move /
taking up oxygen / causing the change in volume / pressure.
Describe how a respirometer can be used to measure the rate of anaerobic respiration (by measuring carbon dioxide release)
● Repeat experiment as above but remove chemical that absorbs CO2
● Make conditions anaerobic, for example:
○ Layer of oil / liquid paraffin above yeast to stop O2 diffusing in
○ Add a chemical that absorbs O2
○ Leave for an hour to allow O2 to be respired and used up
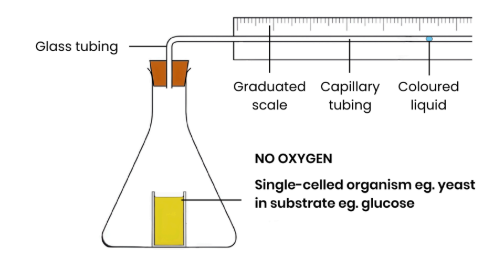
Explain why the liquid moves.

● Yeast anaerobically respire so release CO2
● So volume of gas and pressure in container increase
● So fluid in capillary tube moves down a pressure
gradient away from organism
Explain why the apparatus is left for an hour after the culture has reached a constant temperature.
● Allow time for oxygen to be used / respired
Describe how rate of respiration can be calculated
1. Calculate volume of O2 / CO2 consumed / released (calculate area of a cylinder)
a. Calculate cross-sectional area of capillary tube using π r2
b. Multiply by distance liquid has moved
2. Divide by mass of organism and time taken
3. Units - unit for volume per unit time per unit mass eg. cm3min
-1g
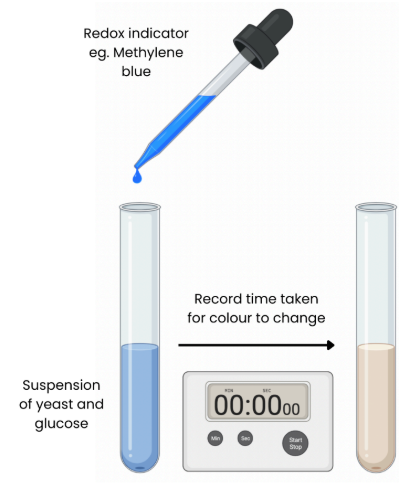
Describe how redox indicator dyes such as Methylene blue can be used to
measure rate of respiration
● Redox indicators (eg. methylene blue) change colour when they accept electrons becoming reduced
● Redox indicators take up hydrogens and get reduced instead of NAD / FAD
→ modelling their reactions
Describe the steps to add the redox indicator to measure rate of respiration
1. Add a set volume of organism eg. yeast and a set volume of respiratory substrate eg. glucose to tubes
2. Add a buffer to keep pH constant
3. Place in water bath at a set temperature and allow to equilibrate for 5 mins
4. Add a set volume of methylene blue, shake for a set time (do not shake again)
5. Record time taken for colour to disappear in tube
6. Rate of respiration (s-1) = 1 / time (sec)
Give two examples of variables that could be controlled.
● Volume of single-celled organism
● Volume / conc. / type of respiratory substrate
● Temperature (with a water bath)
● pH (with a buffer)
● Volume of redox indicator (only control)
Why leave tubes in the water bath for 5 minutes?
● Allow for solutions to equilibrate and reach the same temperature
as the water bath
Suggest a suitable control experiment and explain why it is necessary.
● Add methylene blue to boiled / inactive / dead yeast (boiling denatures enzymes)
● All other conditions the same
● To show change is due to respiration in organisms
Suggest and explain why you must not shake tubes containing methylene blue.
● Shaking would mix solution with oxygen
● Which would oxidise methylene blue / cause it to lose its electrons
● So methylene blue would turn back to its original blue colour
Suggest one source of error in using methylene blue. Explain how this can be reduced.
● Subjective as to determination of colour change / end point
● Compare results to a colour standard (one that has already
changed) OR use a colorimeter for quantitative results
Describe how biomass is formed in plants
● During photosynthesis, plants make organic (carbon) compounds from atmospheric or aquatic CO2
● Most sugars synthesised are used by the plant as respiratory substrates
● Rest used to make other groups of biological molecules (eg. carbs, lipids & proteins) → form biomass
How can biomass be measured?
Mass of carbon or dry mass of tissue per given area
Describe how dry mass of tissue can be measured
1. Sample dried in an oven eg. at 100
oC (avoid combustion)
2. Sample weighed and reheated at regular intervals
3. Until mass remains constant (all water evaporated)
Explain why dry mass is more representative than fresh (wet) mass
Water volume in wet samples will vary but will not affect dry mass
Describe how the chemical energy stored in dry biomass can be estimated
1. Known mass of dry biomass is fully combusted (burnt)
2. Heat energy released heats a known volume of water
3. Increase in temperature of water is used to calculate chemical energy of biomass
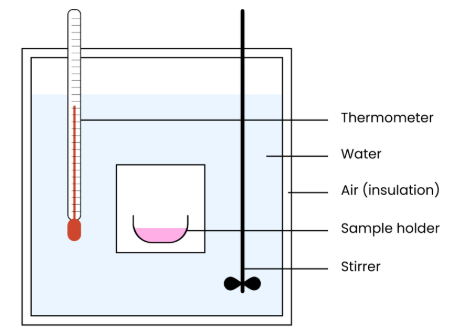
Explain how features of a calorimeter enable valid measurement of heat energy released
● Stirrer → evenly distributes heat energy (in water)
● Air / insulation → reduces heat loss & gain to & from surroundings
● Water → has a high specific heat capacity