Chapter 1: Kinetic Particle Theory
1/16
Earn XP
Description and Tags
Kinetic Particle Theory
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
17 Terms
Kinetic Particle Theory
All matter is made up of particles which are in constant motion. This accounts for the properties of the three states of matter and the changes of states.
Properties Of Solid
Tightly packed. Arranged in an orderly manner.
Can only vibrate upon fixed positions.
Fixed shape.
Fixed volume.
Not easily compressed.
Properties Of liquid
Packed closely together but not as solids. No regular arrangement.
Particles slide against one another.
No fixed shape. Takes on the shape of the container.
Fixed volume.
Not easily compressed.
Properties Of Gas
Spaced far apart from eachother.
Particles move freely at high speeds.
No fixed shap. Takes the shape of the container it is in.
No fixed volume.
Easily compressed.
Changes Of States (Sublimation)
When a solid changes directly into a gas. This happens to only a few solids such as iodine or solid carbon dioxide. The reverse reaction also happens and is also called sublimation (sometimes called deposition or desublimation). Sublimation occurs at a specific temperature which is unique for a pure substance
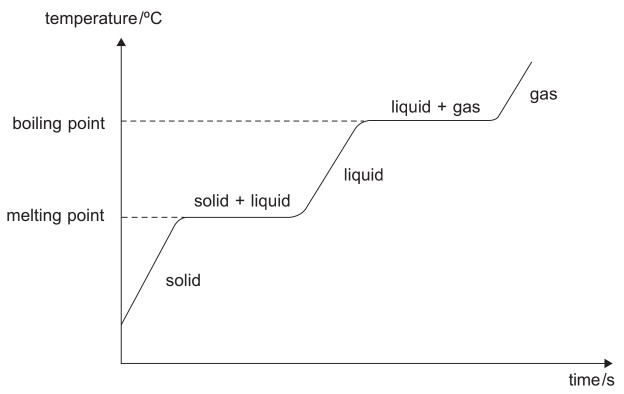
Heating Curve
At parts where the graph rises, heat is supplied to the substance to raise its temperature. The graph becomes flat when the substance undergoes a change in state. The graph remains flat as heat is taken in to overcome the interactions between the particles.
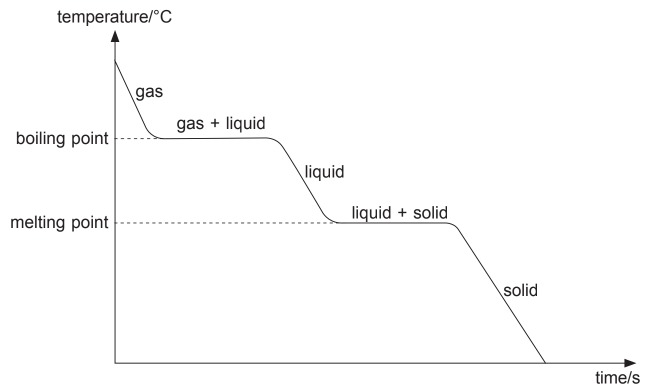
Cooling Curve
At parts where the graph falls, heat is given out from the substance to the surroundings and its temperature decreases. The graph becomes flat when the substance undergoes a change in state. The graph remains flat as the particles form bonds, producing heat which is given out to the surroundings.
Melting
Occurs at the melting point. Particles absorb heat and vibrate more vigorously, allowing them to overcome the interparticle interactions holding them in fixed positions.
Freezing
Occurs at the melting point. Particles release heat and move more slowly. Interparticle interactions are formed and the particles are forced to be held in a fixed and orderly arrangement.
Boiling
Occurs at the boiling point. Particles absorb heat and gain more kinetic energy. The particles move fast enough to completely overcome the forces of attraction.
Evaporation
Occurs below the boiling point. Particles at the surface gain sufficient energy to escape into the surroundings.
Condensation
Occurs at the boiling point. Particles release heat and move more slowly. The forces of attraction are then able to hold the particles closely.
Diffusion
Particles of matter move from a region of higher concentration to a region of lower concentration. Particles with higher mass move more slowly than particles with lower mass. At higher temperature, the rate of diffusion is greater as the particles have more kinetic energy and can move faster.
Gaseous Particles
Gaseous particles are in constant and random motion. An increase in temperature increases the kinetic energy of each particle, as the thermal energy is transformed to kinetic energy, so they move faster. Decreasing the temperature has the opposite effect. The pressure that a gas creates inside a closed container is produced by the gaseous particles hitting the inside walls of the container. As the temperature increases, the particles in the gas move faster, impacting the container’s walls more frequently. Therefore an increase in temperature causes an increase in pressure.
Pure & Impure Solids
Pure substances have a fixed melting point, and boiling point; any change in melting or boiling points indicates impurities. Impurities in liquids increase the boiling point. Impurities in solids decrease the the melting point.
Endothermic
Heat energy ENters from the surroundings to break bonds.
Exothermic
Heat energy EXits into the surroundings to form bonds.