Week 8 Proteins & Amino Acids
1/52
Earn XP
Description and Tags
watch video if you want, i did not
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
What do we use protein to do?
How does our body decide what each protein does?
We use protein to
Diagnose and monitor disease states (Renal, Liver, and Heart problems, etc.)
Evaluate inflammatory response aka see if our body is fighting an infection or injury
Assess nutritional status
How proteins are built (chemically) decides what they do.
What are proteins made up of?
What tells our body how to combine amino acids to make certain proteins?
Can our body make certain proteins?
What proteins can’t our body make?
What is the mnemonic device to memorize the protein our body can’t make?
1) Proteins are made of smaller parts called amino acids,
2) Nucleotide (DNA) sequence tells your body how to put amino acids together to make proteins.
3) Most of our proteins are made by our bodies.
There are some amino acids called essential amino acids our body can't make, so we have to get them from food.
4) "MILL PATH TV" to memorize the essential amino acids
Methionine, Isoleucine, Leucine, Lysine, Phenylalanine, Threonine, Tryptophan, Valine.

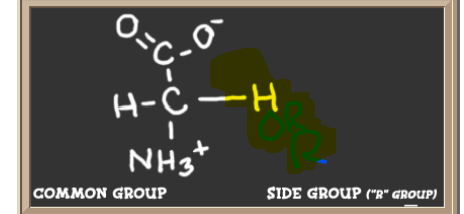
What is the common group of all amino acids?
What does the R group determine?
what can the R group be categorized as?
picture
The "R" group of an amino acid determines how it interacts with other molecules.
R group can be categorized as acidic, basic, water-loving (hydrophilic), water-fearing (hydrophobic), or aromatic.
Central dogma is what?
describe each step
watch video on page 5 powerpoint
The Central Dogma
DNA -> transcription -> RNA -> translation -> Protein
DNA: The instruction manual for building proteins.
Transcription: Copying a part of the DNA into RNA.
RNA: A message that carries the instructions from DNA.
Translation: Using the RNA message to build a protein.
Protein: The final product, which does a job in the cell.
Can amino acids only make protein?
what other do amino acids have?
Amino acids can combine in many ways to make lots of different compounds
Some of the things amino acids make aren't proteins, some amino acids can even be broken down to be used to make glucose (sugar), ketone bodies (energy sources when sugar is low), urea or ammonia
How are amino acids linked together?
where are amino acid linked together?
Define hydration synthesis
Amino acids are linked together covalently by peptide bonds at the C-terminal and the N-terminal
when they join, water is released.
Dehydration synthesis
process of joining molecules by removing water.
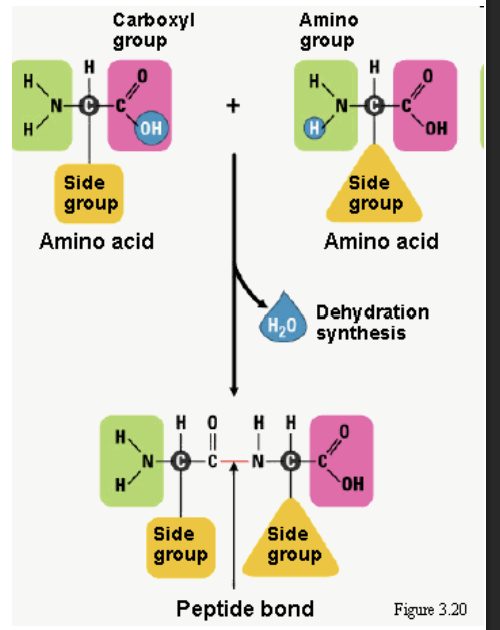
Carboxyl group is what
Amino group is what
Carboxyl is R-COOH
Amino group is R-NH2
Define Aminoacidopathies
Amino-acidopathies are rare inherited disorders:
A problem/defect with an enzyme makes it hard for the body to break down certain amino acids.
The problem can be with the enzyme itself or with the membrane transport system for amino acids (how amino acids move around)
If the body can't break down amino acids, they build up and cause serious health problems like
mental retardation in newborns and often death.
Some Amino-acidopathies; please tell me what they do?
Phenylketonuria
Tyrosinemia
Alkaptonuria
Phenylketonuria (PKU)
PKU is a disease where the body can't break down phenylalanine due to the enzyme that breaks phenylalanine down is missing
phenylalanine hydrolase is the enzyme that is missing
The urine smells like a mouse.
PKU can damage the brain.
Tyrosinemia
Tyrosinemia is a disease where the body can't break down tyrosine and there are 3 types:
Type I is the worst kind and damages liver and kidney, CNS (brain and spine)
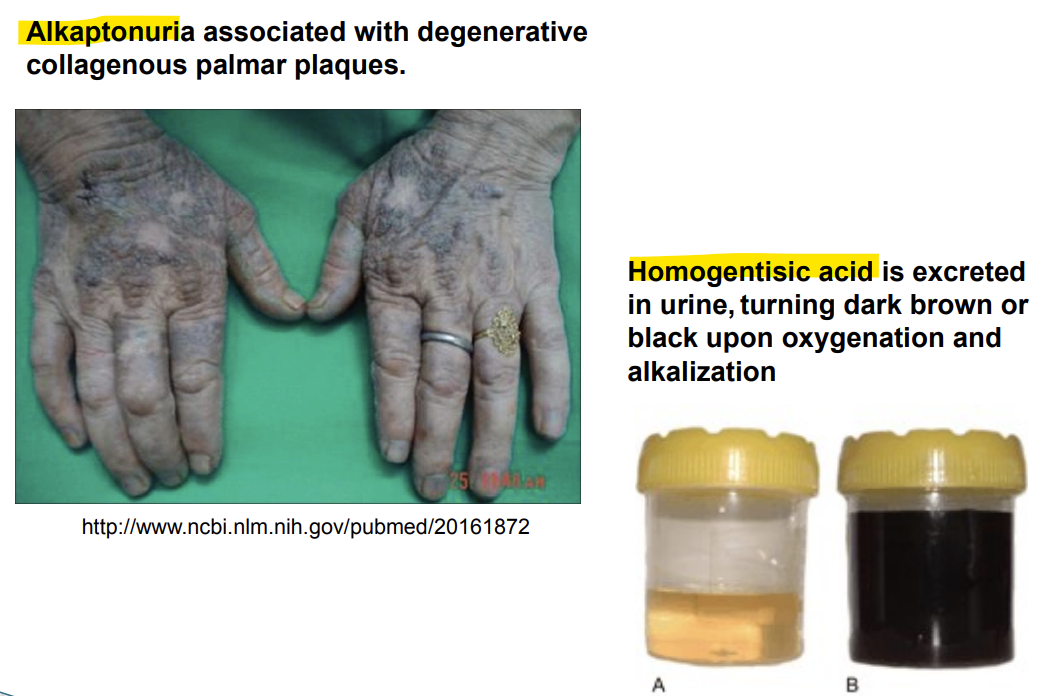
Alkaptonuria
Alkaptonuria is a disease where the body can't break down homogentisic acid due to homogentisate oxidase (enzyme that breaks down homogentisic acid) being missing
Urine turns brownish-black upon exposure to air
Ochronosis (darkening of the tissues of the body) may happen due to excess of homogentisic acid.
A few more Amino-acidopathies; please tell me what they do?
Maple syrup urine disease
Isovaleric Acidemia
Maple syrup urine disease
Absence or reduction of alpha-ketoacid decarboxylase (an enzyme needed to break down certain amino acids.
This disease makes urine smell like maple syrup or burnt sugar in the urine, breath and skin
Isovaleric Acidemia
A disease where the body can't break down isovaleric acid due to the absence of isovaleryl-CoA dehydrogenase enzyme
The body smells like sweaty feet due to build up of isovaleric acid
A few more Amino-acidopathies; please tell me what they do?
Homocystinuria
Argininosuccinic Aciduria
Cystinuria
Homocystinuria
A disease where the body can't break down homocysteine due to the absence of cystathionine-beta-synthetase enzyme
This disease can cause many problems like brain damage, weak bones, heart problems, and blood clots.
Argininosuccinic Aciduria
A disease where the body can't break down argininosuccinic acid due to the deficiency of argininosuccinic acid (ASA) lyase enzyme
This disease leads to Citrullinemia (genetic disorder where the body can't properly remove ammonia, leading to high ammonia levels in the blood and brain damage)
Cystinuria
A disease where the body can't re-absorb certain amino acids properly due to a problem with amino acid transport system (how amino acids move around)
The body has trouble absorbing cystine, lysine, arginine, and ornithine
Cystine cause complications due to its insolubility (doesn't dissolve well.)
Cystinuria disorder results in what visible reults?
what can it also cause?
Cystinuria
Cystinuria is a genetic disorder that causes the body to excrete excessive amounts of cystine (an amino acid) into the urine since they can’t get reabsorb
Cystinuria is the disorder, and cystine crystals (or stones) are the visible result
cystine crystals are flat, colorless, hexagonal plates that form more readily in acidic urine
If these crystals grow larger and aggregate(combine), they can result in kidney stones, which are a common complication of cystinuria
What does CA law say about newborns
When does it have to be done?
California law says all newborns must be tested for certain diseases: PKU, Galactosemia, Thyroid problems, and Sickle cell, etc.
The blood test is done between 12 hours and 6 days after birth.
might be inaccurate if <12 hrs.
what are proteins?
where are proteins found?
Where are most protein made and by what cells
What are the exceptions to proteins? and why
1) Proteins are:
macromolecules that are made up of polymers of amino acids
found in cells, fluids, secretion (release of useful substance like hormones, enzymes, etc.), excretion (remove of waste product from body)
2) Most proteins are made in the liver, where specialized liver cells (hepatocytes) release them into the bloodstream.
3) Immunoglobulins aka antibodies are also protein but are an exception because they are made by plasma cells (a type of white blood cell), not the liver
Plasma cells are primarily located in the bone marrow, lymph nodes, and spleen.
Define monomers
Define macromolecules
Define polymers
define catabolism
define denaturation
A monomer is a small molecule
monomers can combine to form larger molecules
Macromolecules are large and complex molecules, such as proteins, carbohydrates, nucleic acids, or polymers, which are made up of smaller building blocks called monomers
Polymers are large molecules made up of repeating units of monomers bonded together
Catabolism is the process where the body breaks down large molecules (like carbohydrates, proteins, or fats) into smaller ones to release energy
Denaturation happens when a protein loses its normal shape because of external factors like heat, acids, or chemicals.
when a protein denatures, it cannot work the same way anymore
there are other molecules that can denature too not just proteins
What are all protein made out of?
How are protein different from sugar and fats?
what charges do proteins have?
Define isoelectric point?
what is isoelectric point abbreviation?
Proteins are made of carbon, hydrogen, oxygen, and nitrogen, and sometimes sulfur.
Proteins have about 16% nitrogen, this makes them different from sugars and fats.
Proteins carry a charge that changes depending on the pH of their environment.
The isoelectric point (pI) is the pH level where the protein has no overall charge
PI techniques are used for separations of proteins
catabolism means what, one word?
where does catabolism happen
What is made during protein catabolism?
what is the goal of protein catabolism? and
Protein denaturation is what?
1) Catabolism aka breakdown
catabolism mainly happens in the digestive tract (gut), kidneys, and liver.
2) During protein catabolism the goal is to remove nitrogen from the system:
Ammonia is produced which is converted into urea, which excreted in the urine.
Ketoacids are also produced, Ketoacids can then be converted into glucose(sugar) for energy or turned into fat for energy storage
3) Protein denaturation
Denaturation is when a protein's shape changes, and it stops working.
Denaturation is caused by heat, pH change, force, light, chemicals, etc.
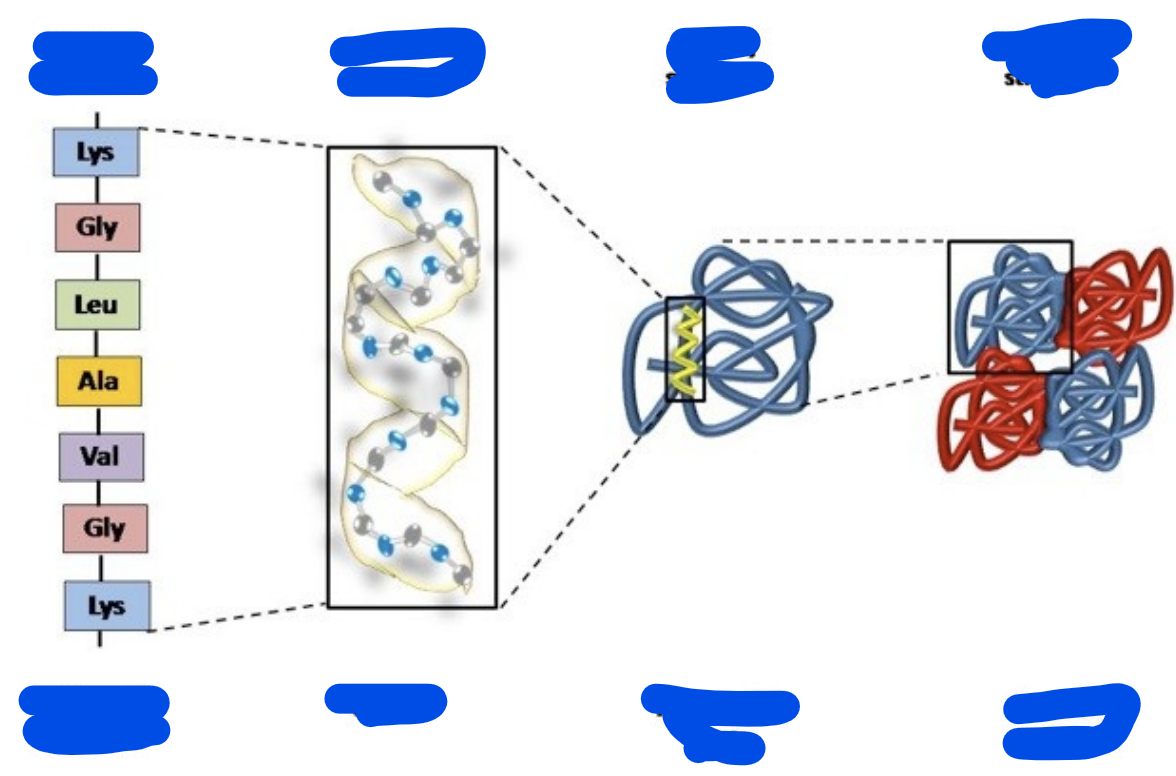
Go in order and describe:
primary structure
secondary structure
Tertiary structure
quaternary structure
Primary structure:
A simple chain of amino acids linked together
A polypeptide chain is when multiple amino acids are together
The secondary structure
The polypeptide chain than interact with itself to form spirals (alpha-helices) or flat sheets (beta-pleated sheets), held together by weak hydrogen bond
The tertiary structure
The polypeptide chain then interacts with itself even more and folds even more and thus, the 3D shape of one individual polypeptide chain protein is formed
The final 3D shape determines the protein’s function
The quaternary structure
2 or more 3D tertiary structures interact with each other to form larger protein complex
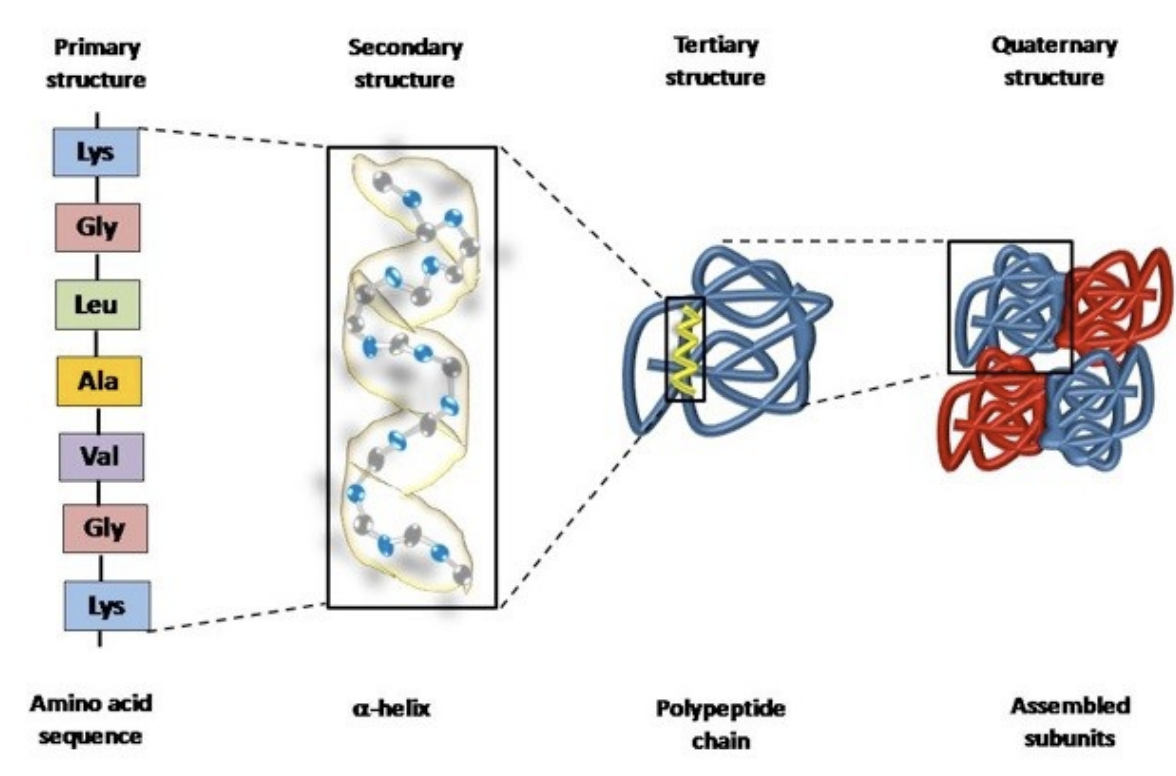
Quaternary structure define again
is albumin a Quaternary structure
is Hemoglobin a Quaternary structure?
what stages can protein denature
what does denature mean
Quaternary structure occurs when two or more 3D tertiary polypeptide structures, come together and interact to form a functional protein complex.
Albumin has only one tertiary polypeptide chain and therefore, would not be considered a quaternary structure.
Hemoglobin on the other hand has 4 globin chains and indeed is considered a quaternary structure.
If anything disrupts the secondary, tertiary, or quaternary structures of a protein—such as changes in pH, temperature, or exposure to chemicals—the protein can lose its shape and become denatured
Even when the protein is fully developed, these disturbances can break the weak bonds holding its structure together, causing the protein to lose its function
Define simple proteins and 2 examples
Define conjugated proteins and define these:
Metalloproteins:
Lipoproteins:
Glycoproteins:
Mucoproteins:
Nucleoproteins:
Phosphoproteins:
Simple proteins, also known as primary proteins, are indeed made up of amino acids linked together
Globular proteins: round
Fibrous proteins: long and stringy
Conjugated proteins, which are proteins with non-protein groups attached, examples are:
Metalloproteins:
metals attached to proteins (hemoglobin with iron).
Lipoproteins:
fats (lipids) attached to protein.
Glycoproteins:
small amount of carbohydrates (<4%) attached to proteins
Mucoproteins:
Large amount of carbohydrates (>40%) attached to proteins
Nucleoproteins:
DNA or RNA attached to proteins
Phosphoproteins:
Proteins attach to substances containing phosphoric acid (e.g., some cell receptors).
Functions of proteins includes:
Enzymes,
Hormones (Send messages to control cells and organs.),
Transport, Immunoglobins aka antibodies, Structural, Storage, Energy
Osmotic force
Help maintain water balance in and between cells and tissues, example includes albumin
Let me know what these proteins do
Myoglobin
Troponin
Brain natriuretic peptide (BNP)
Fibronectin
1) Myoglobin
A protein that stores oxygen in muscle tissue
2) Troponin
A protein that tells us of a Myocardial Infarction (heart attack)
3) Brain natriuretic peptide (BNP)
BNP is a protein that is released by the heart when it's stretched (physical expansion or enlargement of the heart muscles).
Detects heart failure
4) Fibronectin
Fibronectin is a glyco-protein that can help predict if a pregnant woman is at risk of having her baby too early
What does total protein measure?
what can the total protein tell us?
How should total protein specimens be collected?
what should u be careful of?
normal protein level range are what?
1) Total protein measures all of the proteins in our plasma(blood)
2) The amount of total protein in our blood can tell us how well our:
diet, kidneys, liver, and how well other organs are doing
3) To collect total protein specimen (sample)
serum (clotting) or plasma (prevent clotting)
Be careful of hemolysis (breaking of RBC) or lipemia (to much fat in the blood), since they can mess up the total protein results
4) normal protein level is usually
6.4 - 8.3 grams per deciliter (g/dL)

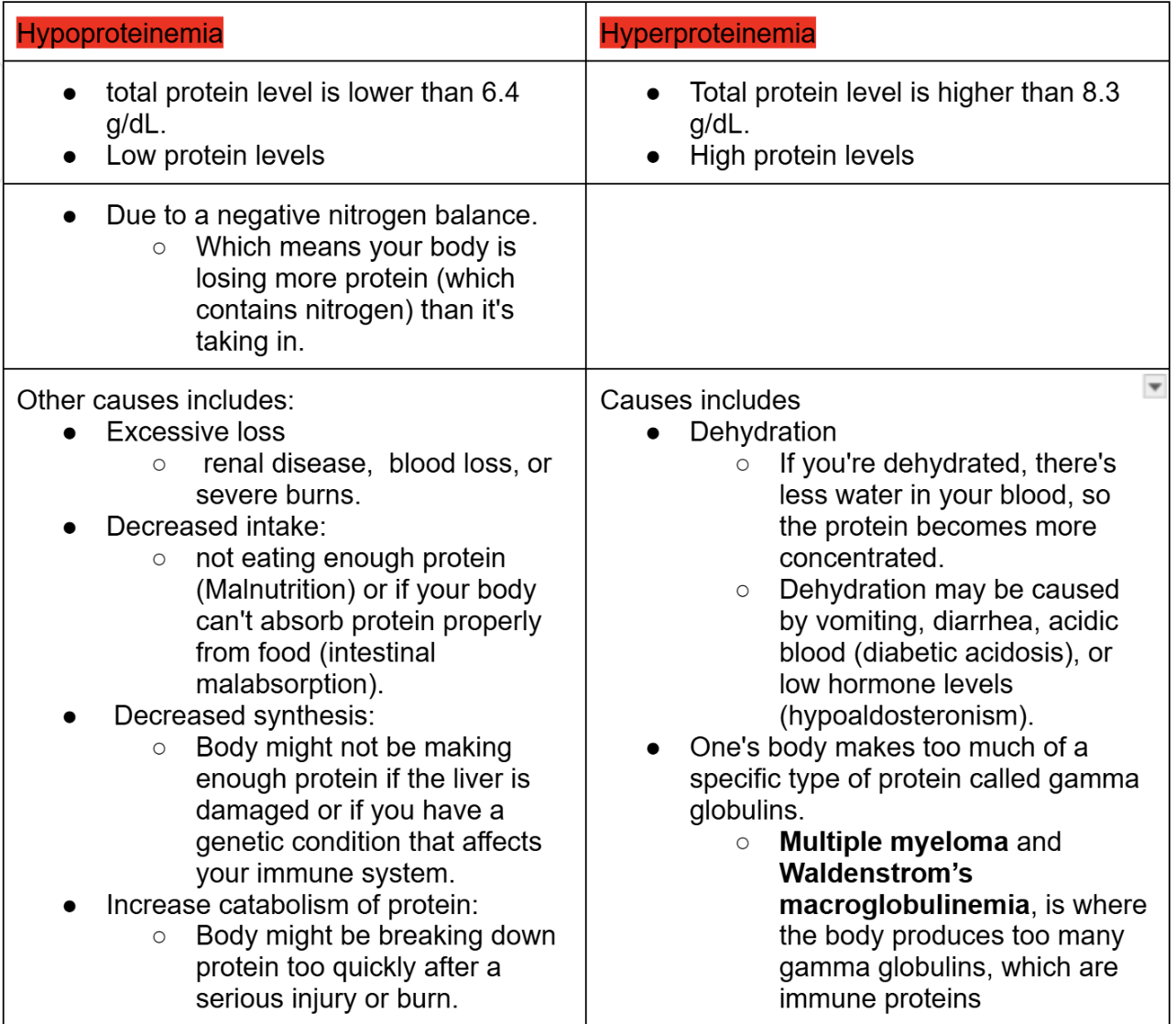
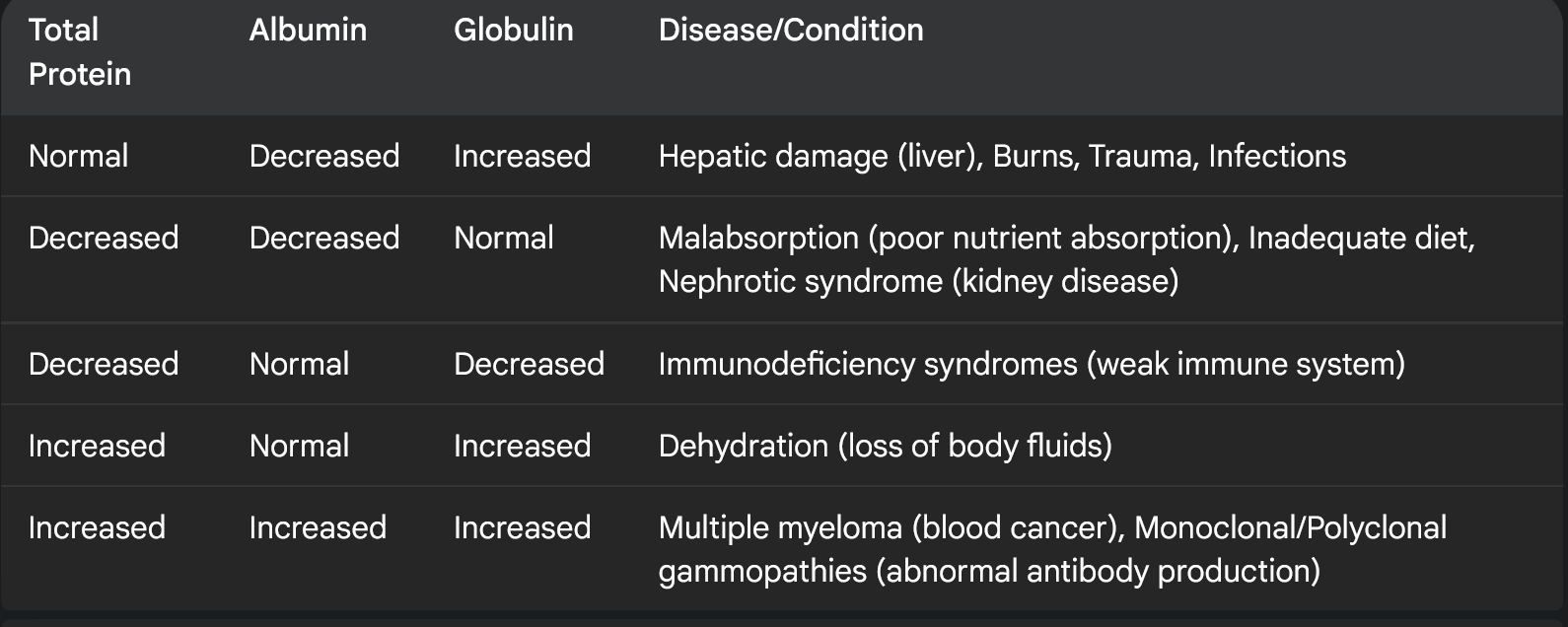
Fill in the box please
tell me about Hypoproteinemia and Hyperproteinemia
what they mean, what level
what causes them
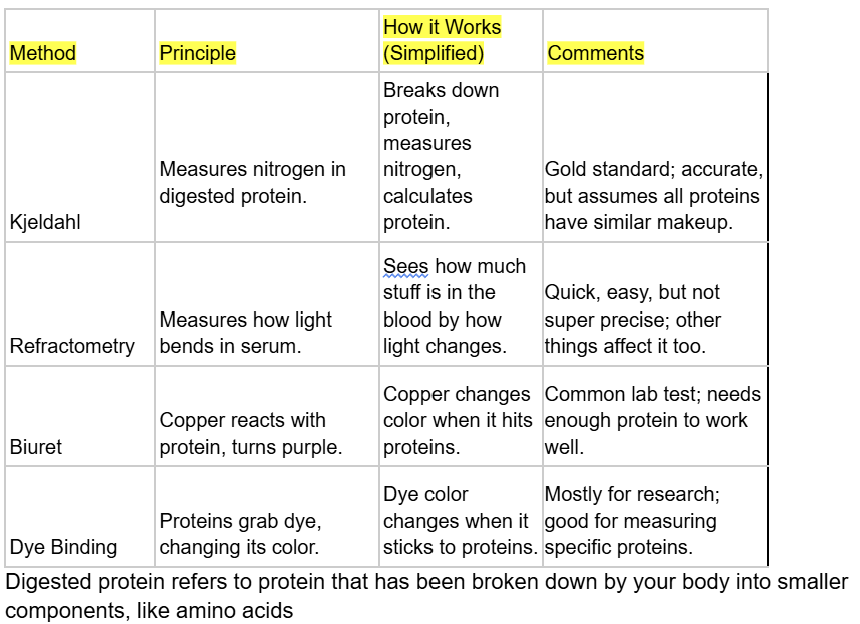
Ways to determine total protein, tell me about each
Kjeldahl
Refractometry
Biuret
Dye binding
Common fluids to look at for protein concentration?
Other fluids we can look at to measure protein concentration?
The most common fluids doctors look at for protein measurement are urine (pee) and CSF (the fluid around your brain and spinal cord).
cerebrospinal fluid (CSF)
Other fluids include:
Fluid around your organs in your belly (peritoneal) in order to lubricates and cushions the organs
fluid found around your lungs (pleural), helps reduce friction during breathing
The protein in your blood can come from where?
If protein shows up in our urine, what does that mean?
What 3 methods did we talk about to test urine for protein? Describe them
Urine Dipstick (Screening Test):
Sulfosalicylic Acid Precipitation:
Quantitative Test (12 or 24-Hour Collection):
1) Protein in your blood can come from
your blood, kidneys, urinary system, or reproductive organs.
2) Our kidneys filter protein out of our blood and then reabsorbs most of it .
If protein shows up in your urine, however, it means the kidneys aren't putting it back correctly.
means they are not being reabsorbed by the renal tubules (go back to kidney chapter if u do not know what this is)
3) Methods used to test urine for various substances, particularly protein
Urine Dipstick (Screening Test):
A quick test using a strip of paper dipped in urine to see if protein is present.
Sulfosalicylic Acid Precipitation:
A method that makes proteins in urine clump together, allowing them to be seen and measured.
Quantitative Test (12 or 24-Hour Collection):
A precise test that measures the total amount of protein in urine collected over a 12- or 24-hour period.
CSF normal range includes?
High range of CSF can be caused by?
CSF:
Normal reference protein range is 15-45 mg/dL.
Higher than normal protein in CSF can be caused by
Infections:
Bacterial, viral, or fungal meningitis.
Injury:
Traumatic tap (blood contaminates the sample).
Nervous system diseases:
Multiple sclerosis, Guillain-Barré Syndrome.
Brain issues:
Tumors (neoplasm), inflammation (encephalitis), or abscesses.
Methods used to detect and quantify proteins in urine and cerebrospinal fluid (CSF)
Turbidimetric
Biuret
Folin-Lowry
Dye Binding
Many more methods too…

What are the 2 main types of proteins in our blood?
Total protein equals what?
if the total protein is normal, does that mean everything is alright?
what does serum protein electrophoresis (SPEP) do?
1) There are two main types of proteins in your blood
Albumin:
This is the most abundant, making up about 60% of all protein in our blood
Globulins:
All the other proteins that aren't albumin.
2) total protein = albumin and globulins
The total protein might look normal, even if there's something wrong. This can happen if, for example, your albumin is low but your globulins are high.
In multiple myeloma, the body makes too many
globulins, so even if albumin is low, the total protein could be in range.
we can use a technique called serum protein electrophoresis (SPEP) to separate albumin and the different types of globulins, so we can see the individual levels of each.
serum protein electrophoresis (SPEP)
When do we do SPEP?
How does all protein differ?
SPEP test, positive charge moves toward what side, negative charge moves towards what side?
How fast a protein moves in the SPEP tests depends on what?
What 5 things do we need for a SPEP test?
Different background slide….IMPORTANT
serum protein electrophoresis (SPEP)
This test is done when your total protein or albumin levels are not normal.
The test separates proteins due to the fact that all proteins have different electrical charges, sizes, and solubility (how well they dissolve.)
In this test, Positive proteins move to the negative side (cathode), and negative proteins move to the positive side (anode).
How fast a protein moves depends on:
The stronger the protein's charge, the faster it moves.
The bigger the protein, the slower it moves.
The thicker the buffer, the slower it moves.
SPEP test needs 5 components:
Electricity to make the proteins move.
A surface (support medium) for the proteins to move on.
A liquid (buffer) that helps the proteins move.
blood sample.
Detection device
More on serum protein electrophoresis (SPEP)
Define ampholyte
What happens in an acidic condition to a protein
pH is lower than the pI, what happens
What happens to a protein in a basic condition?
pH is higher than the pI, what happens?
How does a protein charge become stronger?
Define isoelectric point? abbreviation?
How does the PI relate to the SPEP?
An ampholyte is a molecule (like a protein) that can act as both an acid and a base, meaning it can gain or lose protons depending on the pH of its environment.
In acidic conditions, proteins gain protons and becomes positively charged.
If the pH is lower than the pI, the protein is positively charged.
In basic conditions, proteins lose protons and becomes negatively charged.
If the pH is higher than the pI, the protein is negatively charged.
1) The farther the buffer's pH is from the protein's pI, the stronger the protein's charge becomes.
2) The isoelectric point (pI) is the pH at which a molecule (protein in our case) carries no net electrical charge, meaning it is electrically neutral
When the protein is placed in the electric field, it starts moving because it has a charge (positive or negative)
As it moves through the gel, it encounters different pH levels.
Once it gets to a certain pH, the pH makes the protein have a neutral charge and the protein stops move
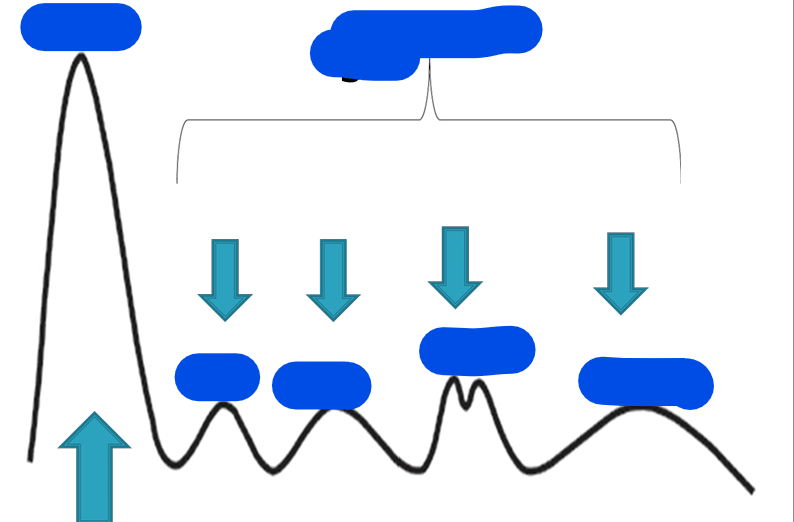
What do we do after the electrophoresis phase for a serum protein electrophoresis (SPEP) test?
What does a densitometer do
what does a densitometer chart show?
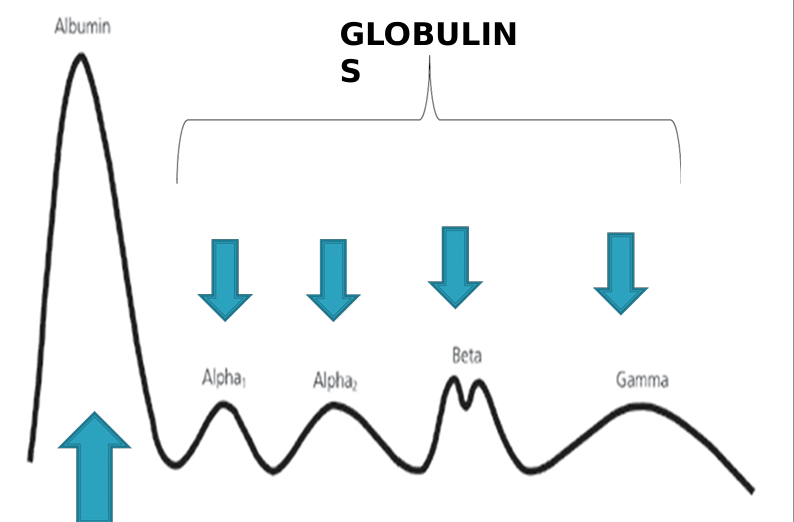
After the electrophoresis, the gel is stained aka dyed to make the protein bands visible.
A machine called a densitometer reads how much light the bands absorb and creates a chart showing the pattern.
The concentration of each protein band is calculated as a percentage.
The chart labels the different protein bands:
albumin,
the globulin regions which includes:
alpha 1
alpha 2
beta,
gamma
Globulin formula
A/G means what
No need to memorize table, just for fun
Globulin = Total Protein - Albumin
A/G ratio
albumin concentration by the calculated globulin.
what is pre-albumin aka
what 2 things can pre-albumin due for us that we discussed?
Albumin is made where ?
what does albumin do? There are 4 we talked about
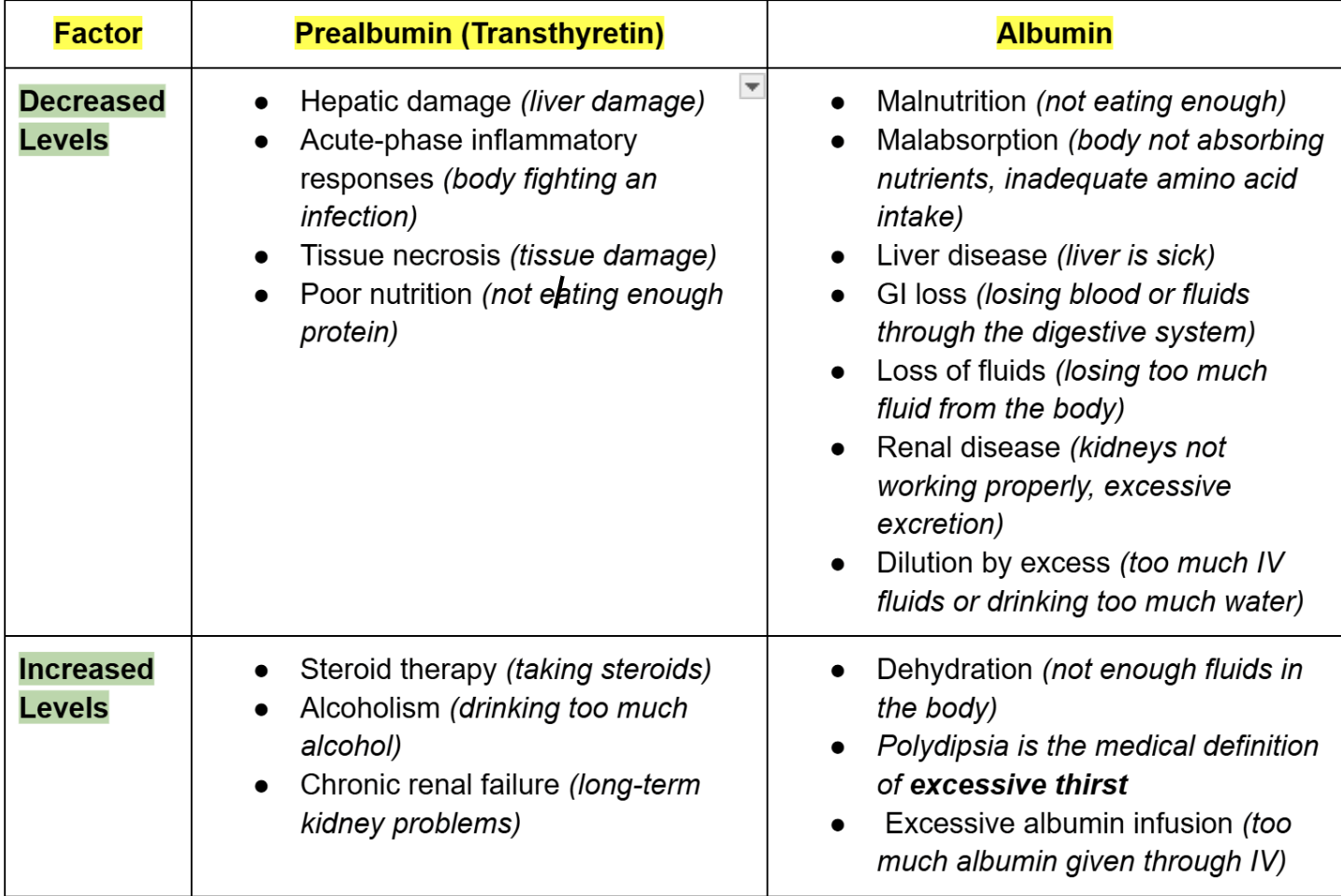
Pre-albumin aka transthyretin is a protein that moves faster than albumin in electrophoresis
Low levels of Pre-albumin are often a sign of poor protein nutrition
Also, a transport protein that carries thyroid hormones and Vitamin A
Albumin is made in your liver and is the most common protein in your blood. Albumin:
Maintains fluid balance in blood vessels aka maintains colloid osmotic pressure
Buffers pH aka keeps our blood’s pH balance
negative acute phase reactant which means albumin decreases when there a sudden inflammation
Binds to substances in the blood and carries that around
Prealbumin vs albumin
What 4 group of globulins proteins will we discuss?
Globulins are a group of proteins in your blood, divided into four main parts which all have different proteins. We will focus on the ones that we mostly encounter in the lab:
alpha 1 aka α 1 fraction
alpha 2 aka α 2 fractions
beta aka ß fraction
gamma aka y fraction
negative acute phase reactant
positive acute phase reactant
acute phase reactant
A negative acute phase reactant is a protein in the blood whose levels decrease in response to inflammation or stress in the body
A positive acute phase reactant is a protein in the blood whose levels increase in response to inflammation, infection, or injury.
When the term acute phase reactant is used without specifying "positive" or "negative," it refers to proteins that change in concentration in response to inflammation
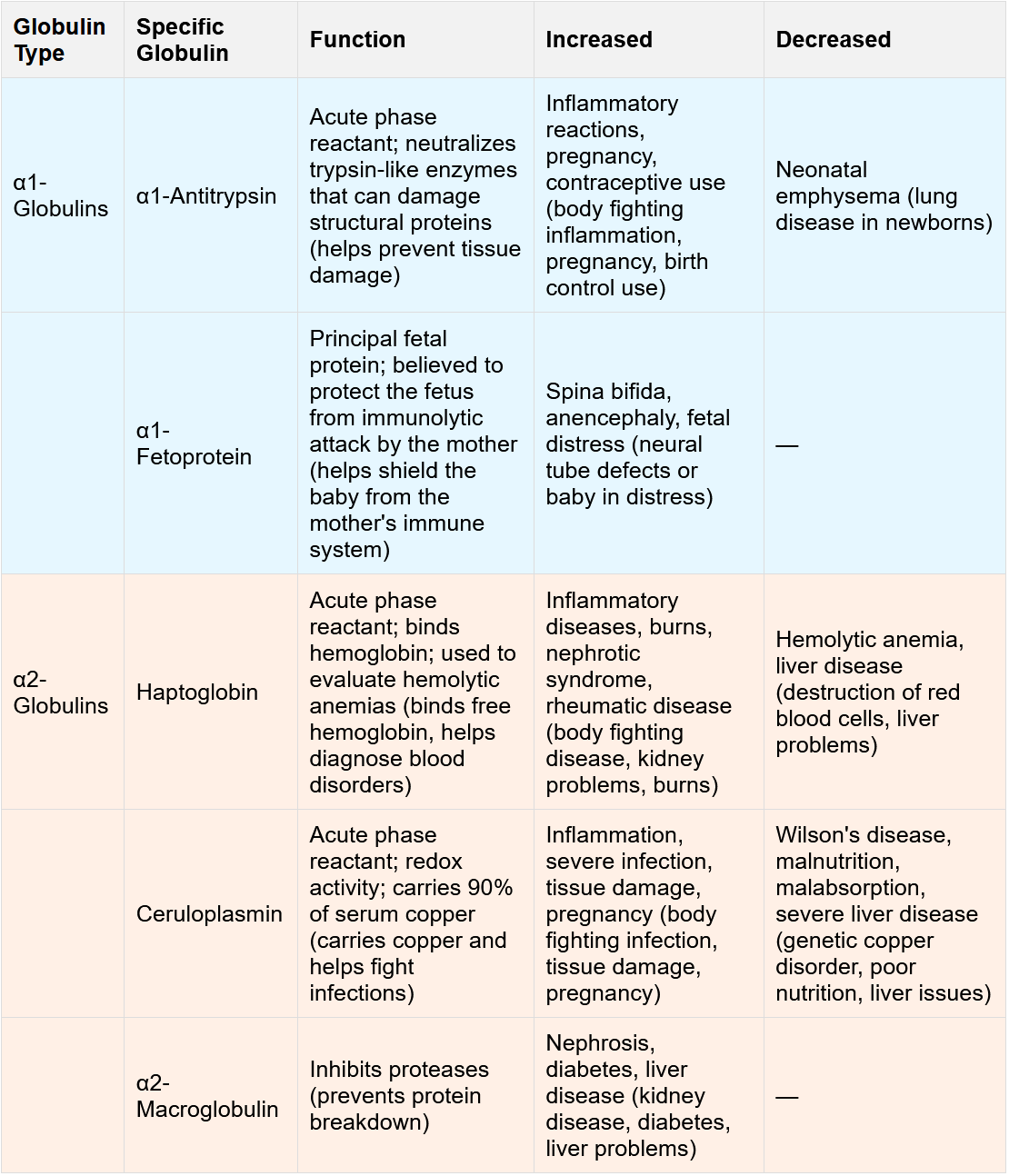
name alpha 1 globulins and functions
name alpha 2 globulins and functions
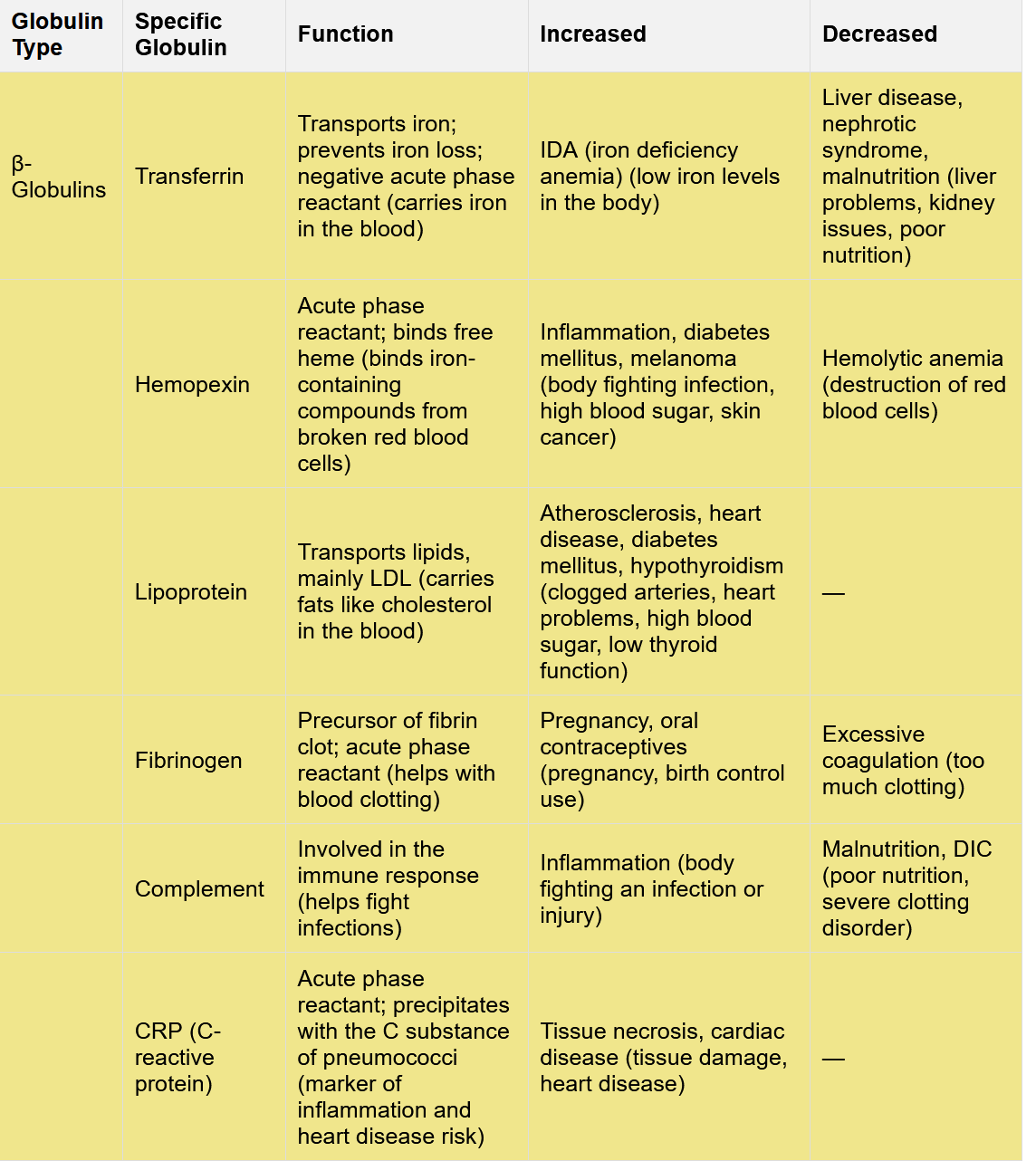
Name the beta globulins and functions
Name the γ- Globulins and their functions?
All of these y-globin are immunoglobulin proteins
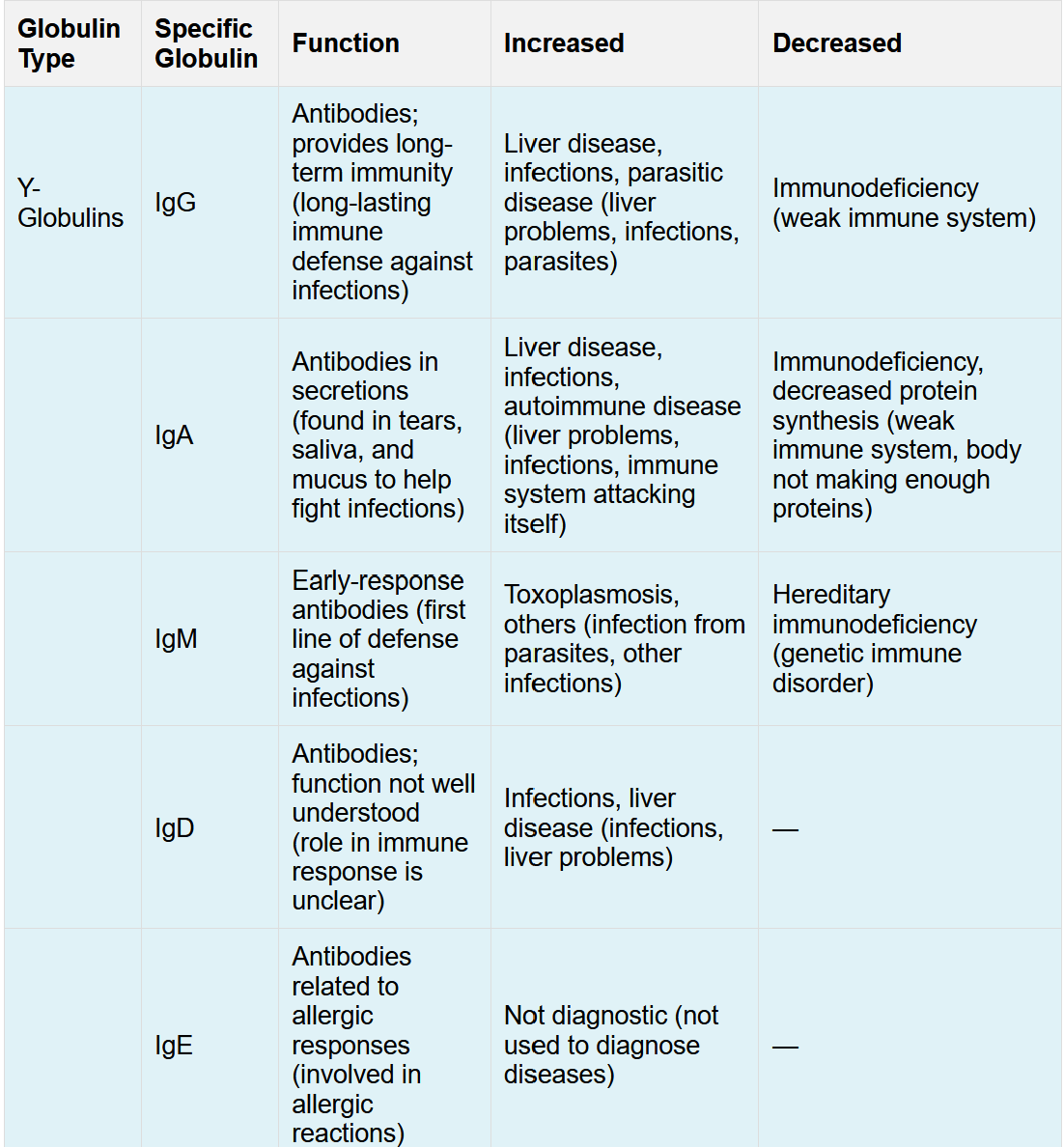
what are γ- Globulins?
Where are γ- Globulins made?
γ-Globulins aka gamma globulins are immunoglobulins protein
immunoglobulins are antibodies that play a key role in the immune system.
γ-Globulins are made by plasma CELLS!!!!
KNOW
What can’t neonates do?
What are antibodies aka?
antibodies are made up of what?
Newborn (Neonates) babies can't make their own immunoglobulins aka antibodies (except for one type, IgM).
Instead, babies get antibodies from their mothers to protect them.
immunoglobulins aka Antibodies are made of proteins. They have two pieces, 2 heavy chain and 2 light chains
Heavy Chains (H): These are the bigger parts of the structure and there are five types: alpha, delta, gamma, epsilon, and mu.
The type of heavy chain decides the isotype ((IgA, IgD, IgG, IgE, IgM,)
Light Chains (L): These are smaller pieces and can either be lambda or kappa.
Define monoclonal immunoglobulins
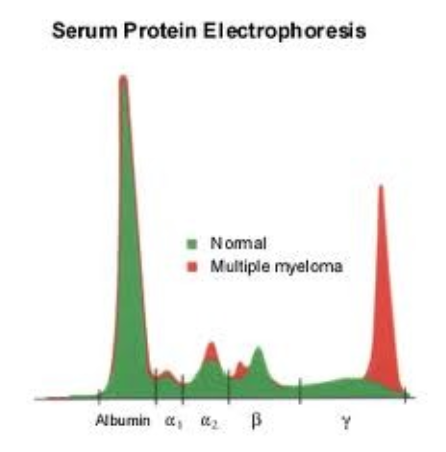
what peak will be the highest?
immunoglobulins aka Antibodies that come from one plasma cell are called monoclonal immunoglobulins
If the plasma cells become cancerous, they start overproducing immunoglobulin aka γ-globulins (gamma globulins)
When this happens, there is a huge peak on the Serum Protein Electrophoresis chart
Define globulins
what globulin includes the immunoglobulin?
what are immunoglobulins?
what type of cell is a plasma cell?
what makes plasma cells?
what do plasma cells produce?
Globulins are a broad category of blood proteins that include alpha (α), beta (β), and gamma (γ) globulins.
only the γ-globulins (gamma globulins) include immunoglobulins, which are antibodies produced by plasma cell
A plasma cell is a specialized type of white blood cell that plays a vital role in your immune system.
Plasma cells are made from B lymphocytes (B cells) and plasma cells produce antibodies (immunoglobulins).
explain what happens to the peak on the SPEP test base on these conditions:
Alpha (α) 1-Antitrypsin deficiency:
Nephrotic Syndrome (kidney problem)
Inflammation (acute phase reactant)
Cirrhosis (liver disease)
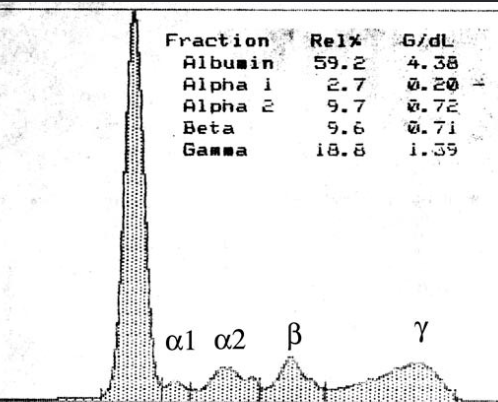
What is this chart showing?
This is a normal strip and normal densitometer scan.
All 5 fractions are within their respective reference intervals
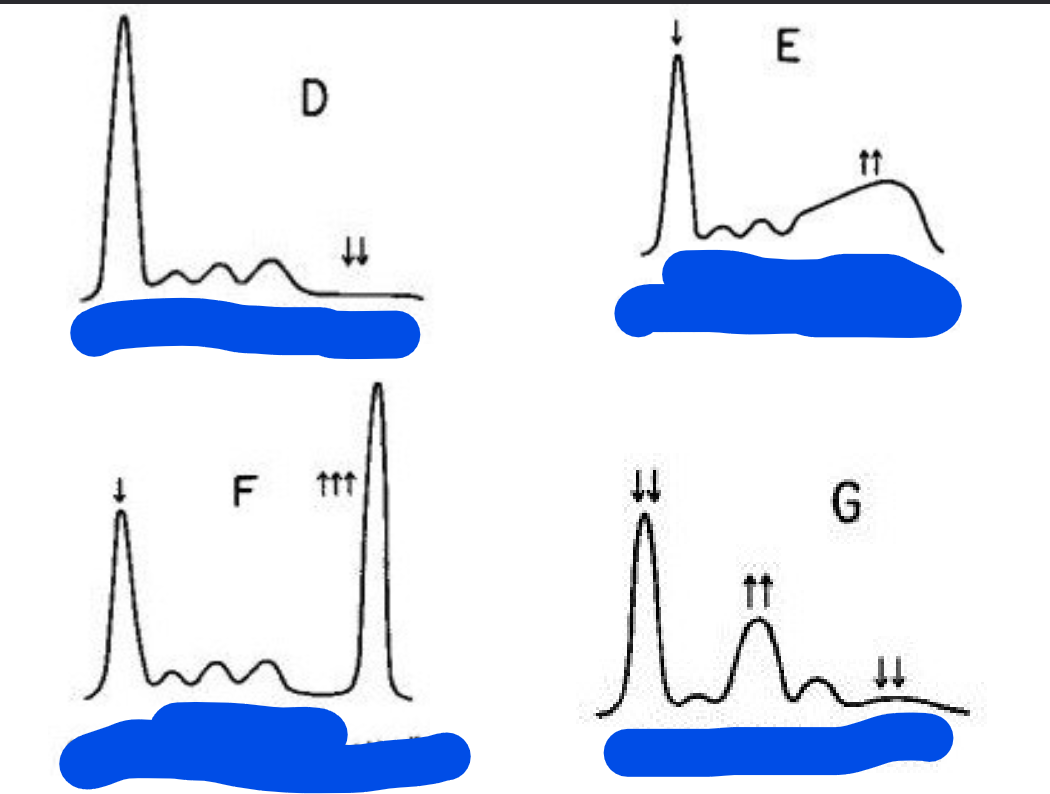
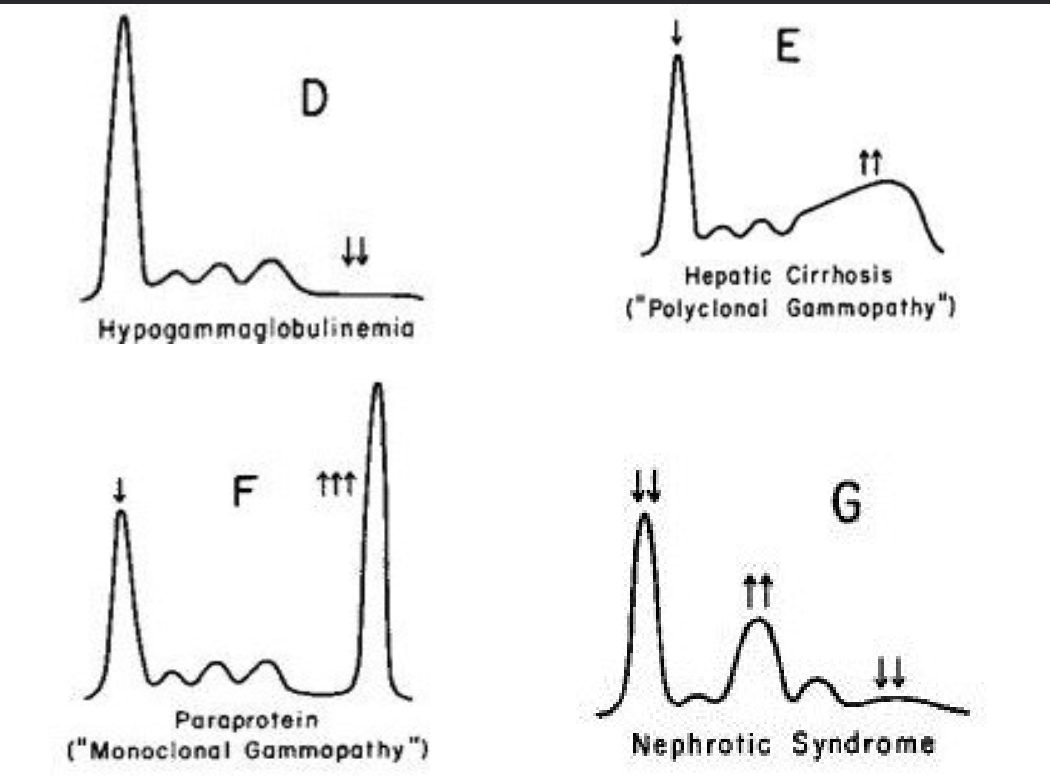
Label each graph with each status
D) Hypogammaglobulinemia
E) Hepatic Cirrhosis
F) Monoclonal Gammopathy
G) Nephrotic Syndrome
Define Monoclonal Gammopathies
What are some diseases that fall under the Monoclonal Gammopathies category?
1) Monoclonal Gammopathies is when a single ß-cell clone produces way too much plasma cell, plasma cell then produces way to many antibodies (immunoglobulins).
2) There are several different diseases that fall into this category, each with different levels of severity:
MGUS (Monoclonal Gammopathy of Undetermined Significance):
Smoldering Multiple Myeloma:
Multiple Myeloma:
Waldenstrom's Macroglobulinemia:
This is another type of cancer where a specific type of antibody (IgM) is produced in excess.
Mutiple Myeloma can cause?
Major complication multiple myeloma can cause
Multiple myeloma can cause a lot of problems:
bone pain, low red blood cell count (anemia)
tiredness (fatigue), holes in your bones (bone lesions),
high calcium levels (hypercalcemia), and kidney/renal problems.
Major complication includes:
Infections and kidney failure.
High calcium levels and thick blood (hyper viscosity) can happen.
Multiple myeloma with IgA antibodies is generally more severe than with IgG antibodies.
Broken pieces of antibodies (Bence Jones proteins) show up in the urine.
These broken antibody pieces are called paraproteins and are light chain segments.
Mutiple myeloma tends to have a poor outcome, and there's a higher chance of developing leukemia.
Waldenstrom's Macroglobulinemia is less or more severe than multiple myeloma
why?
What will you still experience?
People with Waldenstrom's Macroglobulinemia have what in their urine, usually?
Waldenstrom's Macroglobulinemia is less severe than multiple myeloma.
You'll experience less bone pain, fewer bone problems, and fewer bacterial infections.
However, you will still experience
thick blood (hyper-viscosity)
About 10% of people get nerve damage (peripheral neuropathy).
~30-40% have
lymphadenopathy (swollen lymph nodes)
splenomegaly (enlarge spleen), and/or
hepatomegaly (enlarge liver)
Bence Jones proteins (broken antibody pieces) are found in the urine of at least half the patients.
remember since igM is produced in excess for this disease,
(IgM) is different from what's seen in multiple myeloma.
what is analyzed in a Urine and cerebrospinal fluid (CSF) Electrophoresis test?
what do we have to do first and why?
Urine Protein Electrophoresis explain
Cerebrospinal fluid (CSF) Electrophoresis explain
If there are bands that are present in these tests but absent in the SPEP tests, what does this mean?
1) Urine and CSF Electrophoresis test can be used to analyze proteins in urine and cerebrospinal fluid (CSF).
Since there's not a lot of protein in these fluids, we need to concentrate them first.
2) Urine Protein Electrophoresis (UPEP):
We test urine to find broken pieces of antibodies (light chains).
These pieces get filtered into the urine quickly, so they might not show up on a regular blood test (SPEP).
3) Cerebrospinal fluid (CSF) Electrophoresis
we expect to see a protein called pre-albumin.
If someone has multiple sclerosis (MS), you’ll see multiple distinct bands (oligoclonal bands) in the globulin zone of the CSF test.
If these bands are absent in the blood test (SPEP test) but present in the CSF test, it’s significant for multiple sclerosis (MS) or shows that antibodies are being made in the central nervous system.
Multiple sclerosis (MS is what kind of disease?
Multiple sclerosis (MS) is a chronic disease that affects the central nervous system ( brain, spinal cord), and optic nerves.