2.2: Defining and Distinguishing Drugs from Foods, Dietary Supplements, Devices, & Cosmetics
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
90 Terms
Federal Drug Regulation is the
Protection of the public health by ensuring drug products (human and veterinary) are safe and effective
Federal Drug Regulation has a balance of
"Direct regulation" and "Indirect regulation"
Direct regulation for Federal Drug Regulation
FDA Rulemaking authority
Indirect regulation for Federal Drug Regulation
Issue Guidance Documents
The Federal Food and Drug Administration (FDA) is an agency within the
Department of Health and Human Services (DHHS)

Authority for administering the FDCA is vested in the
Secretary of DHHS.
The _________ appoints a commissioner of the FDA who is accountable to the Secretary
President
There are _____ Headquarter offices within the FDA
13
3 example of Headquarter offices in the FDA
Office of Regulatory Affairs
Office of Operations
Office of Import Operations
FDA consists of ___ Center level organization
9
Notice and Consent Rulemaking allows for public input on proposed regulation _________ the FDA issues a final regulation
before
Notice and Consent Rulemaking falls under what title
Title 21 of the Code of Federal Regulations (CFR)
who has a voice in the rulemaking process?
experts, stakeholders, and public
T/F
FDA regulations are federal laws but they are not part of the FDCA
true
FDA issues ________ ____________ which is a less formal way of regulation that describes the agency's current thinking on a regulatory issue
Guidance documents
T/F
Guidance documents
FALSE
FDA incorporates advice from standing committees of outside experts such as
physicians, pharmacists, nurses

What does the FDA regulate?
Foods, drugs, biologics, medical devices, electronic products that give off radiation, cosmetics, veterinary products, and tobacco products
T/F
FDA approves animal drugs and food additives for use in food for animals
true

A drug is recognized by what three organizations
United States Pharmacopeia (USP) or official homeopathic pharmacopeia, or official National Formulary

Establishing that an article is a drug, as opposed to a food, dietary supplement, or cosmetic, provides the FDA with considerably more ________ over the article.
authority!
What established prescription and OTC drugs
Durham-Humphrey Amendment of 1951
Two things in order to change the status of a drug from OTC to prescription
1. the ______ and __________ of use require physician supervision
2. ___________ measures for use require physician supervision
toxicity, method
Collateral
_____has authority to categorize Rx drugs; unsafe for use except under the supervision of a practitioner due to toxicity, method of use, or collateral measures necessary to use the drug
FDA
Rx Drugs are subject to
New Drug Application
a product is a drug if it was intended to diagnose, cure, mitigate, treat, or prevent a disease, or (for articles other than food) was intended to affect the body __________ or ____________
structure
function
A product is a drug if __________ or ____________ claims are made no matter what the disclaimers are in the labeling
therapeutic
structure/function
A product is a drug if the manufacturer cannot ___________ a therapeutic or structure/function claim for a product by proclaiming an article is not a drug
mitigate
A product is a drug if it is the __________- intended use of a product NOT the ____________ intended use
manufacturer
purchaser
Something is a food if it is used for its ordinary ___________ and in an ordinary _________ (people use food for taste, aroma, or nutritive value)
purpose
manner

Foods are excluded in what part of the definition of a drug because it will naturally affect structure of function of the body
Part C- Articles (other than food) intended to affect the structure or any function of the body in man or animals
Two special categories of foods created by the FDA:
Special dietary foods
Medical Foods
Special Dietary Foods are recognized under the ________ and supply dietary need that exists by reason of a _________, _____________ or ___________ condition
FDCA
physical, physiological, pathological

examples of conditions that need supply of dietary need
convalescence, pregnancy, lactation, infancy, allergic hypersensitivity to food, underweight, overweight, or need to control intake of sodium

Medical Foods include food formulated for ________ or _________ use under the supervision of a physician and that are intended for the specific dietary management of a disease or condition for which distinctive ___________ requirements are established by medical evaluation
oral, enteral
nutritional
Examples of a medical food are foods formulated without the amino acid ____________ for people with phenylketonuria
phenylalanine
Medical Foods must be specially formulated, NOT ____________ occurring, and must provide nutritional requirements that would be impossible for the patient to meet through a _________ diet
naturally
normal
______________ and ____________ foods are VAUGE definitions that are NOT specifically defined by law
Nutraceuticals
functional
Functional Foods have been enhanced with
dietary supplements
Example of functional food or neutraceuticals
OJ with Vit D
foods with probiotics

FDA does not approve :
- medical foods
- infant formula
- dietary supplements
- the food label or nutritional facts panel
- structure-function claims on dietary supplements
DA has battled with food manufacturers who have made
health claims for their products
If a manufacturer makes a claim on a food label that fits the definition of a drug, then the FDA may recognize that the manufacturer is claiming that the product can
treat, diagnose, prevent, or cure disease

T/F
FDA can take action against a manufacturer if its claims a product is a drug when its not
true

This act created a new category of FOOD called dietary supplements
Dietary Supplement Health and Education Act of 1994 (DSHEA)
True of False
FDA cannot require premarket approval of "Dietary Supplements
TRUE
FDA must prove what of a dietary supplement before removing it from the market
that it is unsafe
What is the disclaimer on every label of dietary supplement
"These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease
___________ permits manufacturers to make limited health claims that describe the relationship of a food substance and disease
DSHEA
FDA must approve the claim made by these manufacturers by regulation pursuant of the _____________________ standard
"significant scientific agreement
what claims dont require a disclaimer if it meets the significant scientific agreement
unqualified health claims
What claims require the disclaimer when a claim does not meet the significant scientific agreement
qualified health claims
DSHEA provides that a dietary supplement is adulterated if it presents a significant or unreasonable risk of _______ or ______ under the conditions of use recommended or suggested in the labeling or under ordinary conditions of use
illness or injury
This act shows Adverse event reporting requirements for Dietary Supplement manufacturers and non-prescription drug manufacturer
The Dietary Supplement and Nonprescription Drug Consumer Protection Act 2006
Manufacturers, packers, or distributors whose name appears on the label MUST submit to the ______________ program any report of a serious ADR within 15 business days
MedWatch

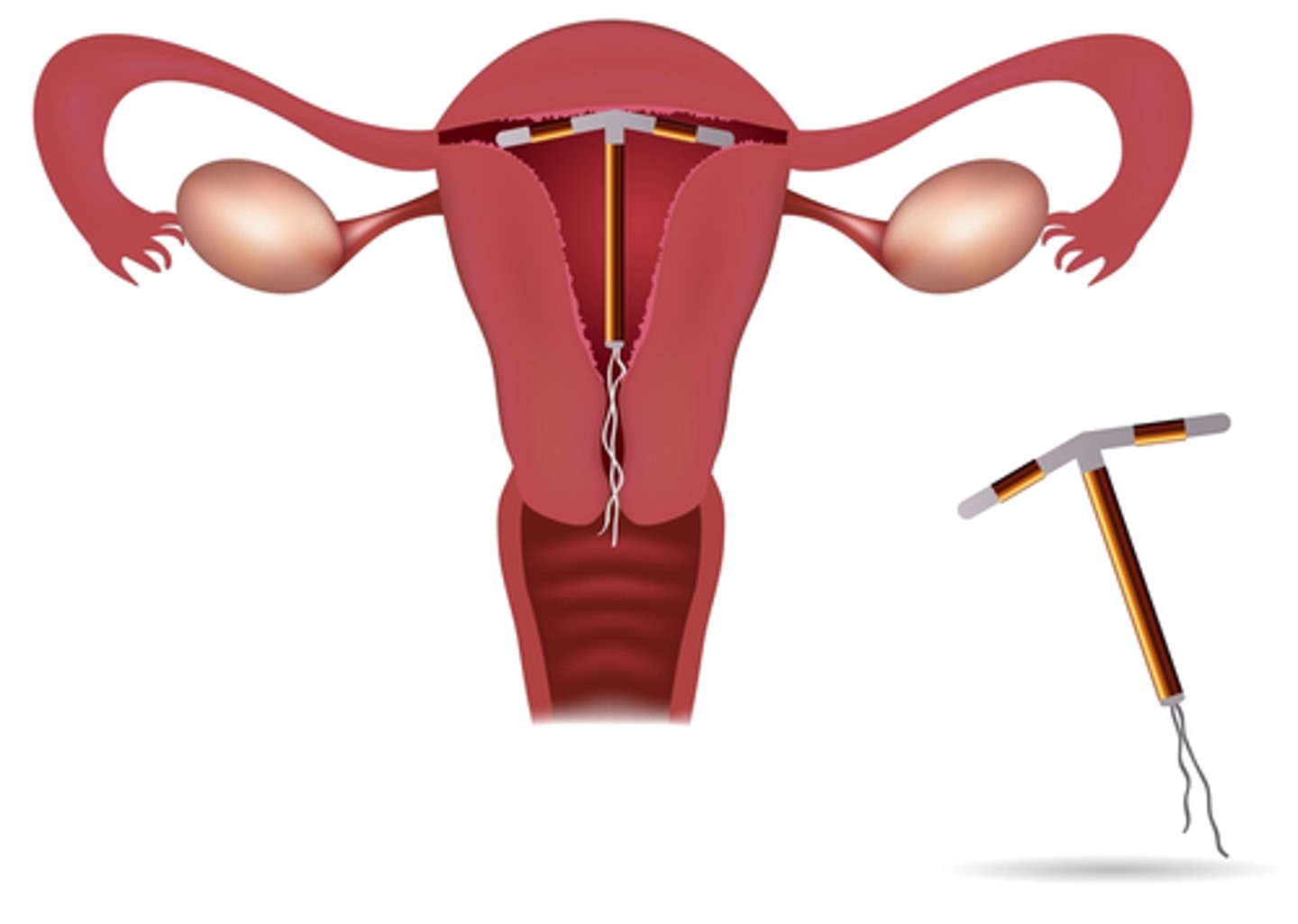
A device differs from a drug because it does not achieve its primary purpose through a ____________ action and not dependent on ___________ for its primary purpose
chemical
metabolism
Some devices are ____________ products that contain drug
combination
A cosmetic can become a drug if the seller makes a health or ______________ claim
structure/function
T/F
FDA does not approve cosmetics
true
___________ prohibits the distribution of cosmetics that are adulterated or misbranded in interstate commerce
FDCA
Some products may be a cosmetic and a drug if a product has
2 intended uses

Can a cosmetic make the following claim?
Cleanses skin and alters appearance
YES

Can a cosmetic make the following claim?
treats acne, eczema or increases collagen
NO
Label means a display of written, printed or graphic matter upon the ___________ container of an article
immediate

Labeling means ALL labels and written, printed or graphic matter upon
any article, its containers or wrappers accompanying it

If the literature is deemed to _______________ the product, it is labeling
accompany
If the literature is deemed NOT to accompany a product, it is
ADVERTISING
Prescription drugs are labeled for the ____________________ not the ______________
healthcare professional
patient

What label exception of a prescription drug is actually for the patient
Medication guide
patient instructions
Regulated specific information required on the commercial label
name/address of Mfr, packer, distributor
name, quantity, proportion of each active ingredient

New package insert requirements
Highlights at the beginning of table of contents
Full Prescribing info
patient counseling information

pamphlet that must"accompany" the drug product and contains the essential scientific and medical information needed for safe and effective use of the drug by healthcare professional
package insert
T/F
package inserts can be promotional in nature
FALSE
Where are package inserts found for reference
DailyMed
Labeling may also include Instructions for
use or medication guides

Pharmacists are required to include FDA approved ______________________ every time a drug is dispensed to help ensure the safe use
medication guides
Medication Guides are _____________ labeling that is part of the FDA approved prescription drug labeling for certain prescription drugs
Patient
Patient labeling could prevent serious __________________
adverse reactions
A drug will be considered misbranded or adulterated under the FDCA if it is not recognized in
USP-NF
Homeopathic Pharmacopeia
__________________ is a private org independent of the FDA created by physicians and pharmacist in 1820 who were concerned about the potency, quality, and composition of medicinal agents and ingredients
USP Convention

_______________ contain the approved titles, definitions, descriptions, and standards for identity, quality, strength, purity, packaging, stability and labeling for a drug
Monographs
The FDA does not subject homeopathic drugs to ______________ approval or require proof of safety or efficacy
pre-market
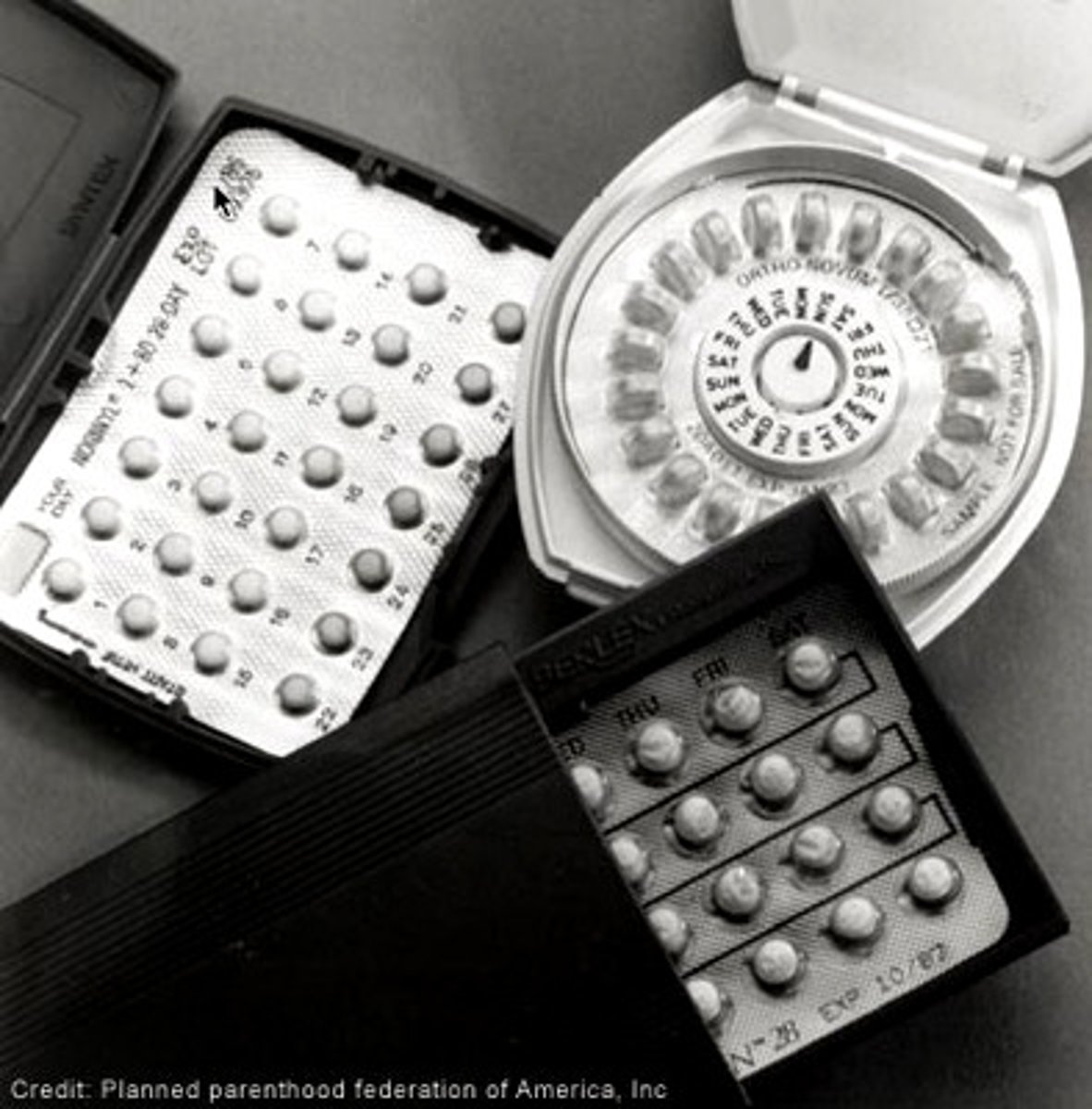
Patient Package Inserts are required for
oral contraceptives and estrogens
T/F
PPIs are patient information and patient labeling that is FDA approved as drug labeling
true
Nonprescription Drug Labeling must be in a ____________- format stating the principle intended use, cautions, warnings, and adequate directions for use and "Drug Facts" panel
User-friendly

Some drugs exists as both OTC and prescription, depending on the _____________ for use
indication
Some indications can be labeled with ______________ directions for use
adequate
It is important for pharmacists to communicate the information on the label for nonprescription drug products accurately and never recommend use for _______________ purposes that are not on the nonprescription drug label or ____________ doses that recommended on the label
unintended
higher

if a patient has a condition that can not be treated by the OTC version of a drug then what is necessary?
guidance of a healthcare professional

Is it unsafe for a patient to use the OTC version product for a condition found on the prescription labeling on the product ?
YES could delay treatment