Atomic Spectroscopy (chptr 20)
1/27
Earn XP
Description and Tags
CHEM 310: Foundations of Analytical Chemistry
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
28 Terms
atomic spectroscopy
determination of elemental composition through evaluation of the characteristic electromagnetic spectrum of atoms
atomic emission spectroscopy (AES)
emission from a thermally populated state
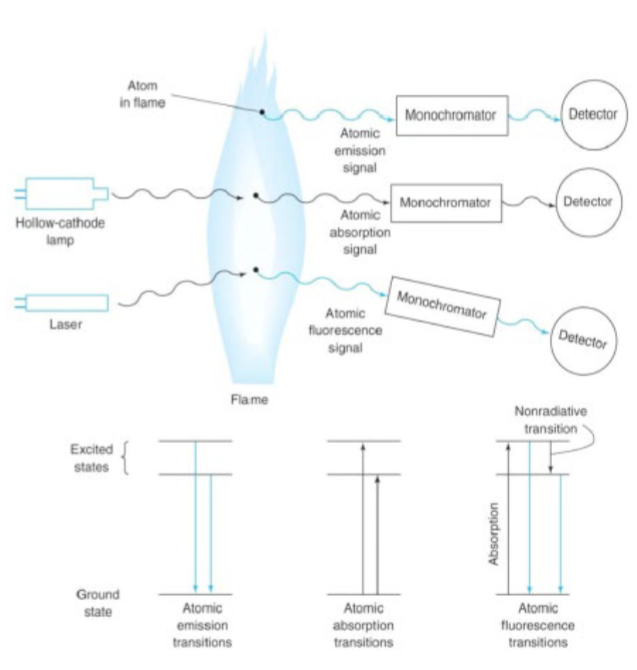
atomic absorption spectroscopy (AAS)
absorption of sharp liens from a hollow cathode lamp
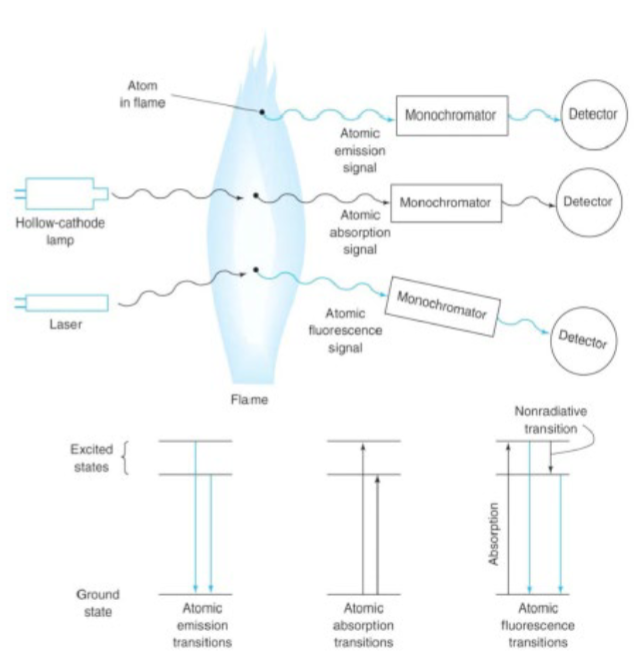
atomic fluorescence spectroscopy (AFS)
fluorescence following absorption of laser radiation
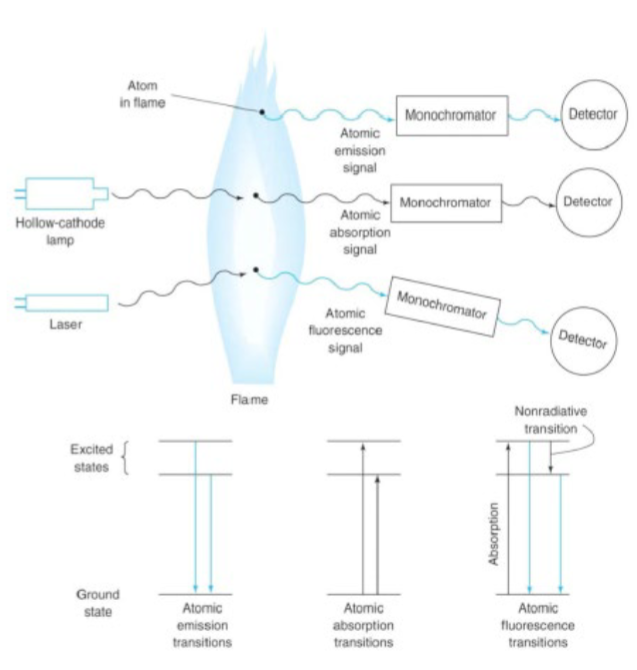
atomization
the process of converting an analyte in solid, liquid or solution form to a free gaseous atom; converting solution into vapor-phase free atoms
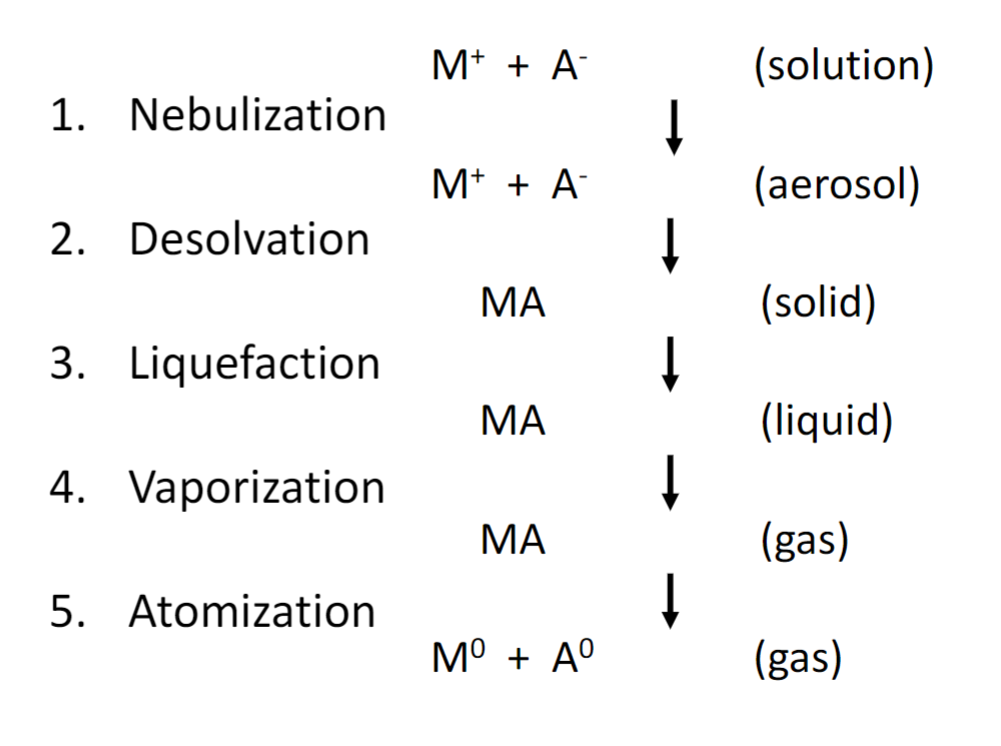
5 steps for atomization
nebulization
desolvation
liquefaction
vaporization
atomization
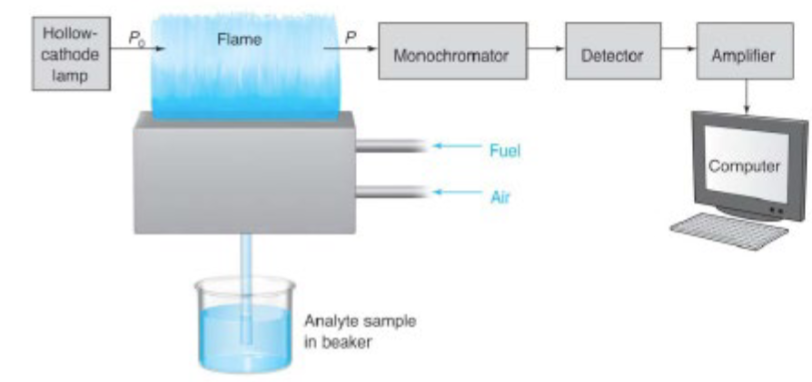
AAS: By measuring the amount of light absorbed, …
a quantitative determination of the amount of analyte element present can be made.
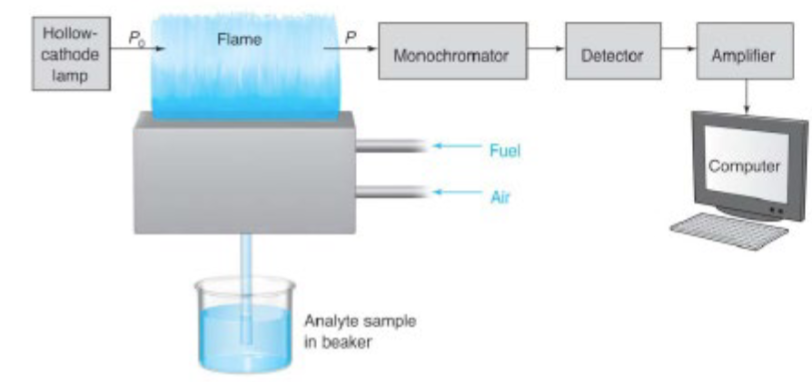
AAS: Absorption spectra of atoms are…
simpler and appear as sharp absorption lines because electronic states do no have vibrational or rotational levels.
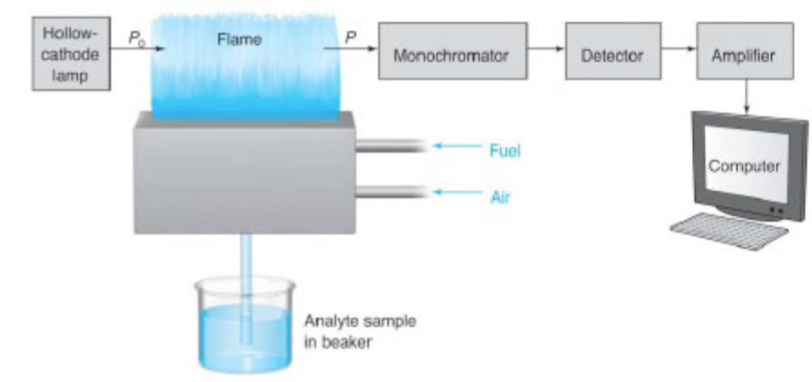
AAS: Use of special light sources and careful selection of wavelength allows
the specific quantitative determination of individual elements in the presence of others.
AAS: Ease and speed to get precise and accurate measurements makes it ideal for…?
metal analysis!
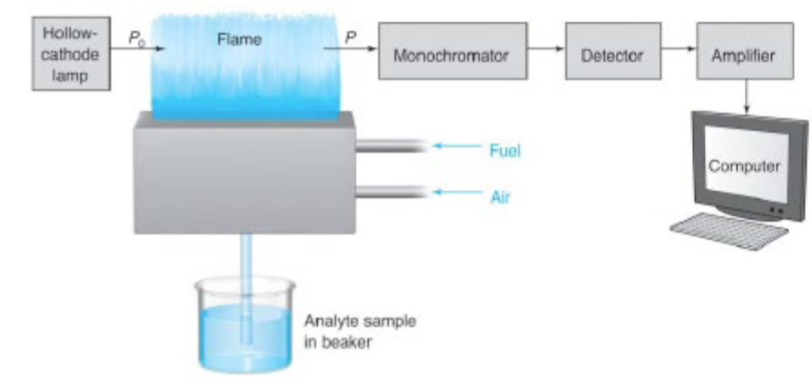
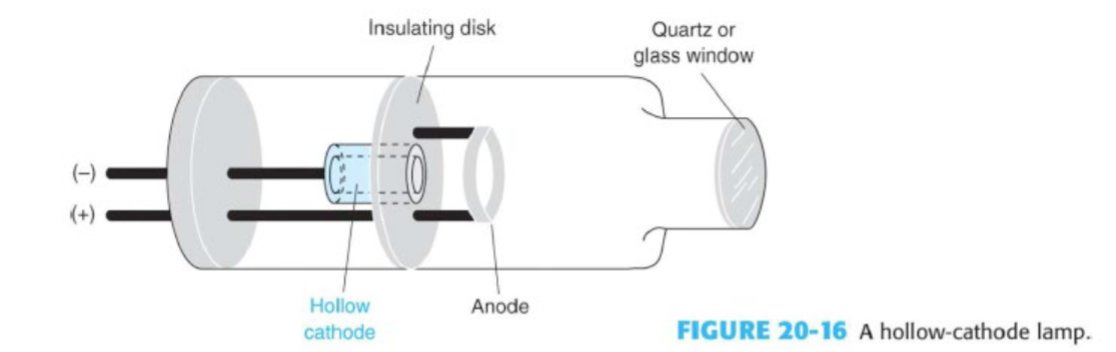
AAS light source: the hollow cathode lamp
inert gas atmosphere at a low pressure
atoms of the gas are ionized in the electric discharge and accelerated to the cathode to dislodge atoms of the cathode metal (sputtering)
collisions with high energy electrons excites sputtered metal atoms
they relax to emit a line spectrum characteristic of the cathode metal
typically limited to one metal
atomization by flame: pre-mixed burners
mixes the fuel, oxidant, and sample before introducing the mixture to the flame
sample is drawn into the nebulizer by the rapid oxidant (usually air) flow past the tip of the sample capillary
spray is directed into a glass bead where droplets of sample is further broken into smaller particles
i.e. nebulization, forming an aerosol
fuel also feeds into the chamber, sample is swept and introduced into the mixed fuel and oxidant gases in the burner
atomization by furnace: graphite furnace
more sensitive than flame and uses less sample
a few uL of sample is placed on graphite rod or within the depression of a tiny graphite crucible
solvent is evaporated by passing a current, temperature is increased very rapidly to 2000-3000C within a few seconds
confines atomized sample in the optical path for several seconds (usually <1s)
leads to higher sensitivity

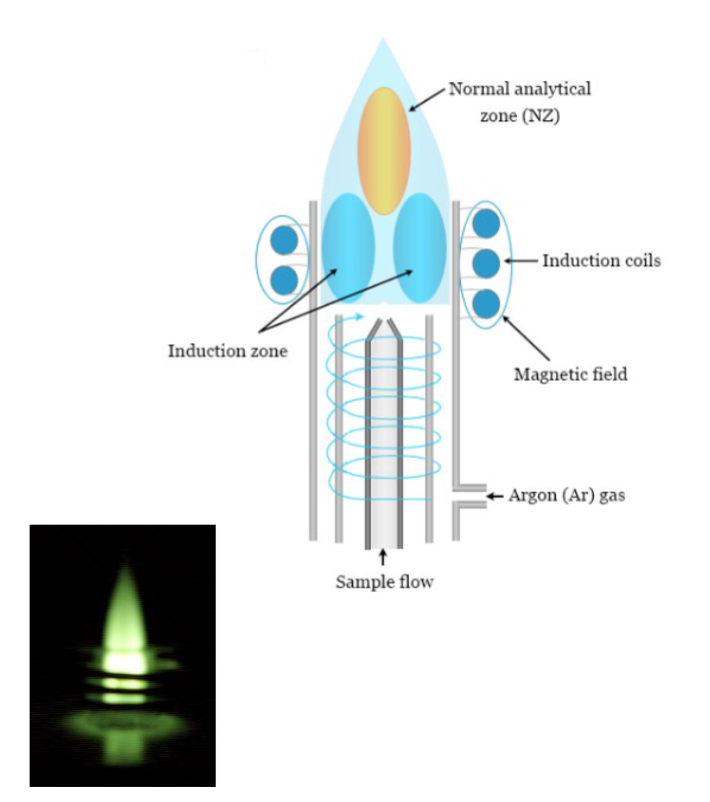
atomization by plasma: inductively coupled plasma (ICP)
twice as hot as a combustion flame
the high temperature, stable, and inert gas environment eliminate much of the interference found in the flame
costs more to purchase and operate than a flame
AES operation
sample is subjected to a high thermal energy environment in order to produce excited state atoms, capable of emitting light
ideal AES source
complete atomization of all elements
controllable excitation energy
sufficient excitation energy to excite all elements
inert chemical environment
no background
accepts solutions, gases, and solids
tolerant to various solution conditions and solvents
simultaneous multi-element analysis
reproducible atomization and excitation conditions
accurate and precise analytical results
inexpensive to maintain
ease of operation
3 potential AES sources
flames
electric discharge (like arcs or sparks)
plasmas
AES source: flame
background signals due to flame fuel and oxidants
continuum emission from recombination reactions
like bands of atomic origin, these molecular bands are fairly broad:
H + OH —> H2O + hv
CO + O —> CO2 + hv
flames used in AES nowadays are only used for a few elements
it’s cheap, but there are limitations
flame AES is often replaced by flame AAS
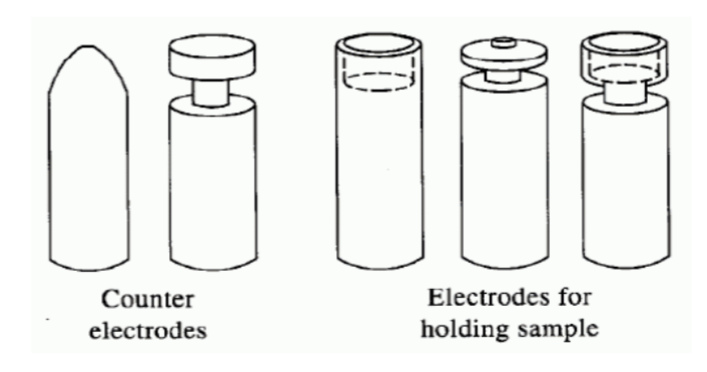
AES source: electric discharge, arcs and sparks
arc - an electrical discharge between two or more conducting electrodes
spark - an intermittent high-voltage discharge (few microseconds)
limited to qualitative and semi-quantitative use
particularly useful for solid samples (which get pressed into the electrode)
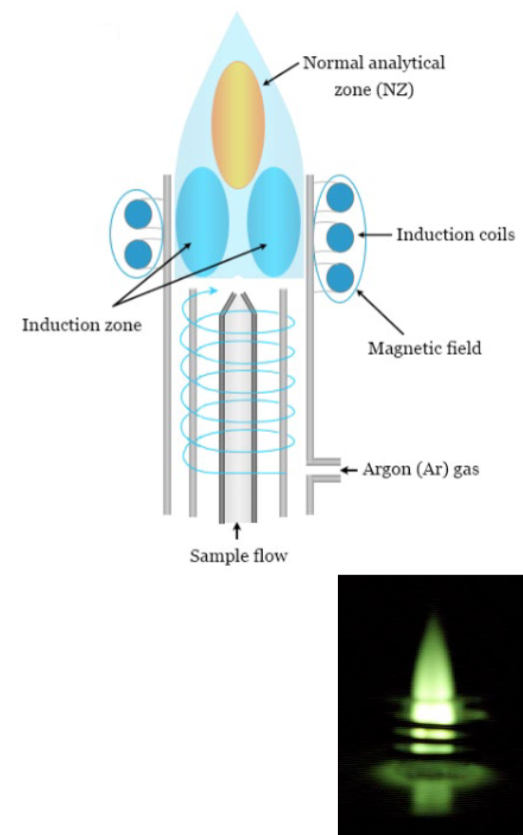
AES source: inductively coupled plasma
long residence time results in high desolvation and volatilization rate
high electron density suppresses ionization interference effects
simultaneous multi-element analysis possible
spectral interference is more likely for plasma than for flame due to the larger population of energetically higher states
background: Ar atomic lines
Costs more than $50k
operating costs are relatively high due to the Ar cost (10-15 mL/min) and training needed
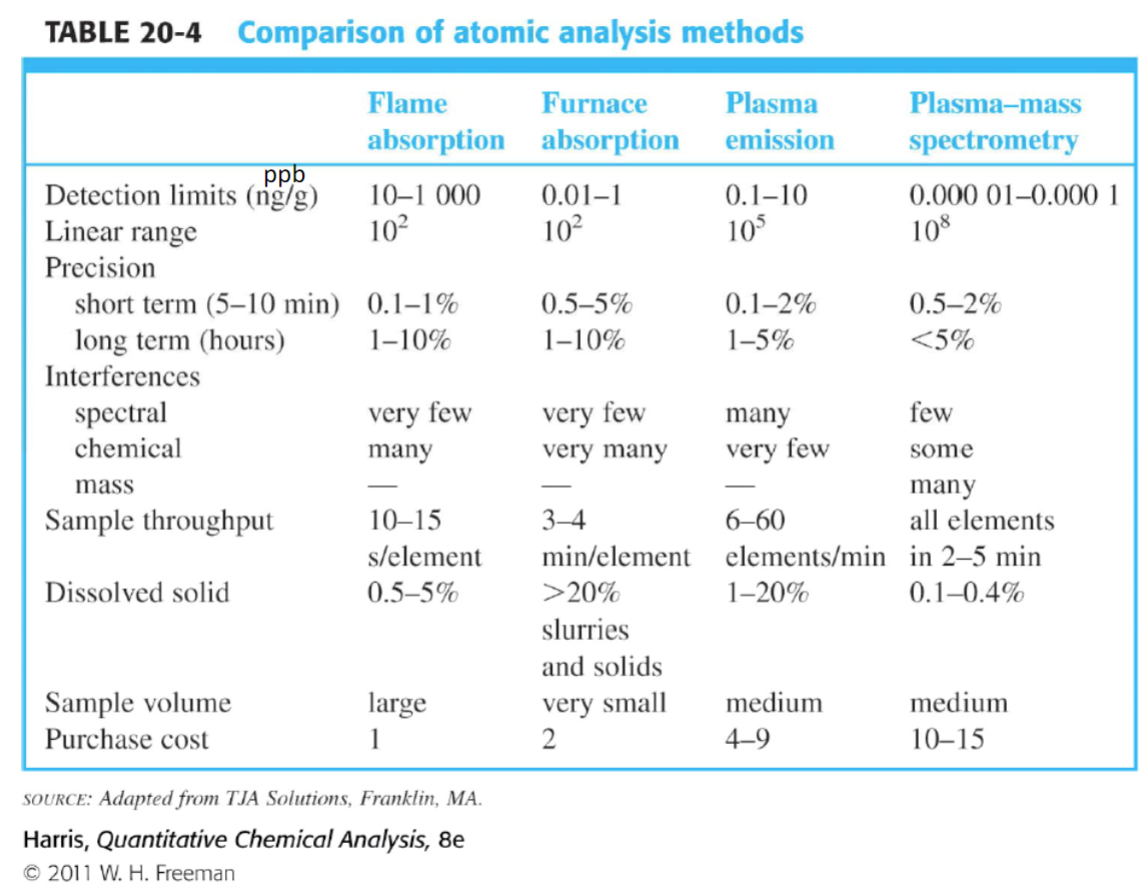
comparison of atomic analysis methods
4 analytical interferences
spectral
ionization
chemical
matrix
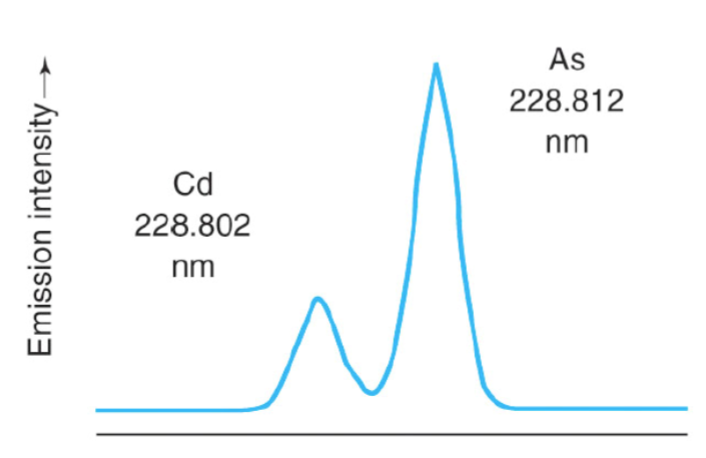
spectral interference
= the overlap of analyte signal with signals due to other elements in the sample or signals due to the flame or furnace
unwanted signals overlapping analyte signal
solutions:
choose another wavelength for analysis
use high resolution spectrometers that are able to resolve closely spaced spectral lines
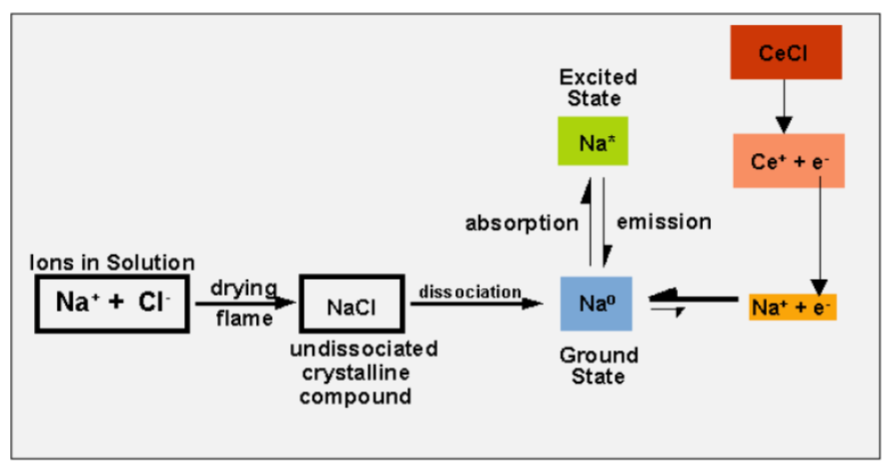
ionization interference
= desired signal is decreased due to ionization of analyte
ionization suppressants
add an excess of an ionization suppressant to standards and samples
there must be another Group I element which is more readily ionized, which will produce high concentrations of electrons that’ll suppress the ionization of your analyte
chemical interference
= caused by interaction with sample component that decreases atomization of analyte
analyte metal forms a bond with an anion which won’t dissociate in the flame
so the ground state is never reached and absorbance doesn’t occur
usually the Group II elements (like Ca2+ and Mg2+) with phosphate
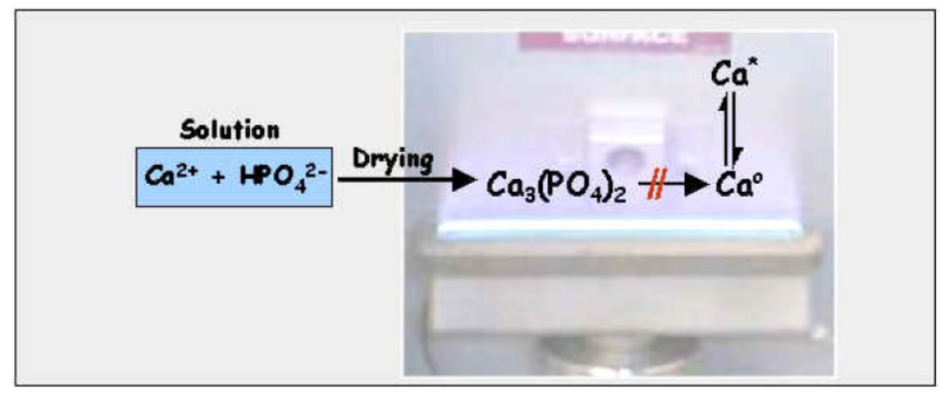
releasing agents
add a releasing agent to standards and samples
it’ll bind the phosphate more strongly than Group II’s
fuel rich flames can reduce certain oxidized analyte species that will be difficult to atomize
higher flame temperatures eliminate many kinds of chemical interferences
matrix interference
= solvent effects; progressive enhancement or decrease in signal based on the nature of the solvent
use method of standard addition
extrapolate backwards to the analyte concentration to automatically match matrix