Experimental Techniques
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
How to measure Mass?
Beam balance
Electronic balance
How to measure time?
Analogue stopwatch
Digital stopwatch
How to measure temperature?
Mercury/Alcohol thermometer
Digital thermometer
Data logger
How to measure volume?
Measuring cylinder
Burette - Very accurate to 0.05cm3
Pipette - Fixed volumes of liquid (typically 20.0cm3 or 25.0cm3
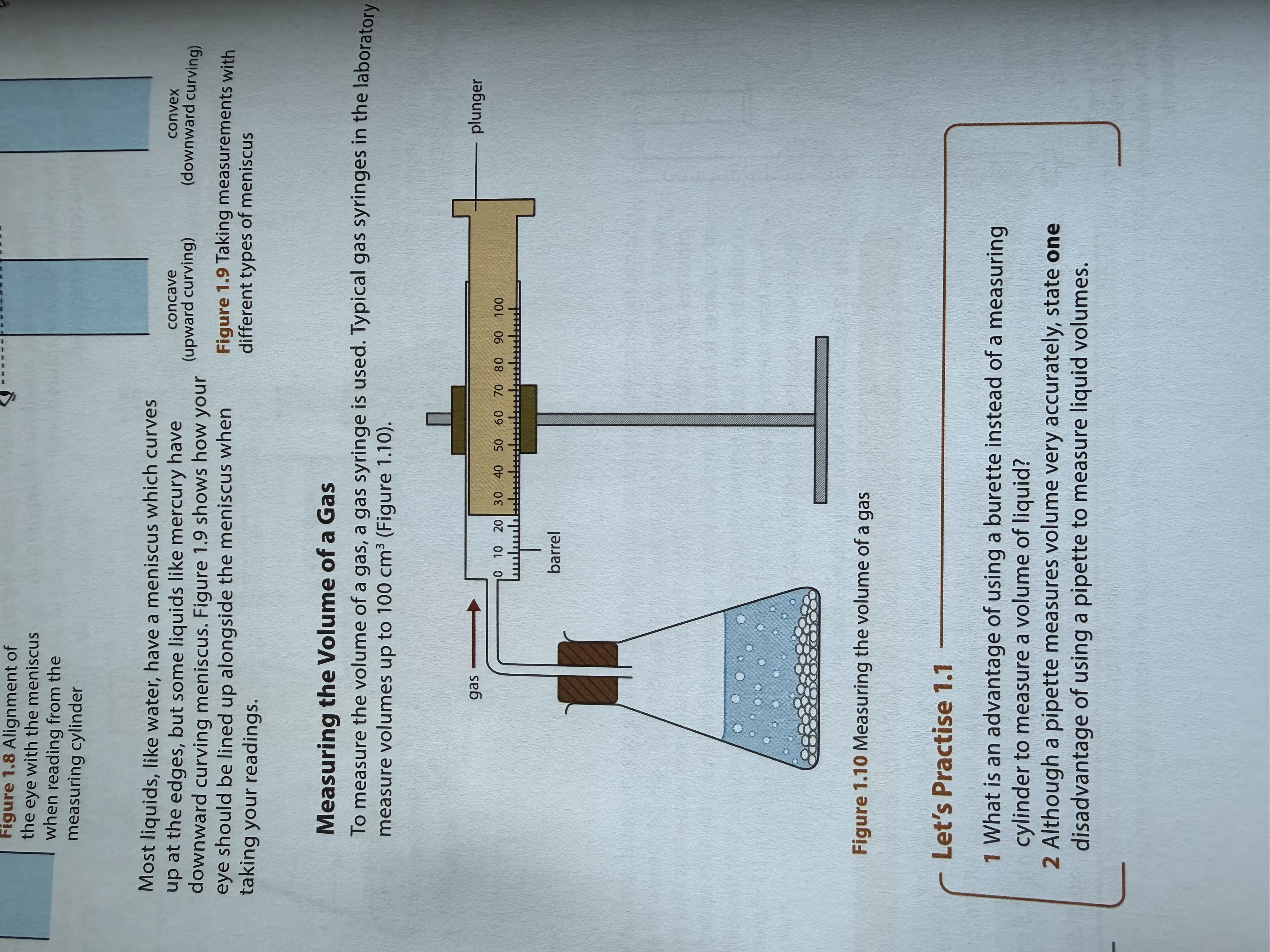
How to measure gas?
Gas syringe
Plunger moves outwards when gas is released
Measure reading on barrel
Max volume of 100cm3
When to use Displacement of Water to collect gas?
When collecting gases insoluble of water
When to use Downward delivery of gas?
When collecting gases denser than air
When to use Upward delivery of gas?
When collecting gas less dense than air
Gases that are not or only slightly soluble
Hydrogen
Oxygen
Carbon dioxide
Gases that are very soluble
Chlorine
Hydrogen
Sulfur dioxide
Ammonia
When to dry gas with Concentrated sulfuric acid?
Most gases can be used, except gases that react with acids (alkalis, ammonia)
When to dry gas with Calcium Oxide (Quicklime)?
Basic, alkaline gases and ammonia. Not suitable for gases that react with a base (acidic gases). Must be heated before use to make sure it is dry.
When to dry gas with Fused calcium chloride?
Most gases can be used, except acidic and basic gases, as well as ammonia.