QCE Chem Flashcards + Questions 2026 syllabus
1/183
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
184 Terms
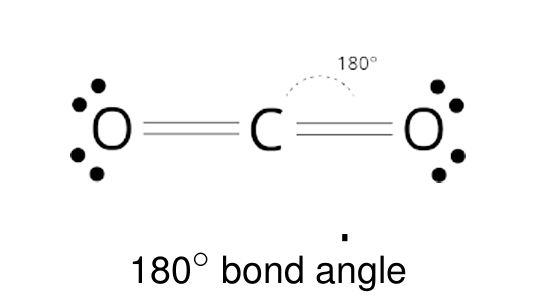
Linear shape
Bent shape
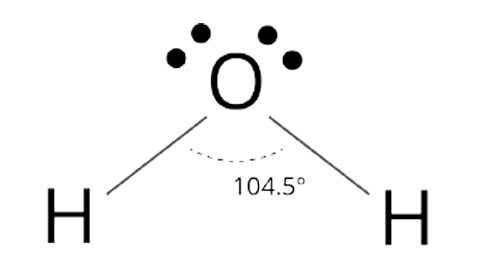
104 degree bond angle
Trigonal planar shape
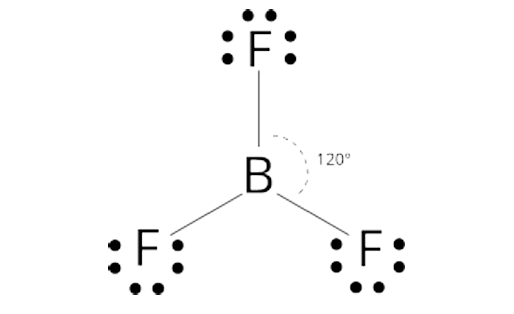
120 degree bond angle
Tetrahedral shape
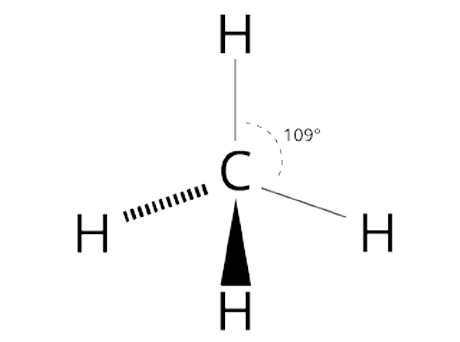
109 degree bond angle
Trigonal pyramidal shape
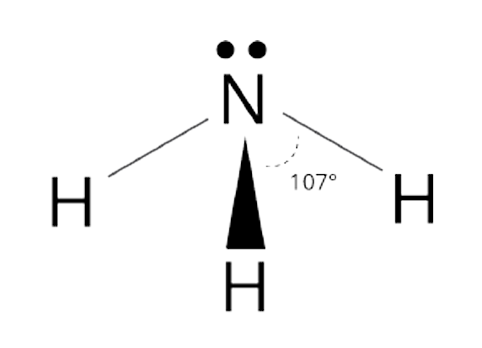
107 degree bond angle
Emission spectrum
Show white lines for emitted light
Absorption spectrum
Rainbow of absorbed light
Electronegativity difference of polar bonds
0.5-1.7 electronegativity difference
Electronegativity difference of non-polar bonds
<0.5 electronegativity difference
2 outer atoms, 1 inner atom
Linear
3 outer atoms, 1 inner atom
Trigonal Planar
4 outer atoms, 1 inner atom
Tetrahedral
3 outer atoms, 1 inner atom, 1 lone pair
Trigonal Pyramidal
2 outer atoms, 1 inner atom with lone pair
Bent
Metals which replace hydrogen in acid and water
Magnesium to potassium
Metals which replace hydrogen in acid only
Lead to aluminium
Metals which don’t replace hydrogen
Platinum to antimony
What is the oxidation number of hydrogen in metal hydrides?
-1
In compounds with nonmetals, what is the oxidation number of hydrogen?
+1
How to balance half-equations
Write the skeleton redox half equations
Balance all elements but oxygen and hydrogen
Balance oxygen with H2O, then balance hydrogen with H+
Balance charges with electrons
Multiply each half-equation by a number for an equal amount of electrons
Parts of a galvanic cell
Anode, cathode, electrolyte, external circuit, salt bridge
Electron flow in galvanic cell
Negative anode to positive cathode
Purpose of salt bridge in galvanic cells
Prevents buildup of charges, keeping the electrodes balanced
Parts of an electrolytic cell
Negative cathode, positive anode, porous barrier, electrolyte, voltage
Redox reaction
Transfer of electrons between chemical species
Low on SEP table
Strong oxidising agent, likely to reduce
High on SEP table
Strong reducing agent, likely to oxidise
Why electrode polarity changes in electrolytic cells
Reduction is always at the cathode
Reduction is electron gain, must be at the half-cell gaining electrons
Therefore the cathode always gains electrons
in galvanic cells, electrons spontaneously flow to positive electrode, so cathode is positive in galvanic cells
in electrolytic cells, electrons artificially flow to negative electrode, so cathode is negative in electrolytic cells
Alkanes
No functional group
Alkenes
Double carbon bond, no true functional group
Alkynes
Triple carbon bond, no true functional group
Alcohols
Hydroxyl OH
Aldehydes
Terminal carbonyl (double-bonded oxygen)
Ketones
Non-terminal carbonyl (double-bonded oxygen)
Carboxylic acids
terminal carboxyl group (carbonyl + hydroxyl)
-oic acid
Haloalkanes
Alkane with halogen in place of hydrogen
Rules of branched molecules
longest continuous chain is parent chain
Name: (position of branches)(name of branches)(parent chain)
order of carbons starts from the end the functional group is closest to. double/triple bonds are functional groups, branches are not
Nomenclature rule
hyphenate between letters and numbers, use comma between numbers
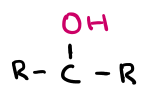
OH
hydroxyl
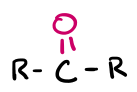
Double-bonded O
carbonyl
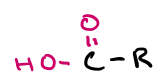
carboxyl group
Ester
Ester functional group (double-bonded O, O in middle)
Product of alcohol and carboxylic acid
Alcohol-yl acid-ate
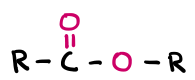
Ester
Amines
Amino functional group (NH2)

Amino group
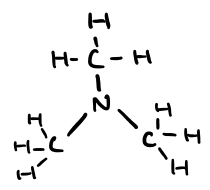
Name this molecule
N,N-dimethylmethanamine (amine)
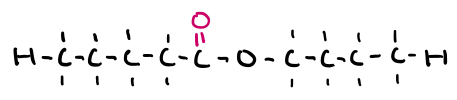
Name this molecule
butyl pentanoate (ester)
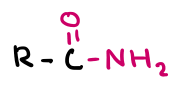
Amide group
Amide
Amide functional group (carbonyl + amino)
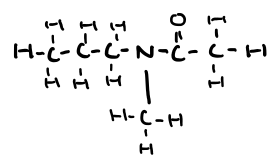
Name this molecule
N-methyl-N-propylethanamide
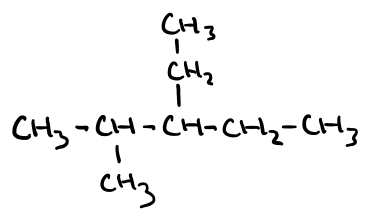
Name this molecule
3-ethyl-2-methylpentane
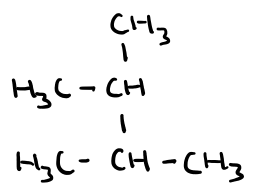
Name this molecule
2,3-dimethylbutane
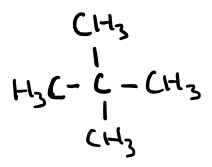
Name this molecule
2,2-dimethylpropane
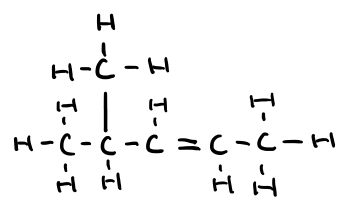
Name this molecule
4-methylpent-2-ene
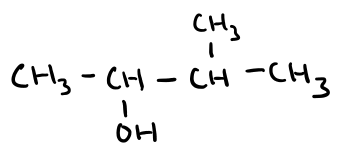
Name this molecule
3-methylbutan-2-ol
Primary Alcohols
Carbon with hydroxyl is bonded to one other carbon
Secondary Alcohols
Carbon with hydroxyl is bonded to two other carbons
Tertiary Alcohols
Carbon with hydroxyl is bonded to three other carbons
Primary Haloalkane
Carbon with halogen is bonded to one other carbon
Secondary Haloalkane
Carbon with halogen is bonded to two other carbons
Tertiary Haloalkane
Carbon with halogen is bonded to three other carbons
Functional group
Atom/group of atoms that impacts reactivity of a molecule
Class
Group of compounds with same functional group
Homologous series
Group of compounds with same functional group and general formula
Successive members differ by CH2
Isomers
Compounds with same molecular formula but different arrangement
Structural isomer
Atoms connected in a different order, different structural formula and name
Functional group isomers
Changed functional group e.g. ketone vs aldehyde
Positional isomers
Changed position of functional group e.g. propan-1-ol vs propal-2-ol
Chain length isomers
Changed main chain length, straight chain vs single or multiple branches off a parent chain
Stereoisomers
Atoms connected in the same order, different arrangement in 3D space
Geometric isomers
Occur when rotation in a molecule is restricted somewhere (e.g. due to double bonds). Cis or trans
Cis isomers
Particular atoms attached to carbons are on the same side
Trans isomers
Particular atoms attached to carbons are on the opposite side
Optical isomers
Same molecular/structural formulas, in 3D space are mirror images of each other.
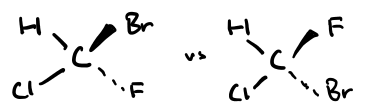
Chiral carbon / chiral centre
Carbon with 4 different groups attached
Chirality
Two objects that are mirror images of each other and cannot be superimposed
Enantiomers
Pair of chiral molecules. Identical physical properties (except optically), but interact differently with other chiral molecules
Intermolecular forces
Forces between molecules
Dispersion force
Weakest intermolecular force
affects all molecules
temporary due to movement of electrons
increases with molecular mass
Dipole-dipole force
stronger than dispersion force
affect polar molecules
positive and negative nodes of two molecules
fixed
Hydrogen bonding
a type of dipole-dipole force
strongest intermolecular force
between a positive H and a lone electron pair on a N, O, or F on another molecule
fixed
Intermolecular forces of alkanes and alkenes
non-polar, only have dispersion forces
Intermolecular forces of alcohols
polar
dispersion forces
due to hydroxyl, hydrogen bonding (as acceptor and donor)
Intermolecular forces of carboxylic acids
polar
dispersion forces
due to hydroxyl, hydrogen bonding (as acceptor and donor)
due to carbonyl, hydrogen bonding (donor only)
Volatility
qualitative measure of how easily a substance changes from liquid to gas
Correlation between boiling point and intermolecular force strength
As intermolecular strength increases, boiling point increases
Correlation between molecular mass and boiling point
As carbon chain length/molecular mass increases, boiling point increases
Correlation between surface area and boiling point
As surface area increases, boiling point increases
Branched molecules have weaker intermolecular forces
Correlation between functional group and boiling point
Different functional groups have different intermolecular forces
How to answer an intermolecular force question
structure of molecules
intermolecular forces present in each molecule
which molecular has stronger intermolecular forces and why
answer
Polarity and solubility
Polarity of each molecule due to functional groups affects solubility
more polar groups a molecule has, the more polar it is and the more soluble it is in polar solvent
Carbon chain length and solubility
as chain length increases, the non-polar portion of the molecule increases → solubility in polar solvent decreases
non-polar regions up to 4 carbons are negligible
Alkane + Oxygen
Oxidation (combustion): → Carbon dioxide + Water
Alkane + Halogen under UV light
Substitution: → Haloalkane + Hydrogen halide
Haloalkane + Halogen under UV light
Substitution: → Dihaloalkane + Hydrogen Halide
Haloalkane + Ammonia with heat and ethanol
Substitution: → Amine + hydrogen halide
Haloalkane + dilute sodium hydroxide
Substitution: → Alcohol + Sodium halide
Haloalkane + concentrated sodium hydroxide under heat
Elimination: → Alkene + Sodium halide + Water
Alkene + Halogen
Addition (halogenation): → Dihaloalkane
Alkene + Hydrogen Halide
Addition (hydrohalogenation): → Haloalkane