Practicalities and Adverse Consequences of antimicrobials
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
29 Terms
What is antimicrobial stewardship?
an organisational or healthcare system-wide approach to promoting and monitoring judicious use of antimicrobials to preserve their future effectiveness
Purpose?
-Monitor & evaluate antimicrobial prescribing
-Produce & review guidance
-Provide regular feedback on prescribing habits and patient safety incidents
-Provide Education and training on stewardship & resistance
-Integrate audit (help track antibiotic prescribing patterns, ensuring compliance with AMS guidelines) into existing QI (Quality improvement) programmes
Who carries out antimicrobial stewardship?
The whole MDT (pharmacist, physician, infection control etc.)
Note:
They do have a separate committee (governance)
What are the benefits of antimicrobial stewardship programmes ?
• Reduce inappropriate antimicrobial prescribing
• Reduce incidence of Healthcare-Associated Infections
• Increase cost-effective prescribing
• Reduce emergence of AMR
Who is part of the antimicrobial Stewardship Team and what is their role?
-Ward-/patient-focused team e.g. AMS pharmacist
Role:
-Review patients at ward-level to ensure they are receiving most appropriate care
-• Liaise with attending teams to discuss cases & educate on practice
-Take on referrals
-Use IT systems to identify caseload
What is "Start Smart, Then Focus" (SSTF) framework - PURPOSE AND PRINCIPLES
-How we practice AMS (antimicrobrial stewardship) in hospitals
-Used in secondary care only
-Designed to reduce the risk of antimicrobial resistance & adverse consequences of antimicrobial use
START SMART SECTION
Principles:
Assess -
-For clear evidence of infection
• undertake risk assessment to guide selection of proportionate treatment
Investigate-
-Appropriate specimens
-implement source control as soon as practical
Prescribe-
-prompt treatment based on local guidelines, avoiding indiscriminate use of broad-spectrum antimicrobials
-take detailed drug allergy history & manage appropriately
Document-
-evidence of infection, working diagnosis, drug name, dose, formulation, route & duration on prescription chart & in note
Record & communicate a clear clinical management plan
FOCUS PART (AFTER ABOUT 48HRS)
Review & revise-
-Clinical diagnosis & continuing need for antimicrobials by 48-72h
-Document clear plan of action (antimicrobial review outcomes)
What are the 5 antimicrobial review outcomes?
-Cease
-Amend (to maybe narrower spectrum antimicrobial)
-Refer (OPAT or specialist)
-Extend
-Switch (drug or formulation)
of treatment option
SSTF Surgical prophylaxis flow chart
How do we carry our AMS in primary care?
-TARGET toolkit for GP's (focuses son UTI's and Respiratory TI's)
• Also has leaflets, audits, quick reference tools • Learning tools & training tools • Resources for community pharmacies
How do we carry our AMS on a national level?
-National Action Plan
-Targets
-Right choice and duration was the focus previously but now focuses on whether to prescribe or not (correct diagnostic choice)
What is the most common drug allergy ?
Penicillin allergy
What does cross reactivity in antibacterial classes mean?
If an individual is allergic to penicillins theyre likely to be allergic to e.g. cephlasporins also
Which class of antibacterials has cross-reactivity with penicillins?
Cephalosporins except ampicillin or amoxicillin
-Cross-reactivity mechanisms now thought to be more related to shared sidechains, rather than beta-lactam ring itself
How does penicillin cross-reactivity occur?
- covalently bind to plasma protein making happen-carrier complexes
- beta-lactam ring binds to Lys residue
- major antigenic determinant forms binding to polylysine matrix
- minor determinant from covalent binding to carboxyl and thiol
What is the mechanism of IgE-mediated (Type 1 -immediate hypersensitivity) allergic reactions?
- Dendritic cells bind to penicillin-bound proteins and present them to T helper cells
- In presence of IL-4, they become TH-2 and induce differentiation of B cells in plasma
- Secrete a penicillin-specific IgE
- bind to mast cells and basophils
- re-exposure of penicillin causes mast cell degranulation and release trienes, histamine, PGs and leukotrienes
(Image shows 3 main mechanism - only fussed about whats in my notes here really)
What are the other mechanisms of allergic reactions?
1) delayed = non-covalent binding which alters HLA peptide
2) delayed T-cell mediated reactions e.g., DRESS, SJS, TEN, AGEP
Clinical implications of a penicillin allergy label
-Receive more vancomycin, quinolones & clindamycin
-Have higher mortality for MSSA bacteraemia (BSI) due to worse choice as we are avoiding giving the a beta lactam
-Have increased rates of MRSA, VRE & Clostridioides difficile infections
-Have longer lengths of hospital stay
-Have increased readmission rates
-Have higher rates of surgical site infections Image from: NEJMterm-5
ALL MAJORLY BASED ON THE ALTERNATIVES ARENT AS GOOD AS BETA LACTAMS (PENICILLIN)
What are patients with a penicillin allergy given instead and what are they susceptible to?
Vancomycin
Quinolones
Clindamycin
- higher mortality
- higher failure rate
- increased MRSA/CD
- longer hospital stays and readmission
- higher surgical site infections
How is penicillin allergy diagnosed?
For IgE mediated = skin testing with increasing amounts over time then observe patient for an hour
For delayed allergy = patch, prick test or intradermal
Low-risk give 250 mg amoxicillin
Pharmacists can do this
What is C.difficile?
Gram-positive, spore-forming, anaerobic, bacillus
these toxin producing strains cause diarrhoea
- Toxin A = enterotoxin
- Toxin B = cytotoxin
How does C.difficile manifest?
- Faecal-oral spores
- Pass through GIT and contact bile acids
- Stimulates germination
- Produce toxins
What is the pathogenesis of C.difficile?
- C.difficile lives in gut microflora disruption by broad-spectrum antibiotic use
- overgrowth = symptom causing/fatal
- diarrhoea = less bacterial diversity
- disruption can last weeks after finishing antibiotics
- absence of antibodies can lead to infection
How can an individual be infected with C. Difficile ?
Infection occurs by ingestion of spores via faecal-oral route which can
-These spores are 'hardy' (can survive in harsh conditions)
-Spores pass through GI tract & come into contact with bile acids & other substances
->stimulates germination of the spores =production of toxins, depending on the surrounding microflora (If the normal gut bacteria (microflora) are disrupted (e.g., due to antibiotics), C. difficile overgrows) = inflammation, diarrhea, and damage to the colon
name the risk factors for CDI
- prolonged use of broad-spectrum antibiotics as they can kill other gut flora
- acid suppressors e.g., PPI/H2
- 65+
- GID (as a co-morbidity)
What are the symptoms of CDI?
- mucosal irritation
- diarrhoea (can have constipation also - sign of an ileus develop = may need surgery)
- severe colitis
- dehydration - needs to be managed
- high WBC, temp, sepsis
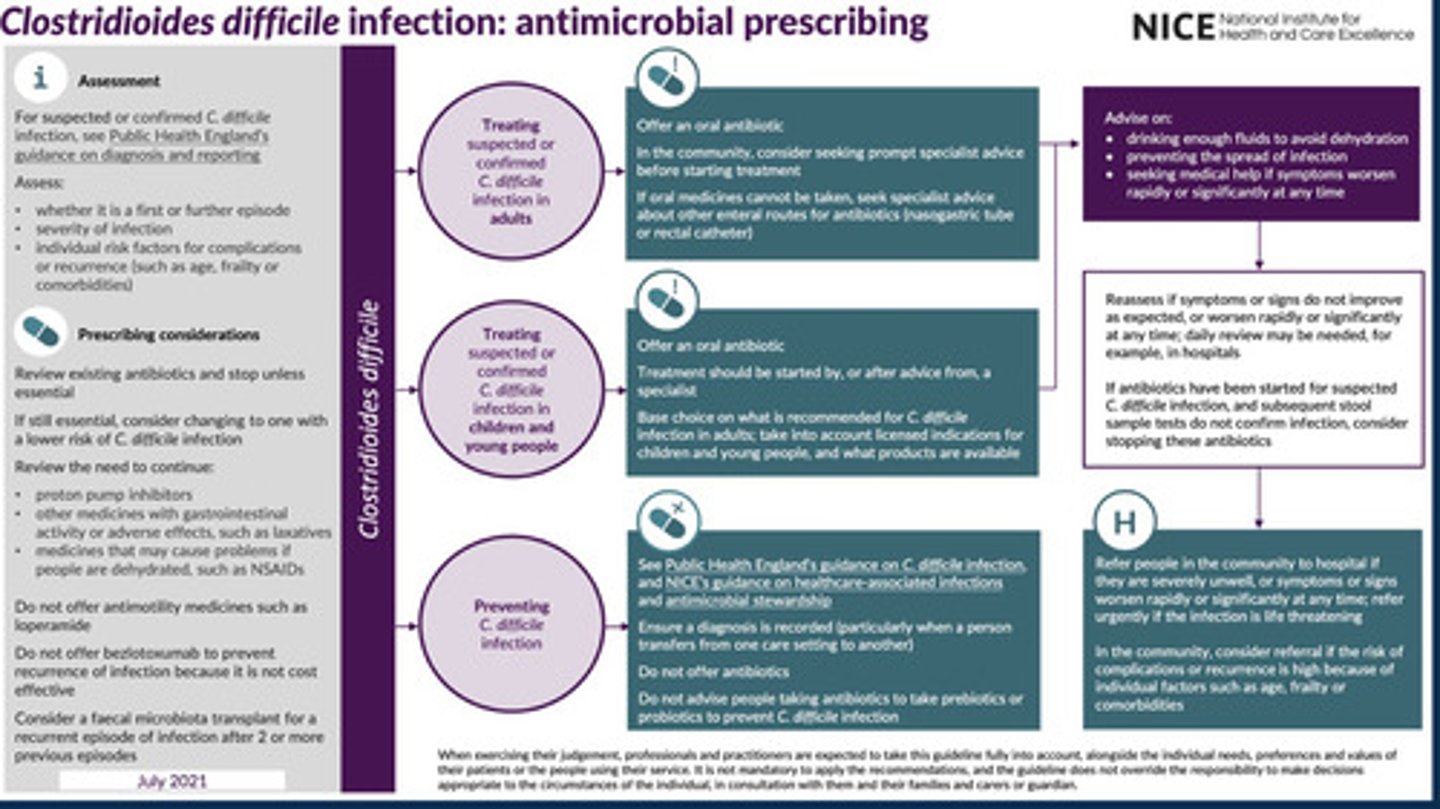
Management (Treatment) of CDI in Adults and children guidlines
-Get specialist advice before starting treatment
What is CDI shown to be resistant to?
- Quinolones
- Cephalosporins
- Clindamycin
What is the infection control steps for CDI?
- isolate patient
- alcohol-based product do not work
- mechanically remove spores
- AVOID 2nd and 3rd generation cephalosporins
- minimise quinolones and carbapenems
What is the treatment for the first episode of CDI?
-
- Mild = oral Vancomycin 125mg orally QDS for 10 days 200mg orally bds for 10 days
-2nd line: Fidaxomicin
- Severe = vancomycin 500mg (increased dose) qds 10-14 days or Fidaxomicin 200mg orally bds for 10 days
- AVOID loperamide and opioids
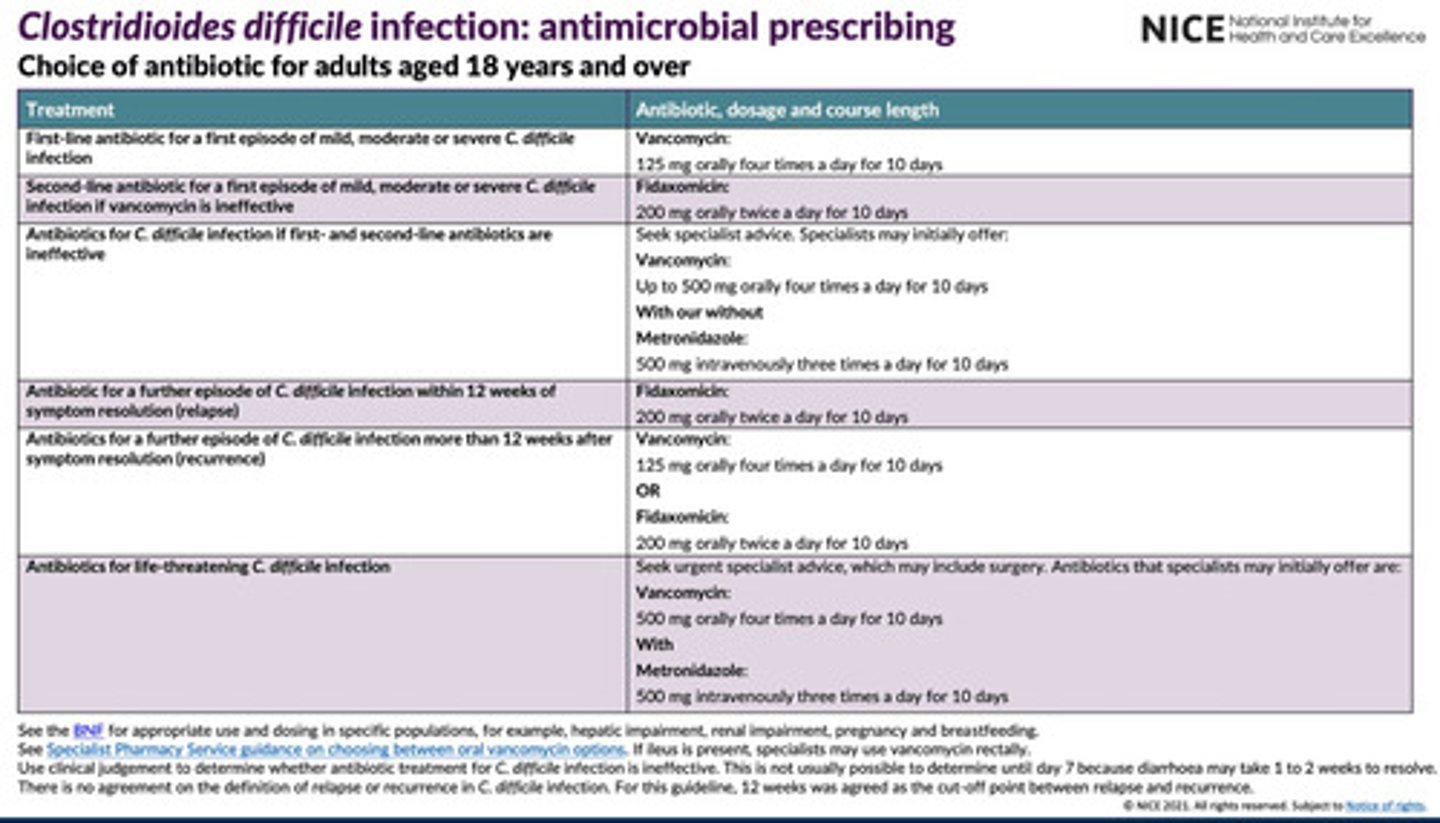
What is the treatment for recurrent CDI?
- recurrent diarrhoea within 30 days 1st EP
- Fidaxomicin bd or
- Vancomycin qds
10-14 days