Chapter 15 Dienes And Aromaticity
1/110
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
111 Terms
15.1 Structure And Stability Of Dienes
What does conjugated dienes cause?
Special stability
What is the main reason for the greater stability of conjugated dienes?
Conjugation
Overlap of 2p orbitals across the carbon-carbon single bond connecting the two alkene units. Not only does pi bonds occur within each of the alkene units, but between them as well
What is delocalization energy?
The energetic advantage of conjugation
What happens as the fraction of s character in the component orbitals increases?
The length of the bond decreases

What is s-trans conformation?
Trans position
The s prefix emphasizes that this refers to rotation about a single bond
Sometimes called the anti-conformation
What happens to 2p orbitals in the s-trans conformation?
The 2p orbitals of all carbons are coplanar and can overlap

What happens to 2p orbitals of one double bond in the gauche conformation?
The 2p orbitals of one double bond are twisted 38o relative to those of the other, at the cost of some orbital overlap

What happens to 2p orbitals in the s-cis conformation?
The 2p orbitals are coplanar, but van der Waals repulsions between two of the hydrogens destabilize this conformation; in the gauche conformation, the offending hydrogens are further apart

What is the hybridization of the central carbon of allenes?
Because the central carbon of allene is bound to two groups, the carbon skeleton of this molecule is linear. A carbon atom with 180o bond angles is sp-hybridized. The central carbon of allene, like the carbons in an alkyne triple bond, is sp-hybridized. The two remaining carbons of the cumulated diene are sp2-hybridized and have trigonal planar geometry

Can allenes be chiral?
Because of their geometries, some allenes are chiral even though they do not contain an asymmetric carbon atom. The following is an example of twist chirality

Do allenes have greater heats of formation than other types of isomeric dienes?
Yes
What is the least stable arrangement of two double bonds?
Cumulated arrangement
What is a common reaction of allenes?
Isomerization of alkynes
15.2 Ultraviolet-Visible Spectroscopy And Fluorescence
What is ultraviolet-visible spectroscopy?
It can be used to analyze and identify organic compounds containing conjugated double and triple bonds
The absorption of radiation in the ultraviolet or visible region of the spectrum is recorded as a function of wavelength
The part of the ultraviolet spectrum of greatest interest to organic chemists is the near ultraviolet
What is the graph for the UV-vis spectrum?
The graph of radiation absorption by the substance versus the wavelength of the radiation
What is on the horizontal and vertical axis of the UV spectrum?
Horizontal axis: the wavelength lambda of the reaction. The units is nanometer
Vertical axis: the absorbance, a measure of the amount of radiant energy absorbed. Also known as the optical density

What is the formula for absorbance?
Suppose the radiation entering a sample has intensity I0, and the light emerging from the sample has intensity I. The absorbance A is defined as the logarithm of the ratio I0/I:
A = log(I0/I)
What happens as more radiant energy is absorbed (I decreases)?
The ratio I0/I increases, as does the absorbance
What does the absorbance at a given wavelength depend on?
The number of molecules in the light path
What is Beer’s Law?
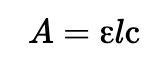
ε: molar extinction coefficient (molar absorptivity). Units are L mol-1 cm-1, or M-1 cm-1. Each absorption in a given spectrum has a unique extinction coefficient that depends on wavelength, solvent, and temperature. he larger the value, the greater is the light absorption at a given concentration c and path length l
Extinction coefficients of 104-105 M-1 cm-1 are common for what molecules?
Molecules with conjugated pi-electron systems
How can strong absorptions be obtained?
From very dilute solutions, solutions with concentrations on the order of 10-4 to 10-6 M with a typical path length of 1 cm
What absorb ultraviolet and/or visible radiation?
Pi electrons and, in some cases, by the nonbonding pairs in organic compounds
Are absorptions by compounds containing only single bonds and nonbonding electron pairs weak or strong
Generally quite weak
When does intense absorption of UV and visible radiation occur?
When a compound contains pi electrons
What is UV-vis spectroscopy useful for diagnosing?
Conjugated double or triple bonds

What is chromophore?
The structural feature of a molecule responsible for its UV-vis absorption
Which spectroscopy has limited utility in structure determination?
UV-vis spectroscopy
When UV-vis spectroscopy is used to determine chemical structure, what is the most important aspect of a spectrum?
Lambda max values
What is the structural feature of a compound that is most important in determining the lambda max?
The number of consecutive conjugated double (or triple) bonds. The more consecutive conjugated multiple bonds there are, the higher the wavelength if the absorption
What happens in a conjugated diene?
The UV-vis absorption at highest wavelength results in promotion of a pi electron from the bonding MO of highest energy, called the HOMO (highest occupied molecular orbital) to the anti-bonding MO of lowest energy, called the LUMO (lowest unoccupied molecular orbital)
The HOMO-LUMO energy gap, the energy difference between these two MOs, determines the wavelength of the absoprtion
What happens to the HOMO-LUMO gap as it becomes smaller?
The number of conjugated double bonds increases → the energy of the radiation required for absorption becomes smaller and the wavelength greater, as the number of double bonds increases
What increases as the number of conjugated double bonds increase?
Lambda max, as well as the extinction coefficient, increases. Each additional conjugated double bond increases lambda max by 30 to 50 nm
What do molecules that contain many conjugated double bonds have?
Several absorption peaks
What happens if a compound has enough double bonds in conjugation?
One or more of its lambda max values will be large enough to fall within the visible region of the electromagnetic spectrum, and the compound will be colored
What is another thing that determines the lambda max of an organic compound?
The conformation of a diene unit about its central single bond, whether the diene is in an s-cis or an s-trans conformation
What does an acyclic diene assume?
The lower energy s-trans conformation
What do dienes locked into s-cis conformations have?
Higher values of lambda max and lower extinction coefficients than comparably substituted s-trans compounds
What is a third variable that affects lambda max?
The presence of substituent groups on the double bond
For example, each alkyl group (regardless of size) on a conjugated double bond adds about 5 nm to the lambda max of a conjugated alkene. The two methyl groups of 2,3-dimethyl-1,3-butadiene therefore add (2x5)=10 nm to the lambda max of 1,3-butadiene, which is 217 nm. The predicted lambda max is (217+10)=227 nm; the observed value is 226 nm

When is lambda max greater?
Greater for compounds containing more conjugated double bonds
What affects lambda max?
Substituents, conformation, and other structural characteristics of the conjugated pi-electron system
15.3 The Diels-Alder Reaction
What is the Diels-Alder reaction?
Reaction between a conjugated diene and an alkene
An example of a cycloaddition reaction, an addition reaction that results in the formation of a ring
An important method for making rings
What is the Diels-Alder reaction an example of?
A 1,4-addition or conjugate addition
Occurs across the outer carbons (carbons 1 and 4) of the diene
What is a characteristic type of reaction of conjugated dienes?
Conjugate addition
What does conjugate addition result in the formation of?
A double bond between carbons 2 and 3. The numbers indicate the relative locations of the carbons involved in the addition; they have nothing to do with the numbering of the diene used in its substitutive nomenclature

In Diels-Alder reactions, what is the diene and dienophile?
Diene: the conjugated diene reactant
Dienophile: the alkene with which it reacts
How many steps are in the Diels-Alder reaction?
Single step involving a cyclic flow of electrons
The curved arrows for this mechanism can be drawn in either a clockwise or a counterclockwise direction

What is a pericyclic reaction?
A concerted reaction that involves a cyclic flow of electrons
What do some of the dienophiles that react most rapidly in the Diels-Alder reaction bear?
Substituent groups such as esters, nitriles, or certain other unsaturated, electronegative groups
Are these substituents needed for Diels-Alder reactions?
No because the reactions of many other alkenes can be promoted by heat or pressure
Can alkynes serve as dienophiles in Diels-Alder reactions?
Yes

What happens if the diene is cyclic instead of a simple diene in the Diels-Alder reaction?
A second ring is formed. This can be used to prepare certain bicyclic compounds

Can a Diels-Alder reaction occur between two alkenes or between two dienes?
No
Can “locked” s-trans conformation dienes react in Diels-Alder reactions?
No because the s-trans single bonds of the diene would become a trans double bond in the Diels-Alder product. It would contain a trans double bond in a membered ring. The product is a bicyclic compound containing a bridgehead double bond

What dienes are the most reactive?
Cyclic dienes locked into s-cis conformations are considerably more reactive than their corresponding non-cyclic dienes
Non-cyclic dienes must overcome an energy barrier to adopt the s-cis conformation, while cyclic dienes locked in the s-cis conformation avoid this barrier and react more rapidly

Why are the Diels–Alder reactions of (Z)-1,3-pentadiene slower than those of (E)-1,3-pentadiene?
The s-cis conformation of (Z)-1,3-pentadiene is destabilized by van der Waals repulsion between the methyl group and a hydrogen, which increases the energy of the transition state. In contrast, (E)-1,3-pentadiene experiences less severe repulsion in its s-cis conformation, making its reactions faster. The s-cis conformation is better than s-trans, but how stable the s-cis conformation is depends on the structure of the diene!


In the Diels-Alder reaction, if we use a dienophile that is a cis alkene, what happens to the product?
Groups that are cis in the alkene starting material are also cis in the product

In the Diels-Alder reaction, if we use a dienophile that is a trans alkene, what happens to the product?
Groups that are trans in the alkene starting material are also trans in the product

How is syn addition to the diene reveled?
If the terminal carbons of the diene unit are stereocenters. We first draw the diene in its s-cis conformation and then classify the groups at the terminal carbons as inner substituents (Ri) or outer substituents (Ro)

What does a syn addition require in the Diels-Alder product?
The two inner substituents always have a cis relationship, the two outer substituents are also cis, and an inner subsistent on one carbon is always trans to an outer subsistent on the other



For a given diene and dienophile, are two diastereomeric syn-addition products possible?
Yes. This issue arises when both the terminal carbons of the diene and the carbons of the dienophile are stereocenters

What are two possibilities which a cis alkene reacts with a diene?

What is endo and exo stereochemistry?
Endo stereochemistry: the product in which the alkene substituents R are cis to the outer diene substituents Ro
Endo stereochemistry: the product in which the alkene substituents R are trans to the outer diene substituents Ro
Because they are diastereomers, both products are typically formed in different amounts

15.4 Addition Of Hydrogen Halides To Conjugated Dienes
What happens when conjugated dienes react with hydrogen halides?
It gives two types of addition product. (The numbers are for reference in describing the addition; they are not associated with nomenclature)

What is 1,2-Addition?
Addition occurs at adjacent carbons
The major product is a 1,2-addition product
What is 1,4-Addition (conjugate addition)?
Addition occurs to carbons that have 1,4-relationship
The minor product results from 1,4-addition, or conjugate addition
What does protonation of the diene at either of the equivalent carbons labeled as 1 and 4 give?
A resonance-stabilized carbocation. The resonance structures for this carbocation show that the positive charge in this ion is not localized, but is instead shared by two different carbons

Why are two constitutional isomers formed?
Because the bromide ion can react at either of the electron-deficient carbons

What does protonation at carbon-2 of the diene give?
A different carbocation, which would react with bromide ion to give an alkyl halide that is different from those obtained experimentally

Which products derived from which carbocation are preferred?
The carbocation from the protonation of the diene at either of the equivalent carbons labeled as 1 and 4 is preferred because of resonance-stability so the products from this carbocation are observed
Resonance accounts for the greater stability of the carbocation intermediate that is formed

What can sometimes happen after a reaction?
Sometimes the less stable product is formed in larger amount
Ex: When a conjugated diene reacts with a hydrogen halide to give a mixture of 1,2- and 1,4-addition products, the 1,2-addition product predominates at low temperature

When the less stable product of a reaction is the major product, what two things must be true?
The less stable product must form more rapidly than the other products. The reaction that forms the less stable product is faster
The products must not come to equilibrium under the reaction conditions, because other wise the more stable compound would be present in larger amount. In the addition of HCl to conjugated dienes, the predominance of the less stable product shows that 1,2-addition, which gives the less stable product, is faster than 1,4-addition

What are does kinetically controlled and thermodynamically controlled refer to?
Kinetically controlled: when the products of a reaction do not come to equilibrium under the reaction conditions. The relative proportions of products are controlled solely by the relative rates at which they are formed
Thermodynamically controlled: if the products of a reaction come to equilibrium under the reaction conditions
What might a given kinetically controlled reaction give?
The same distribution products as would be obtained if the products were allowed to come to equilibrium
What is impossible for a thermodynamically controlled reaction?
To give a product distribution other than the equilibrium distribution
What can we tell when we obtain a product distribution that is clearly different from the obtained at equilibrium?
We know immediately that the reaction must be kinetically controlled
In hydrogen halide addition to a conjugated diene, what is the first and rate-limiting step in the formation of both 1,2 and 1,4-addition products?
Protonation of the double bond
What must the product distribution be determined by?
The relative rates of the product-determining steps: the nucleophilic reaction of the halide ion at one or the other of the electron-deficient carbons of the allylic carbocation intermediate

Why is the 1,2-addition product formed more rapidly?
The reaction is typically carried out in solvents in which the HCl is not dissociated
The diene reactions with undissociated HCl
The carbocation and its chloride counter-ion, when first formed, exist as an ion pair
The chloride ion and the carbocation are closely associated
The chloride ion simply finds itself closer to the positively charged carbon adjacent to the site of protonation than to the other carbon
Addition is completed at the nearer site of positive charge, giving the 1,2-addition product

What determines the relative amounts of products in a kinetically controlled reaction?
The relative free energies of the transition states for each of the product-determining steps and not by the relative free energies of the products
15.5 Diene Polymers
What can 1,3-butadiene be polymerized to give?
Poly(butadiene). This polymer can result either from 1,4-addition of butadiene molecules to each other or from 1,2-addition. The 1,4-addition polymers can contain cis or trans double bonds

What does the most useful butadiene polymers contain
Mostly cis double bonds. This type of polymer is produced by the action of transition-metal organometallic catalysts. The cis double bond is a consequence of the way that the pi bonds of the diene interact with the catalysts. Different types of catalysts can be used to give the different types of diene polymers
What can 1,3-butadiene be polymerized together with?
Styrene, usually in about a 3:1 ratio, to give another type of synthetic rubber called styrene-butadiene rubber (SBR)

What can the butadiene units in SBR be polymerized in?
Either a 1,4- or 1,2-addition, and the 1,4-addition polymer can contain cis or trans double-bond stereochemistry
What is SBR an example of?
A copolymer, a polymer produced by the simultaneous polymerization of two or more monomers
What do crosslinks do?
They increase the rigidity and strength of the polymer at the cost of some flexibility

15.6 The Connection Between Resonance And Stability
What is an allylic carbocation?
A carbocation in which the positively charged, electron-deficient carbon is adjacent to a double bond

Are branched nonallylic alkyl carbocations more stable than allylic carbocations?
Allylic carbocations are more stable than comparably branched nonallylic alkyl carbocations. An allylic carbocation is about as stable as a nonallylic alkyl carbocation with one additional alkyl branch

What is a nonbonding MO (NBMO)?
An MO that has the same energy as an isolated 2p orbital
What is the connection between molecular stability and resonance?
Resonance structures provide a symbolic way to show electron delocalization in the low-energy bonding MOS with lewis structures

What is the logic behind resonance stabilization?
Resonance structures symbolizes electron delocalization
Electron delocalization is stabilizing because it results in additional bonding associated with the formation of low-energy binding MOs
Resonance is a stabilizing effect
What is resonance energy?
Because resonance describes electron delocalization, delocalization energy, the energetic advantage of electron delocalization, is also referred to as resonance energy
Is there a positive charge on the central carbon of an allylic cation?
No
15.7 Introduction To Aromatic Compounds