Unit 5: Kinetics
1/43
Earn XP
Description and Tags
5.1 Reaction Rates, 5.2 Introduction to Rate Law, 5.3 Concentration Changes Over Time, 5.4 Elementary Reactions, 5.5 Collision Model, 5.6 Reaction Energy Profile, 5.7 Introduction to Reaction Mechanisms, 5.8 Reaction Mechanism and Rate Law, 5.9 Pre-Equilibrium Approximation, 5.10 Multistep Reaction Energy Profile, 5.11 Catalysis
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
kinetics
rate at which reactions occur and the factors that affect them
what happens to concentration of reactants and products as reaction progresses
[reactant] decreases, [product] increases
rate unit
usually M/s
rate with respect to reactant A
−ΔtΔ[A]
rate with respect to product B
ΔtΔ[B]
why is the reactant’s rate negative
it is being consumed, so the concentration at the end is lower than the beginning
what must occur in order for a reaction to occur (collision theory)
particles must collide with correct orientation and sufficient energy
how does increased SA (breaking up into pieces) affect reaction rate
increase frequency of collisions, increase rate
how does increased concentration of reactants affect reaction rate
more molecules, increase rate
how does increased pressure (in gas) affect reaction rate
forces particles closer together, increase rate
how does decreasing the volume (of gas) affect reaction rate
forces particles closer together, increase rate
how does increased temperature affect reaction rate
molecules move faster & have more kinetic energy, increase rate
how does using a catalyst affect reaction rate
lowers activation energy, increases rate
rate law
rate = k [reactant1]n[reactant2]m
what does k represent in rate law
rate constant
initial rate
reaction rate as close to t = 0 as possible
reaction order
how impactful reactant is on the rate of reaction
what happens if you double concentration of a 0 order reactant
rate stays the same
what happens if you double concentration of a 1st order reactant
rate doubles
what happens if you double concentration of a 2nd order reactant
rate quadruples
overall order of a reaction
add up the individual orders of the reactants
how to determine rate law experimentally
find trials where only one reactant changes concentration, then calculate its order based on how the rate of reaction changes
how to find units for rate constant
1/Mxs, where x = overall order - 1
what does 0 order graph look like
linear when y-axis is [A], slope is negative
what does 1st order graph look like
linear when y-axis is ln[A], slope is negative
what does 2nd order graph look like
linear when y-axis is 1/[A], slope is positive
what is half-life associated with
1st order reactions (change exponentially)
elementary reactions
series of simple reactions
reaction mechanism
sequence of steps in a reaction
rate-determining step in elementary reactions
slow step
when can we directly derive rate law from the equation
if the slow step is the first step, coefficient of reactants is their order
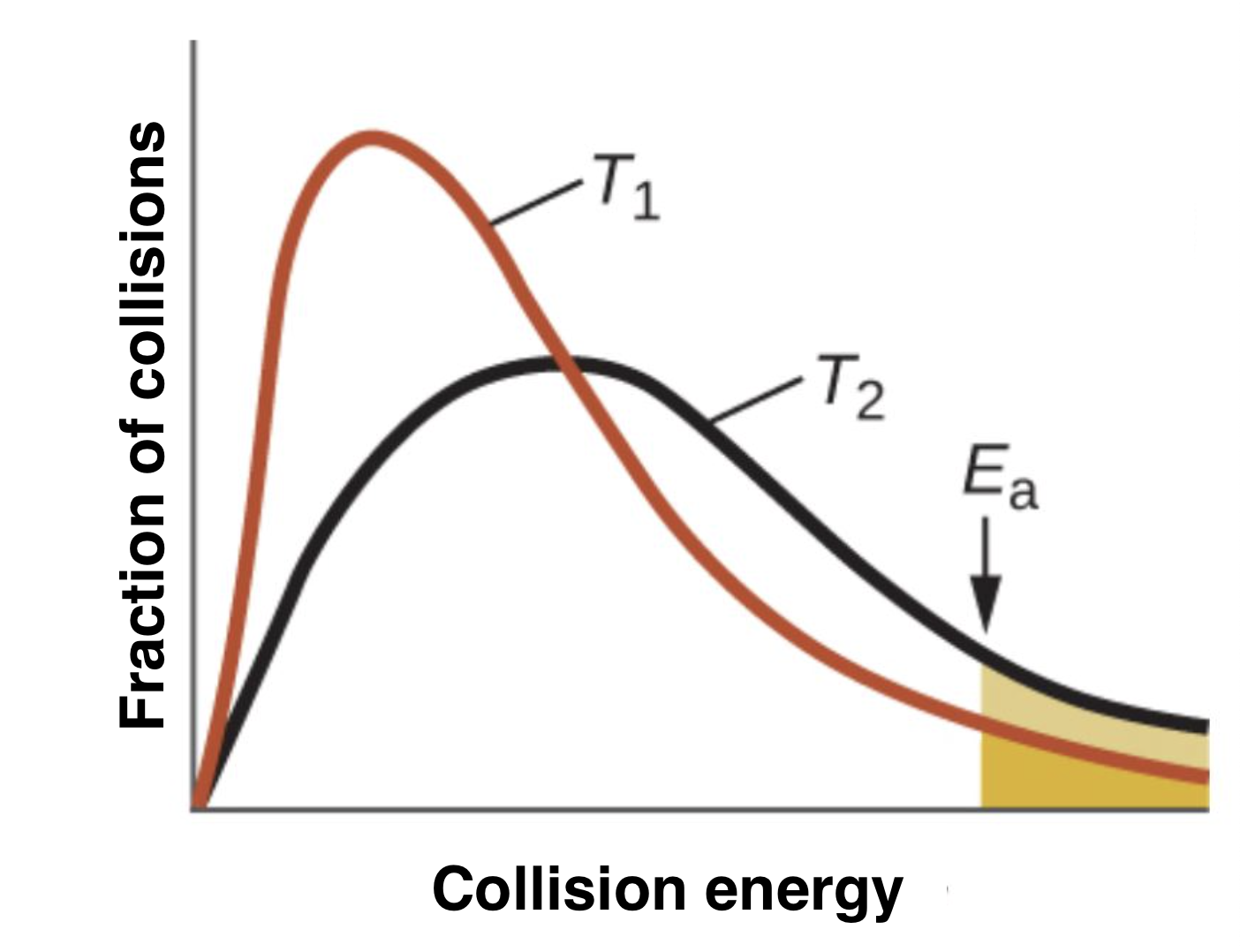
which temperature is greater?
T2 because there are more collisions/molecules at the activation energy
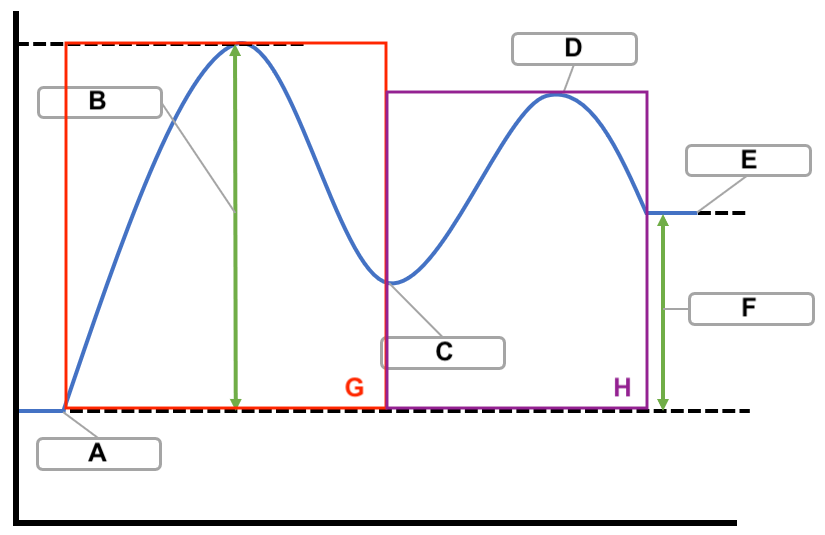
label
A - reactants
B - activation energy
C - intermediate
D - transition state/activated complex
E - products
F - ΔH
G - slow step
H - fast step
how to tell if reaction is exo or endothermic
positive ΔH is endothermic, negative ΔH is exothermic
relationship between temperature and rate constant
direct according to the Arrhenius equation
intermediate
produced during the reaction, then immediately used up; concentration stays relatively low; does not appear in overall equation
catalyst
speeds up reaction without chemically changing, present in beginning and and ending steps
pre-equilibrium approximation
used to determine rate law if the first step is fast and reversible and the second step is slow; use substitution
how do you deal with a -[product] when performing pre-equilibrium approximation
put it in the denominator of the rate law
how does catalyst speed up reaction
lowers activation energy, can provide alternate mechanism, can assist in orientation of reactants
types of catalysts
heterogeneous, homogeneous, enzymatic
heterogeneous catalyst
usually metals, interacts with reactants on the surface
homogeneous catalyst
catalyst fully mixes with reactants, same phase
enzymatic catalyst
contains an active site that facilitates the breaking/formation of bonds, drastically lowers activation energy