Immunology
1/267
Earn XP
Description and Tags
For Professor Lu's BICD 140 course (Recommendation: Study cards to memorize individual concepts with bolded terms being important, practice connecting them by writing/drawing)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
268 Terms
How Does the Immune System Affect Our Lives? | Good Way
Infectious Diseases: Protection from almost any deficiency in immunity
Cancer: Immune system functions in tumor surveillance
How Does the Immune System Affect Our Lives? | Bad Way
Autoimmune Diseases: Type I diabetes, lupus, etc.
Hypersensitivity Diseases: Allergy incidence rose 6-20%, Asthma up 3-8%
Transplantation: Immune response from the recipient is key if transplants are accepted or rejected
Heart Disease: Chronic inflammation disease
Obsessive Compulsive Disorder (OCD): Cured OCD in mice with bone marrow transplant
What is Immunity? | Self vs. Non-Self
Protection from infectious diseases
What is Immunity? | Healthy vs. Sick
Immunopathology: Mechanisms that eliminate pathogens may also cause tissue damage
What is Immunity? | Primary vs. Secondary
Immunity may infer that an individual is capable of resolving an infection after an initial encounter or protected from reinfection with that same pathogen
4 Pathogen Classes
May always cause overt disease or be opportunistic pathogens that strike when the immune system is weakened
All classes can be found inside or outside of the cell, but proliferate in specific areas
4 Pathogen Classes | Parasites
Proliferate intracellularly AND extracellularly
4 Pathogen Classes | Fungus
Proliferate extracellularly
4 Pathogen Classes | Bacteria
Proliferate intracellularly AND extracellularly
4 Pathogen Classes | Viruses
Proliferate intracellularly
3 Big Killers
Do not have a perfect vaccine that confers full immunity
Malaria
HIV
Tuberculosis
New Diseases, New Battles
SARS: 2003 in Hong Kong/China
Ebola
MERS
Covid-19
Virulence Theory
Relative pathogenicity or the relative ability to do damage to the host of an infectious agent
Pathogen is selected to carry out replication and transmission, not selected to be more destructive (calibrate virulence based on the host)
If it kills the host before it is transmitted, then the pathogen dies out
More hosts available = Better for parasitic transmission and vice versa
How is the Immune System Selected?
The immune system does not prevent disease, but it is selected to be slightly different, usually more effective
Innate Immunity
Manifest in virtually all cells in the body
Ready to go at all times w/ immediate response
Limited specificity in recognizing the different classes of pathogens
Molecular patterns as unmethylated DNA, dsRNA, cell wall components, etc.
The specificities of the innate pathogen-receptors are encoded
Adaptive Immunity
Only exists in vertebrates
Have specialized immune cells (B/T Lymphocytes)
“Right” cells are selected from a lymphocyte pool; slower response but can provide long-lasting protection
Highly specific response to unique components of pathogen
Specificities of the adaptive pathogen receptors are acquired through gene rearrangement during the organism’s lifetime
Various Ways to Prevent Bugs from Crossing Epithelia
Three Methods:
Mechanical: Usually flow of fluid, mucus, etc.
Chemical: Enzymes, antimicrobial peptides, high acidity
Microbiological: Normal flora of microbiome
Not All Bugs are Bad
Sometimes antibiotics are given, but they wipe out good and bad bugs which can have a higher risk of pathogenic microbes invading
All of the Cells of the Immune System
These cells all come from hematopoietic stem cells in the bone marrow, and there are 4 major types to know
Cells of the Immune System | Neutrophils
Phagocytosis
Reactive Oxygen/Nitrogen species
Antimicrobial Peptides: Trap pathogens w/ sticky DNA then kill themselves
Cells of the Immune System | Macrophages
Garbage Collectors who eat dead pathogens
Phagocytosis
Inflammatory mediators
Cytokines
Reactive Oxygen/Nitrogen Species
Cells of the Immune System | Dendritic Cells
Detective cells who eat dead cells but also go back to the T Cells and report what pathogen is present
T cells cannot directly recognize pathogens unlike innate immune cells
Can present antigens to B cells as well
Highly phagocytic
Costimulatory signals
Antigen presentation
Link innate and adaptive immunity
Act as APCs (antigen presenting cells)
Ralph Steinmann
Discovered dendritic cells, got Nobel Prize posthumously
Cells of the Immune System | Natural Killer Cells
Border Patrol cells that do well fighting intracellular pathogens
Macrophage activation
Lysis of viral-infected cells
Have two types of receptors:
Inhibitory Receptor: Tells them to not kill uninfected cells due to the detection of MHC Class I produced by healthy cells
Activating Receptor: Tells them to kill target because MHC Class I production is inhibited in diseased cells
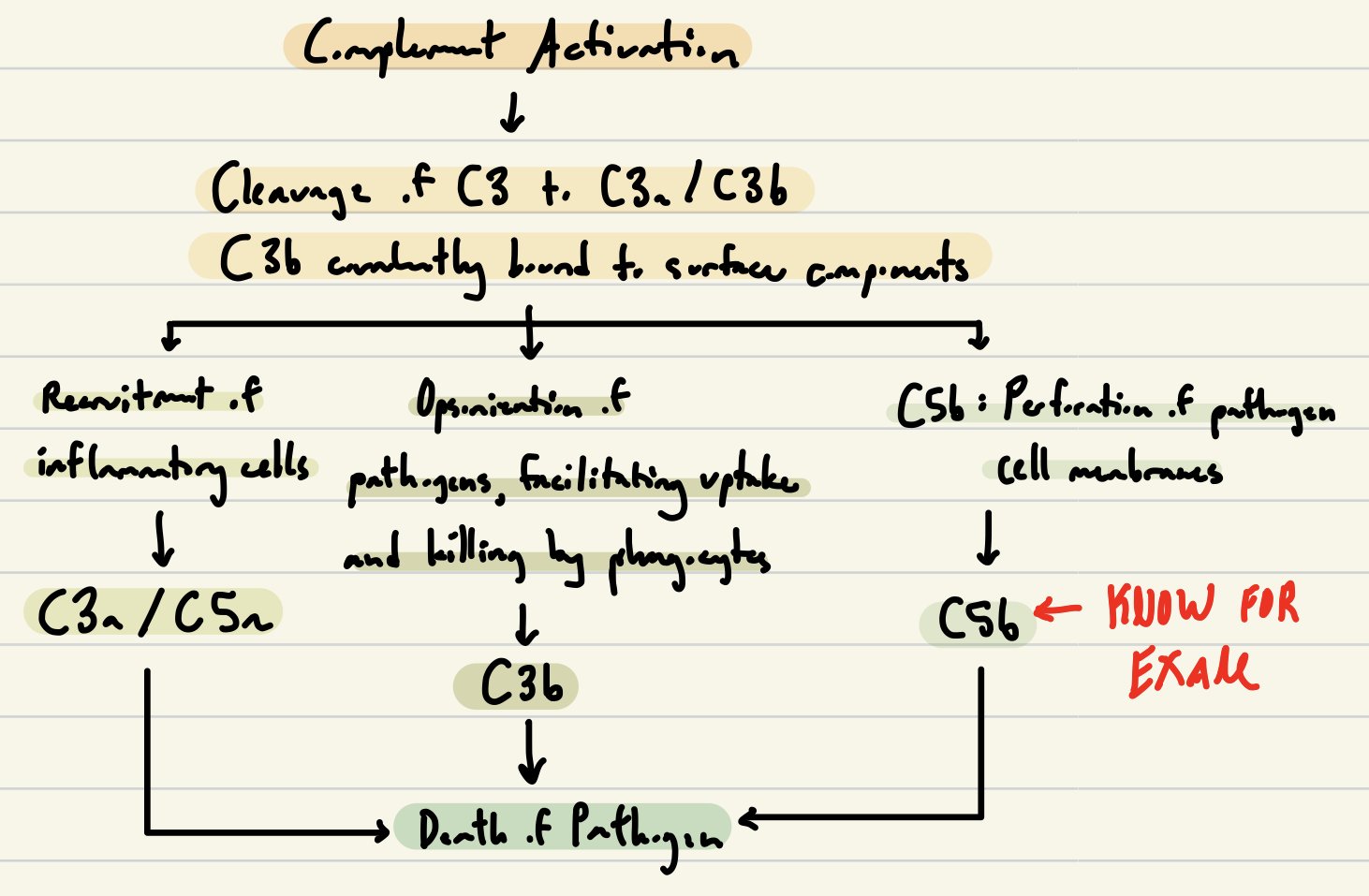
Main Innate Immune Mechanisms | Direct Killing
Phagocytosis: Eat and digest
Secrete antimicrobial peptides (antibiotics)
Lysis of microbes
Perforation of pathogen cell membrane through complement protein
Main Innate Immune Mechanisms | Accessory Role
Opsonization: Increase pathogen uptake via Complement
Recruit more immune cells (Complement and Chemokines)
Activate more immune cells (Cytokines)
Induce systemic inflammatory responses like fever (Cytokines)
What are Cytokines?
Small cell-signaling protein molecules secreted by numerous cells to affect the behavior of other cells
What are Interleukins?
Group of cytokines first seen expressed by white blood cells (leukocytes)
What are Chemokines?
Induce directed chemotaxis in nearby responsive cells (chemotactic cells)
What is Phagocytosis?
Eat and digest!
Bacterium or pathogen is phagocytosed by a neutrophil
The neutrophil ultimately dies (apoptosis) and is consumed by a macrophage
Secreting Anti-Microbial Properties
Two methods:
Transmembrane pore-forming
Modes of intracellular killing
Lysis of Microbes
Perforation of pathogen cell membranes
C5b: Subunit required for perforation
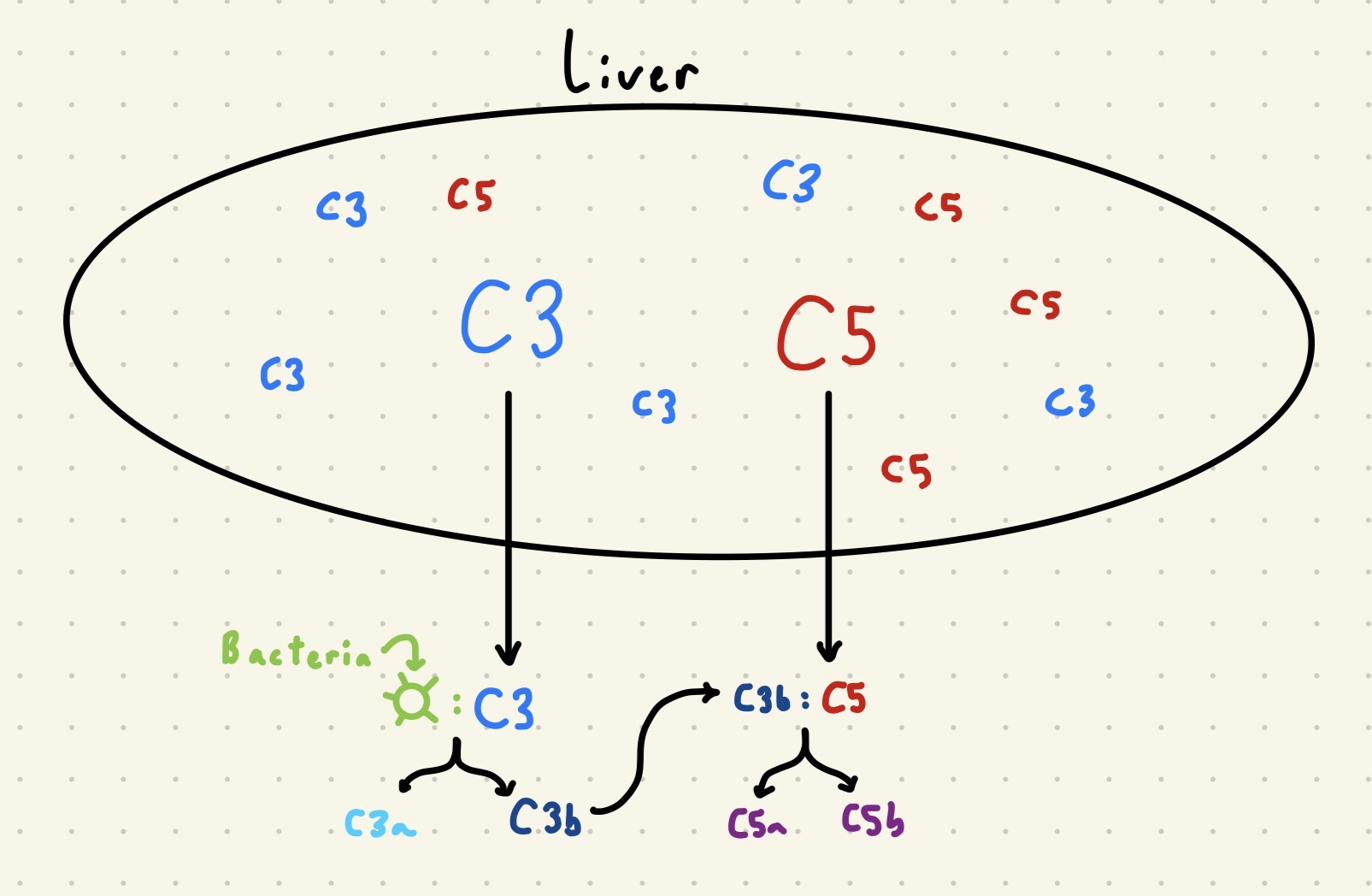
What is Complement?
System of plasma proteins made by the liver that play multiple roles in innate immunity
C3 and C5: Continuously made by the liver
C3 is in its inactive form until it binds to bacteria, changing its conformation and initiating auto-cleavage
C3a: Released
C3b: On surface of pathogen (IMPORTANT for cleavage of C5, generating C5b and C5a)
Opsonization
For large molecules
C3b facilitates phagocytosis by attaching to the bacterial cell surface
CR1 on the macrophage binds C3b on bacterium (C3b is an opsonin)
Opsonins: Any molecule that enhances phagocytosis by marking an antigen for an immune response
Wagyu analogy: Wagyu is good on its own, but seasoning makes it more delicious/irresistible (opsonins are the “seasoning” for bacteria)
Recruiting More Immune Cells: Inflammation
C3a/C5a increase vascular permeability, recruiting inflammatory cells
Can cause heat, pain, swelling, and redness!
Complement Activation Pathway (practice drawing the full pathway)
Three Pathways for Complement Activation | Alternative Pathway
First to act
Pathogen surface creates local environment conducive to complement activation
Three Pathways for Complement Activation | Lectin Pathway
Second to act
Mannose-binding lectin binds to pathogen surface
Three Pathways for Complement Activation | Classical Pathway
Third to act
Sometimes acts second, happens simultaneously with the lectin pathway
C-Reactive protein or antibody binds to specific antigen on pathogen surface (antibodies needed too)
Needs IL-6 signaling for CRP production
Innate Immune Cells Able to Produce Cytokines After Encountering Pathogens: IL-6
Cytokines can induce production of proteins in liver
IL-6: Informs liver to make mannose-binding lectin and C-reactive proteins
C-Reactive: Binds phosphocholins on bacterial surfaces
Mannose-Binding Lectin: Binds to carbohydrates on bacterial surface
Interferons
Virus-infected cells lead to interferon response (IFN-α and IFN-β)
Induce resistance to viral replication
Increase expression of ligands
Activate NK cells to kill infected cells
Innate immune cells like NK cells always ready to kill, but their effector functions increased 20-100 fold when stimulated w/ cytokines made by macrophages
Effector Cells
Mature, activated cells
Naïve Cells
Fully developed, not activated cells (have not encountered any pathogens)
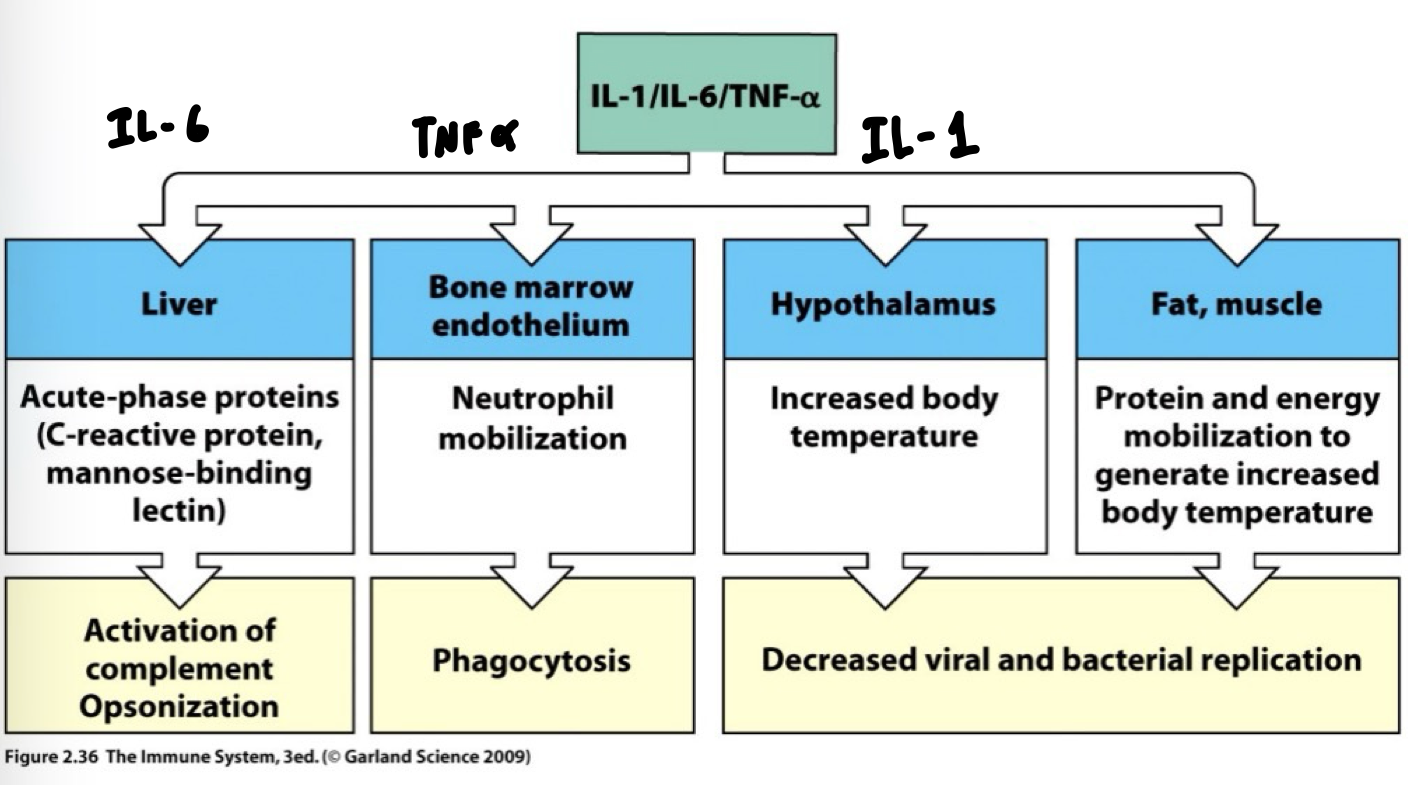
TNFα
TNFα: Get more neutrophils out of the bone marrow
Chemokines recruit cells from distal locations, provide directional signals for migrating cells
IL-1
Induce systemic inflammatory response (systemic response)
IL-1: Secreted by phagocytes travels in blood to hypothalamus, muscle
Increased body temperature to point set by the hypothalamic thermostat
Decreased viral/bacterial replication
Overview of Cytokine Functions
Septic Shock
Too much systemic innate response can be deadly
TNFα made by macrophages causes cells to make platelet activation factor which normally prevents pathogens from entering the blood
Septic Shock: Systemic edema followed by excessive coagulation and organs are starved and shut down
PAMPs
Pathogen-Associated Molecular Patterns: Molecules associated w/ groups of pathogens that are recognized by cells of the innate immune system
PRRs
Pattern Recognition Receptor: Innate immune receptors recognize PAMPs
Where Our Immune Cells Find Pathogens | Extracellular
Sites of Infection: Interstitial Spaces, Blood, Lymph Nodes
Defense: Have complement macrophages and neutrophils
Sites of Infection: Epithelial surfaces (Have TLR4/5 Receptors)
Defense: Antimicrobial peptides
Where Our Immune Cells Find Pathogens | Intracellular
Sites of Infection: Cytoplasmic (RIG-I, cGAS, and Nod2 receptors)
Defense: NK cells
Sites of Infection: Vesicular (Have TLR 3, 7, and 9 receptors)
Defense: Activated Macrophages
Different Receptor Systems
3 to know:
On Surface: Toll-like receptor
Endosomal: Toll-like receptor
Cytoplasm: Nod-like sensors for intracellular bacterial detection
CARD-family sensors for virus detection
TLRs
Toll-Like Receptors: Similar to the protein produced by Drosophila’s Toll genes (susceptible to fungal infections)
Toll Processors
Charles A. Janeway Jr. and Ruslan Medzhitov identified these receptors, caused a large drama in 2011 Nobel Prize decision
Localization of Different TLRS
Surface: TLR4-LPS and TLR5-Flagellum
Endosomal:
TLR3: dsRNA
TLR7: ssRNA
TLR9: CpGDNA
LPS (Lipopolysaccharide)
Component of cell wall in gram- bacteria
Endotoxin: Kept “within” bacterial cells; presence of endotoxins in the blood can cause unwanted inflammatory response → Septic Shock
Multiple TLRs May Recognize Different Structures in the Same Pathogen
LPS recognized by TLR4
Recognition requires LPS Binding Receptors CD14 w/ TLR4
CD: Cluster of Designation is a protocol used for the identification and investigation of cell surface molecules providing targets for immunophenotyping of cell (KNOW CD14 FOR EXAM)
MyD88 binds TLR4 and activates IRAK4 to phosphorylate TRAF6
Leads to release of NFKB, activating transcription of genes for inflammatory response
MyD88 Pathways
Two cytoplasmic pathways:
Dependent: Uses IRAK4 as adaptor protein
Trigger NFKB Pathway
Produce inflammatory cytokines (IL-1, IL-6, TNFα)
Independent: Use TRIF as an adaptor protein triggered by TLR4
Trigger IRF3 pathway
Produce IFNs to fight viral infections (IFNα/β)
Will continue to function if the Dependent pathway is shut down
IRFs
Interferon Regulatory Factor proteins that regulate transcription of interferons
Nod-like receptor can sense bacterial infection in cytoplasm and induced NFKB-mediated inflammation
CARD-family sensors (RIG-1) can recognize cytoplasmic dsRNA (and ssRNA) viral infection, use interferons to respond
Cyclic GMP-AMP Synthase
cGAS: Recognize cytoplasmic DNA viral infection
Dendritic Cells
Highly phagocytic, link innate and adaptive immunity
Professional antigen presenting cells (APCs)
Antigen: Part of pathogen that antibodies bind to
Adaptive immune cells (ie T Cells) do not recognize pathogens unlike innate immune cells
Ralph Steinmann
Contributed to dendritic cell discovery/function
What is the Immune System?
Varying locations depending on whether it’s adaptive or innate
Innate Immune System:
Tissue (mostly exists here!)
Lymphoid Organs, Blood
Adaptive Immune System:
Lymphoid Organs (mostly exists here!)
Blood, Tissue
Lymph
Plasma that has leaked from the blood into the tissues, collected through lymphatic vessels
Lymphoid Organs
Contain lymphocytes, but also other types of cells and structure to support the production, maintenance, and circulation of lymphocytes
Lymphatic vessels collect lymph to carry back to lymphoid organs
Primary Lymphoid Organs
Central Lymphoid Organ: Where lymphocytes are generated (from immature progenitor cells)
From bone marrow
T Cells go to the Thymus to finish maturation
Secondary Lymphoid Organs
Peripheral Lymphoid Organs: When mature naïve lymphocytes reside and an adaptive immune response is initiated
Lymphocytes Continuously Survey the Secondary Lymphoid Organs for Evidence of Infection
Lymphocytes are unique as they travel through blood and lymph
Secondary lymphoid organs compartmentalize the infection and provide a meeting place for the cells of the adaptive immune response
T-Cell Area: Mostly T-Cells
Exist in Lymph Nodes
Arterial Vein: “I-5” that lets them travel to rest of body
Efferent Lymphatic Vessel
Lymphoid Follicle
Dedicated area for B Cells in lymph nodes
Spleen
Deals with pathogens that make it to the blood
No connection to lymphatics
Key Difference: Only connected via blood vessels unlike LN, where both pathogens and lymphocytes enter spleen via the blood
Gut-Associated Lymphoid Tissue
“The adjacent city”: Only separated w/ epithelial cells from the gut
Similar microanatomy to spleen and LN, but differ in:
Route of pathogen entry (direct delivery across mucosa)
Migration pattern of lymphocytes after activation (tend to stay within mucosal system)
M Cells: Create a “window” in the epithelium that allows for immune cells, especially dendritic cells to reach out and detect pathogens
The Adaptive Response is Specific to the Current Infection
Antibodies made during infection w/ vaccine bind to the virus and prevent reinfection w/ virus
Ex: Antibodies made for measles don’t bind to influenza
The Adaptive Immune Response
Diversity and Clonal Selection
Only adaptive immune population w/ receptors that recognize the specific pathogen will respond during a particular infection
T-Cells: Activated by dendritic cells, only specific few
Clonal Selection
Four Principles (KNOW FOR EXAM):
Every lymphocyte bears a single type of receptor with a unique specificity
Lymphocyte Activation: Interaction between a foreign molecule and a lymphocyte receptor capable of binding that molecule w/ high affinity
Differential effector cells derived from an activated lymphocyte will bear receptors of identical specificity to those of the parental cell
Lymphocytes bearing receptors specific for ubiquitous self-molecules are deleted at an early stage in lymphoid cell development (deleted from mature repertoire)
Effectors of Adaptive Immunity
Two types:
B Cells: Secrete their antigen receptor (antibody)
Humoral immunity: Involves substances found in the humours (body fluids)
Attack external bacteria (via antibodies)
T Cells: Do not secrete their antigen receptor
Cell-Mediated Immunity: Activation of macrophages lead to microbial killing and lysis of infected cells
Attack virus-infected cells and phagocytosed microbes in macrophages
Isotype Switching
The constant region of the heavy chain of an antibody changes to interact with different effector molecules, which may increase the affinity the antibody has for a specific antigen (antigen specificity DOES NOT change)
T Helper Lymphocytes
CD4: Helper T Cells
Interact w/ macrophages to activate them (TH1)
Cytotoxic Lymphocytes
CD8: Kill virus-infected cells
Antigen Receptors
Recognition of Pathogens
B Cell Receptor: Recognize pathogens in native form
Bound to B Cell: Y shape w/ two binding sites (antibody)
T Cells: Need APCs to process and present antigens to them
TCR
Binds to antigen derived peptides bound to MHC (Major Histocompatibility Complex) molecules
CD8 T cells cannot “see” antigen w/o MHC Class I
CD4 T cells need MHC Class II on surface of dendritic cell
Functional Outcome of B Cell Activation is Antibody Production
Antibodies bind directly to toxin/pathogens
Neutralization: Pathogen cannot invade cells
Opsonization: Increase phagocytosis
Complement Deposition
Ultimately leads to degradation by a macrophage
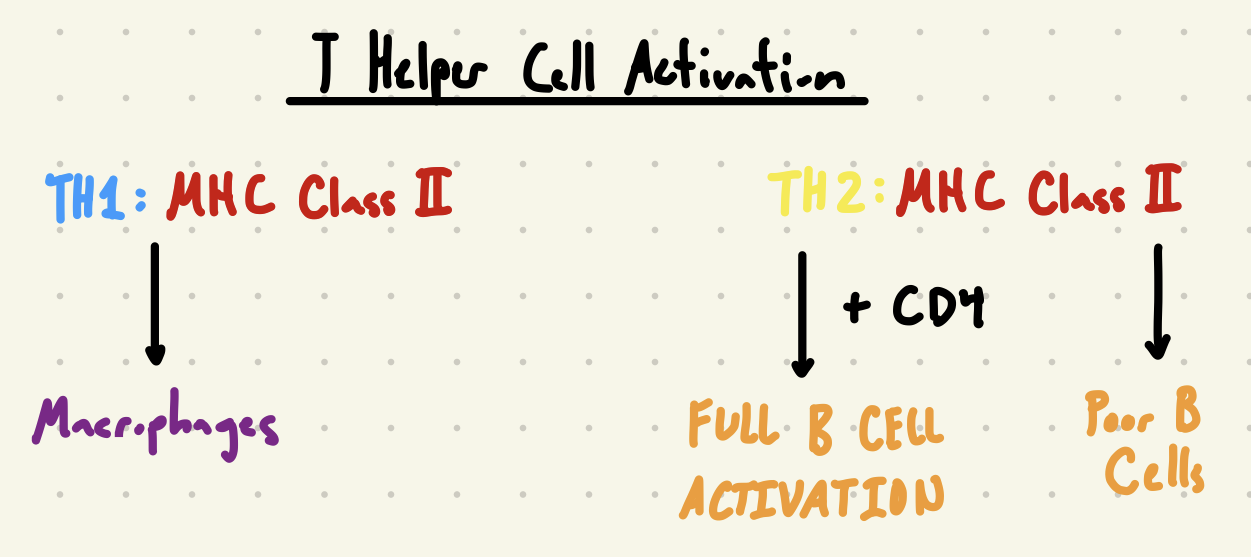
Functional Outcome of T Helper Cell Activation is the Activation of Other Cell Types
TH1: Recognizes complex of peptide antigen w/ MHC Class II and activate macrophages
TH2: Recognizes complex of peptide antigen w/ MHC Class II and activates B Cell (and CD4-TH2 cells for full B Cell Activation)
Poor quality antibodies created without T Cells, so B Cells’ response is limited
Functional Outcome of Cytotoxic T Cell Activation is the Lysis of Infected Cells
Peptide fragments of viral proteins bound by MHC Class I in ER, transported to cell surface where cytotoxic T Cells recognize complex of virus infection
NK cells are Plan B when these cells fail to kill infected cells
Principle Antigen Presenting Cells (APCs) for T Cells
Dendritic cells found in the lymphoid organs, skin, and connective tissue
Macrophages found everywhere in body
Circulating Monocytes: Can turn into macrophages or dendritic cells early on
B Cells: Present antigens bound to antibody receptors
Flow Cytometry Applications
Visualization of samples on a single cell level
Can find a rare cell type amongst a sample comprised of non-relevant counts
Sorting of distinct populations within a heterogeneous sample
Widely used in immunology
How Does Flow Cytometry Work?
The Basics: A sample of cells is labeled with antibodies conjugated with fluorochromes via incubation at low temp
Antibodies specific for cell surface receptors, intracellular receptors
Cell suspension is forced into a tiny stream of liquid so that only one cell enters the machine at a time
Each cell and any antibody attached to it intercept intercept the laser beam and excited to a higher energy state
Energy is released as a photon of light (w/ fluorochromes, photon has distinct spectral properties)
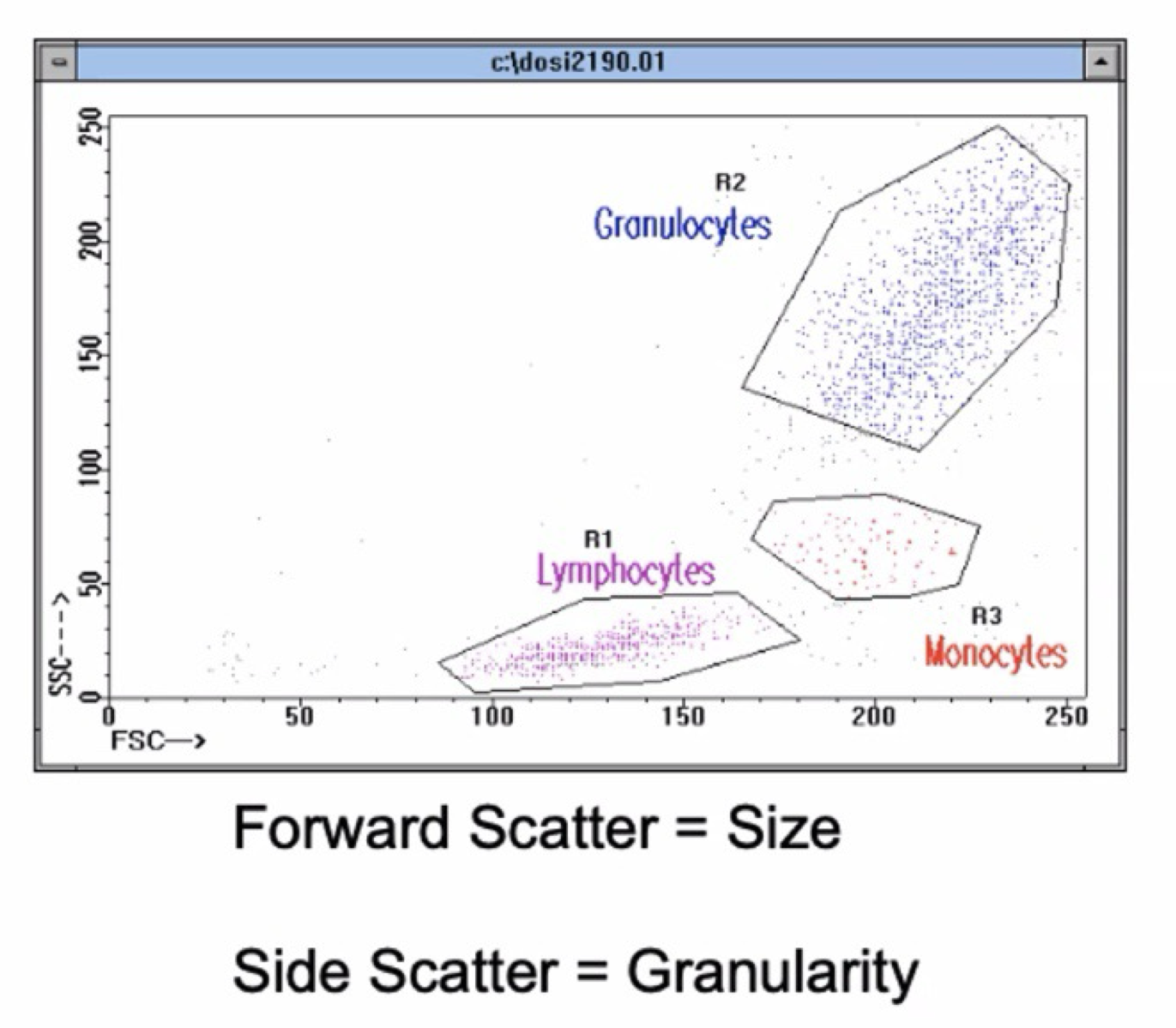
Interpreting Graphs: Scatter
Cells are not opaque, they can create side scatter due to granularity
Granularity: Number of things in the cell
Low side scatter can indicate that the cells are lymphocytes
Forward scatter: Due to size
Density: Color brightness
Identify populations by high or low, not by the specific number
Location on graph is relative
KNOW GRAPH SHOWN FOR EXAM
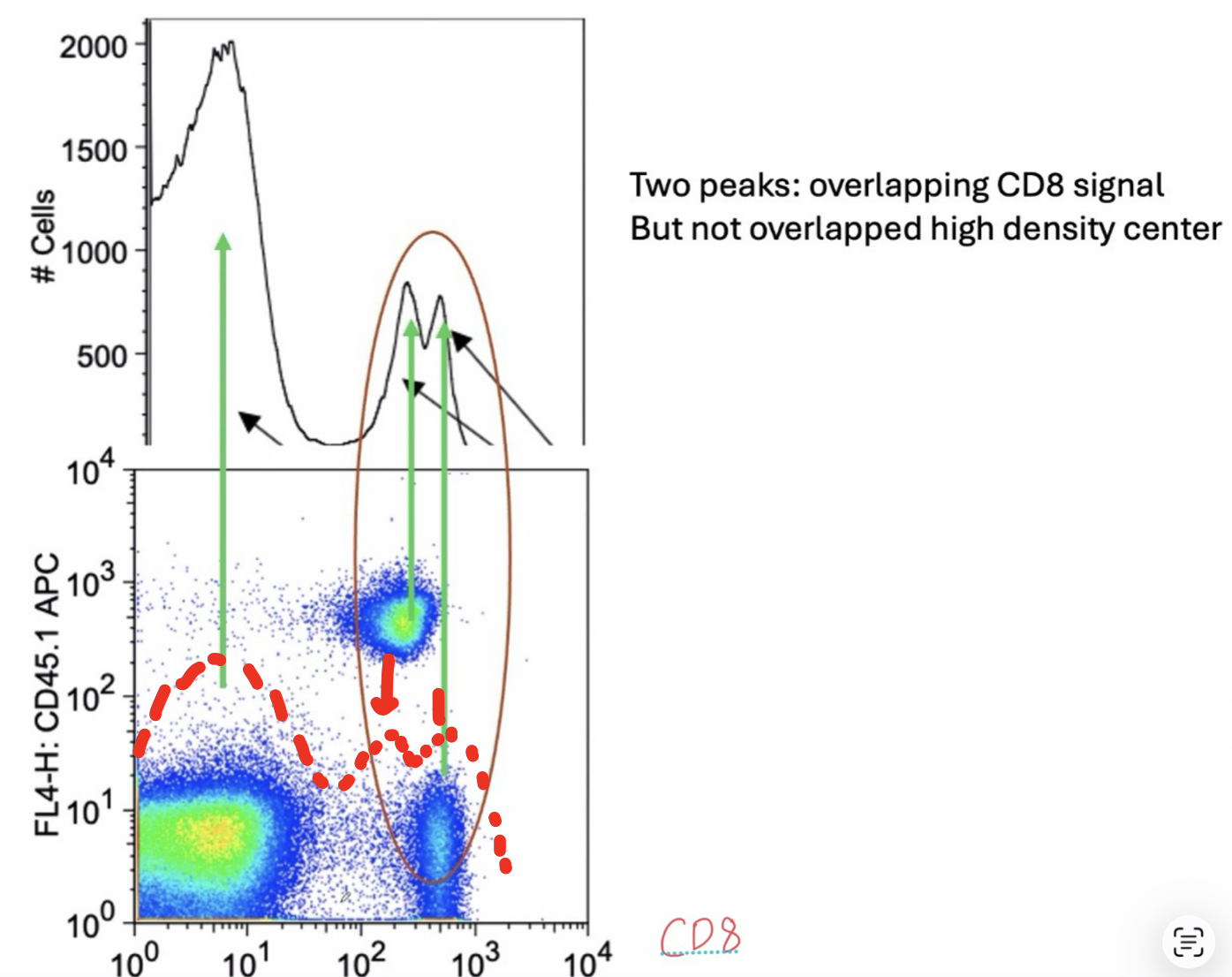
Translating Flow Cytometry Graphs to Peaks
Darker color: Less cells
x/y axis: Fluorescent Intensity = Expression level
Bottom left corner: Double negative population (does not have high levels of either cell population listed on axes)
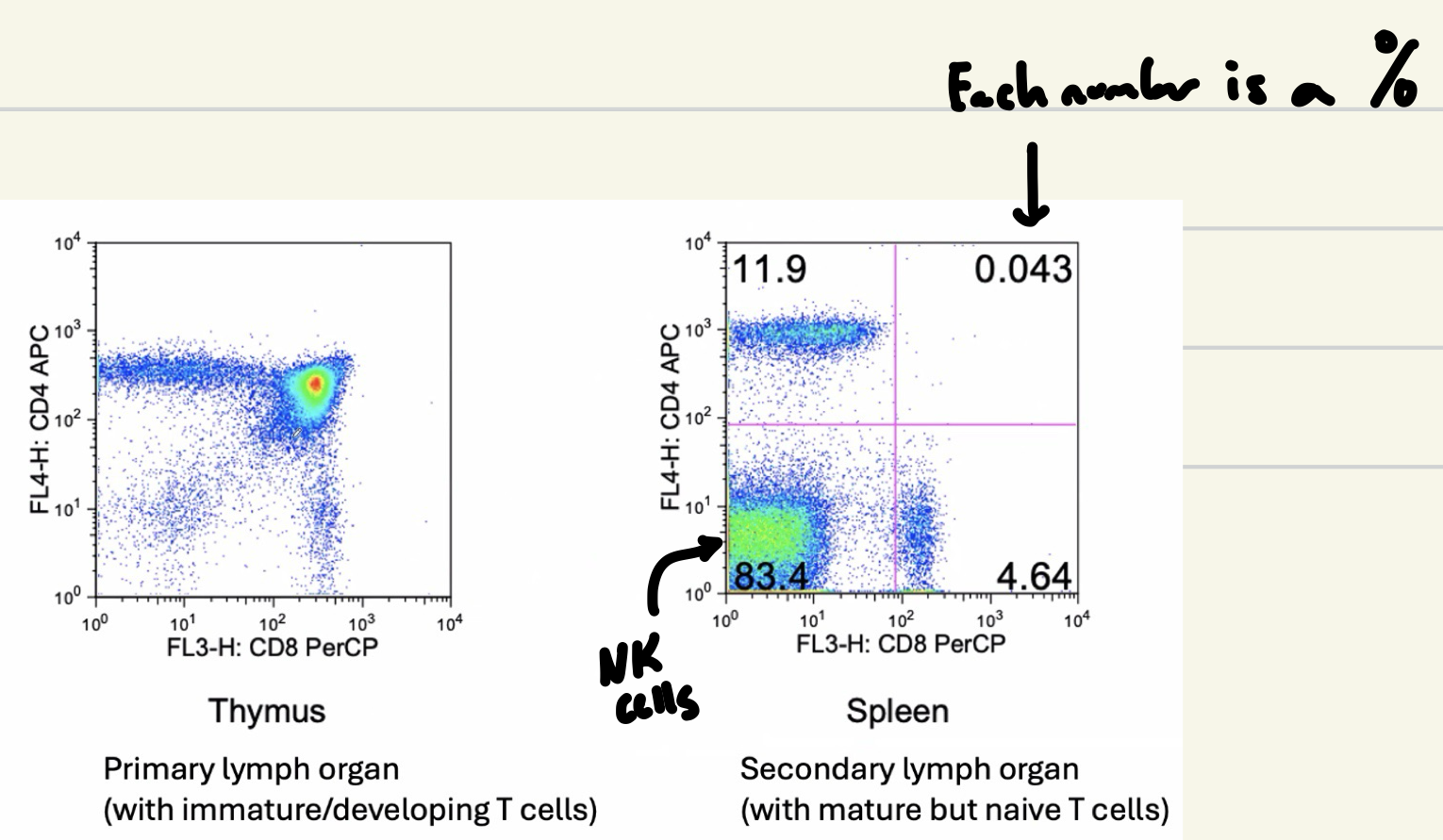
Differentiating Between CD4 (Helper) and CD8 (Cytotoxic) T Cells
Thymus: High population of mature T cells here
Spleen: Secondary lymph organ with mature but naïve T cells
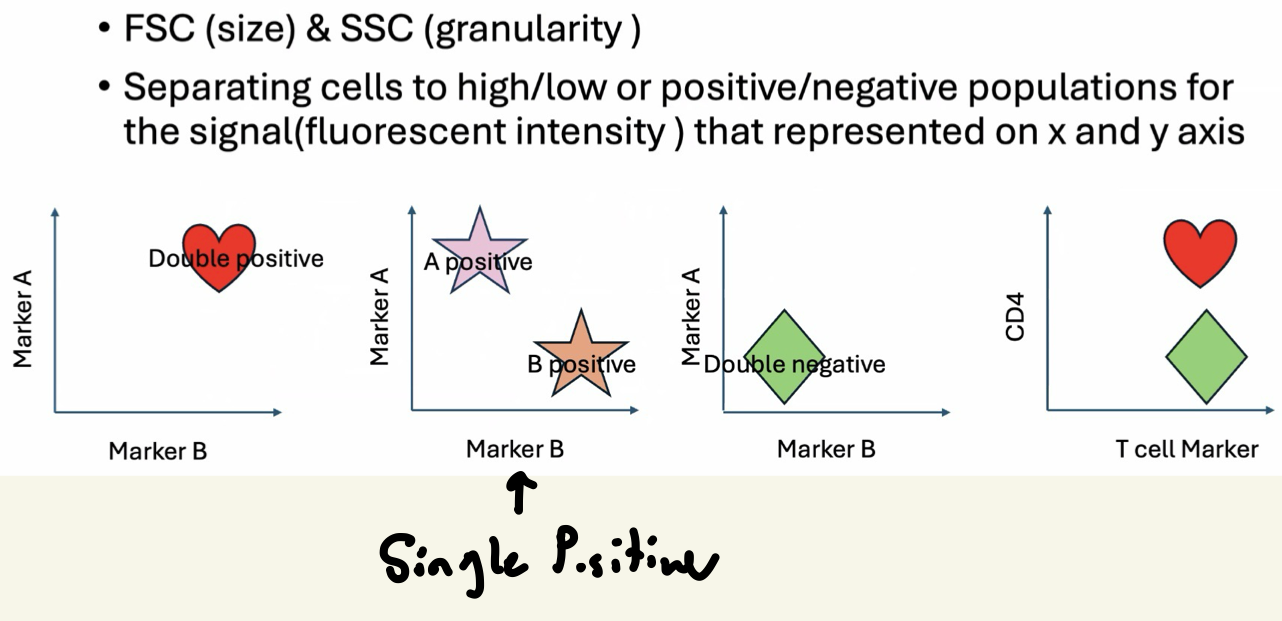
Flow Cytometry Overview (KNOW FOR EXAM)
INF-α and INF-β
Three functions:
Slow down viral replication
Make cells a better target for NK cells
Can activate NK cells directly
In covid, viral load was reduced and symptoms decreased w/ MORE interferons present, and vice versa
Type I interferons have delayed response, caused more immunopathology any many deaths (increase pathology later on)
GALT
Gut-associated lymphoid tissue is a specialized immune system in our digestive tract
Tolerance
Our body needs tolerance against things that are not dangerous (ie commensal bacteria in our gut) to avoid constantly attacking everything (leads to autoimmune disease, allergies otherwise)
How do We Produce an Infinite Variety of Antibodies?
Each B Cell expresses a unique antibody receptor that is selected by antigen
Plasma Cells
Effector B Cells that secrete large volumes of antibodies
Antibody Structure and Generation of B Cell Receptor/Antibody
Antibodies (Ab) are globular proteins that specifically bind to foreign molecules
Made by B Cells, differentiated to become plasma cells
That ONE antibody is a copy of its B Cell receptor, so antibodies are secreted B Cell receptors
Antigens are anything that bind to an antibody
Fc and Fc receptor interaction facilitates antibody-mediated opsonization!
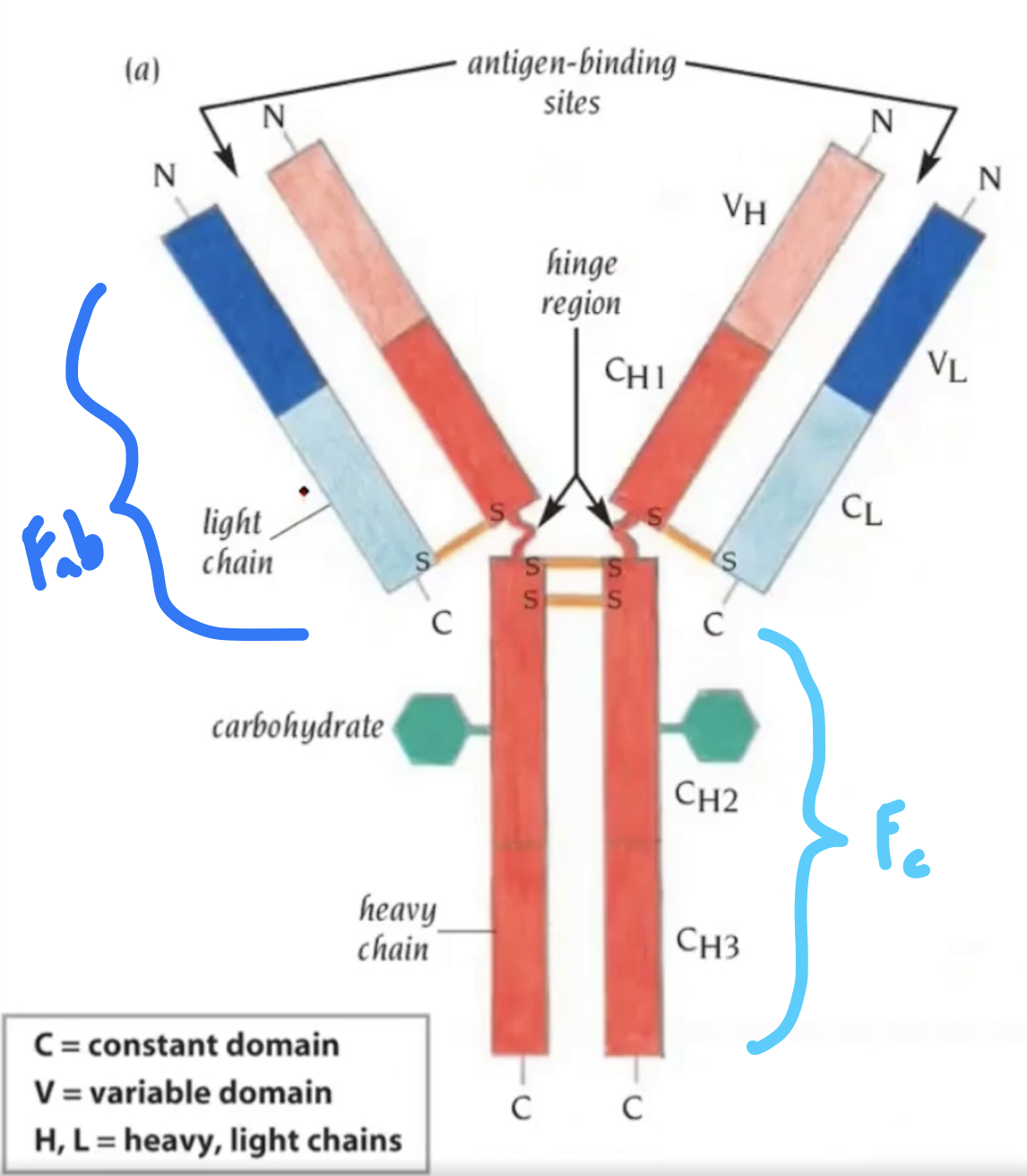
Antibody Structure | Heavy Chain
5 different constant regions (IgM, IgG (1-4), IgA (1-2), IgD, IgE…) with 9 total in humans
Antibody Structure | Light Chain
5 total in humans
Igκ and Igλ (1-4) in humans
Antibody Structure | Variable Region
Antigen recognition/binding site with 10,000,000,000,000 possible variable regions
Unlimited diversity
Antibody Structure | Constant Region
Biological activity occurs on heavy chains in constant region