Orgo Chapter 6: Chemical Reactivity and Mechanisms
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
20 Terms
What are ionic reactions/polar reactions?
Reactions that involve the participation of ions as reactants, intermediates, or products, intermediates in most cases
What is a nucleophile?
An electron-rich molecule or ion that is capable of donating a pair of electrons
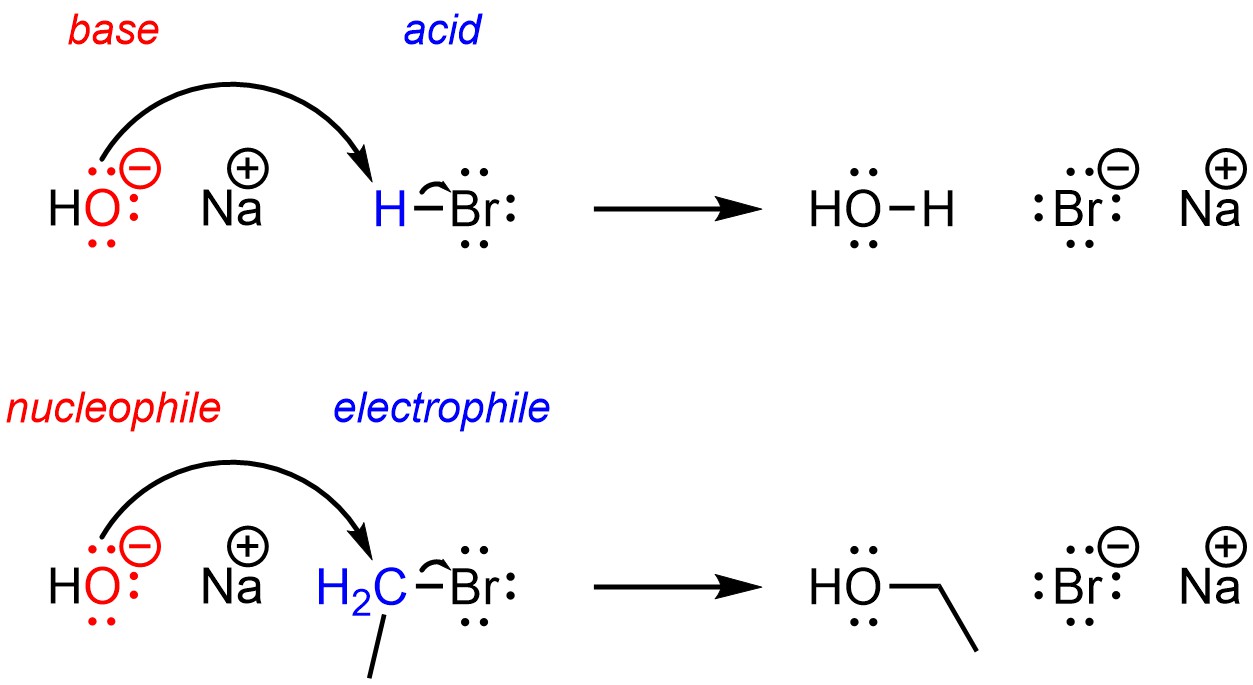
What is an electrophile?
An electron-deficient molecule or ion that is capable of accepting a pair of electrons

Electrtophiles are what type of acids?
Lewis Acids
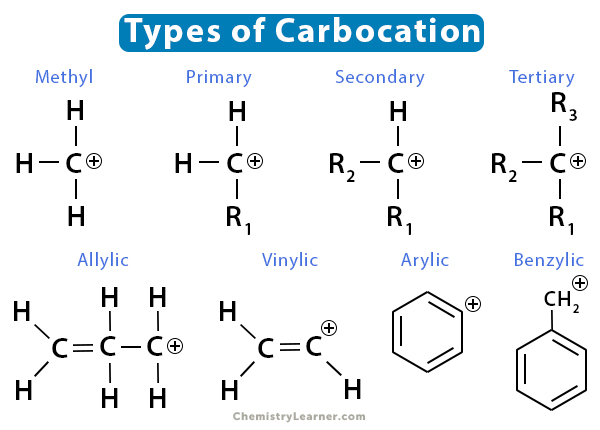
What are carbocations?
Ions in which a carbon atom bears a positive charge (as a results of an empty p orbital) and are electrophilic
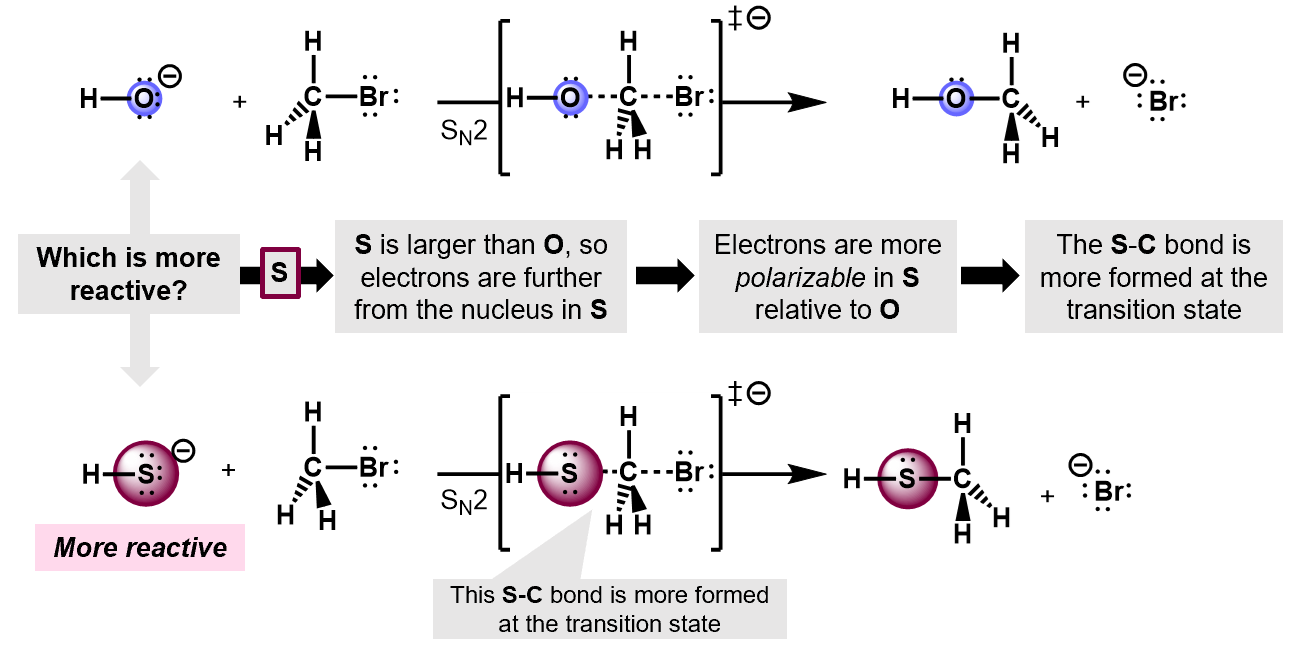
What is polarizability?
Describes the ability of an atom to distribute its electron density unevenly as a result of external influences
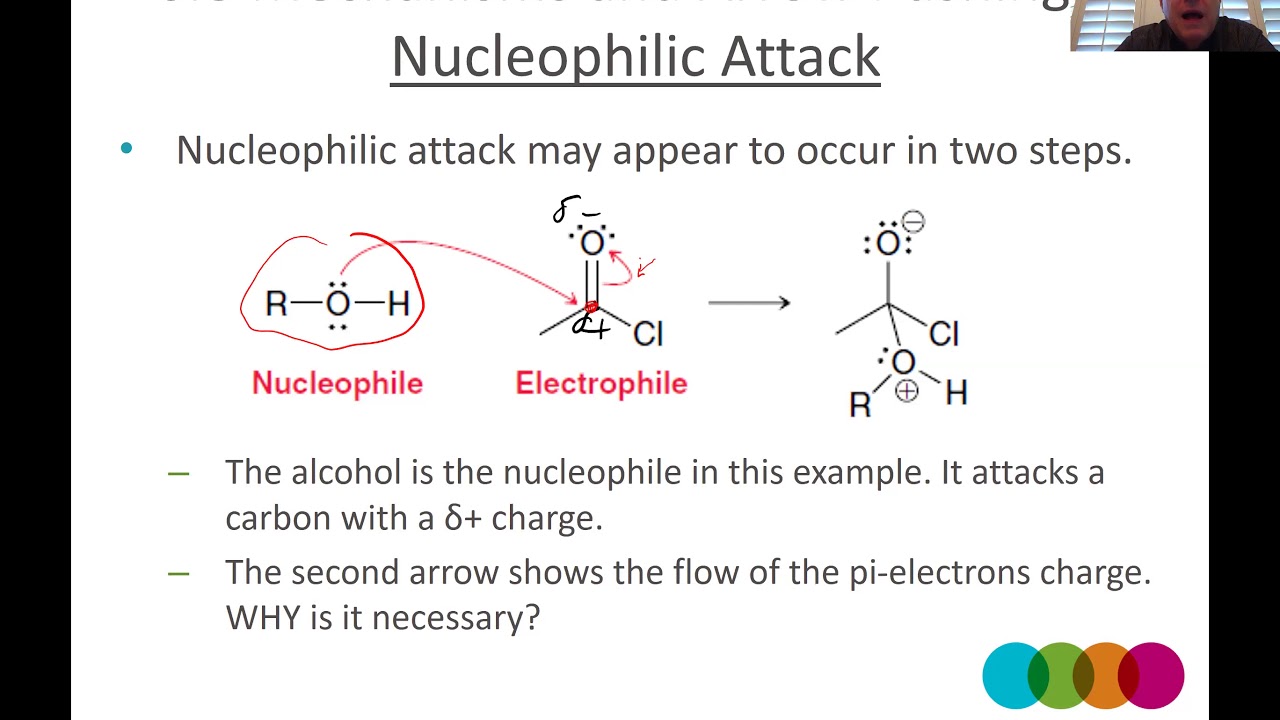
What is a nucleophilic attack?
A nucleophilic (electron-rich) donates an electron pair to form a bond with an electrophile (electron-deficient). Nucleophile uses its lone pair of electrons or a pi bond to form a new bond
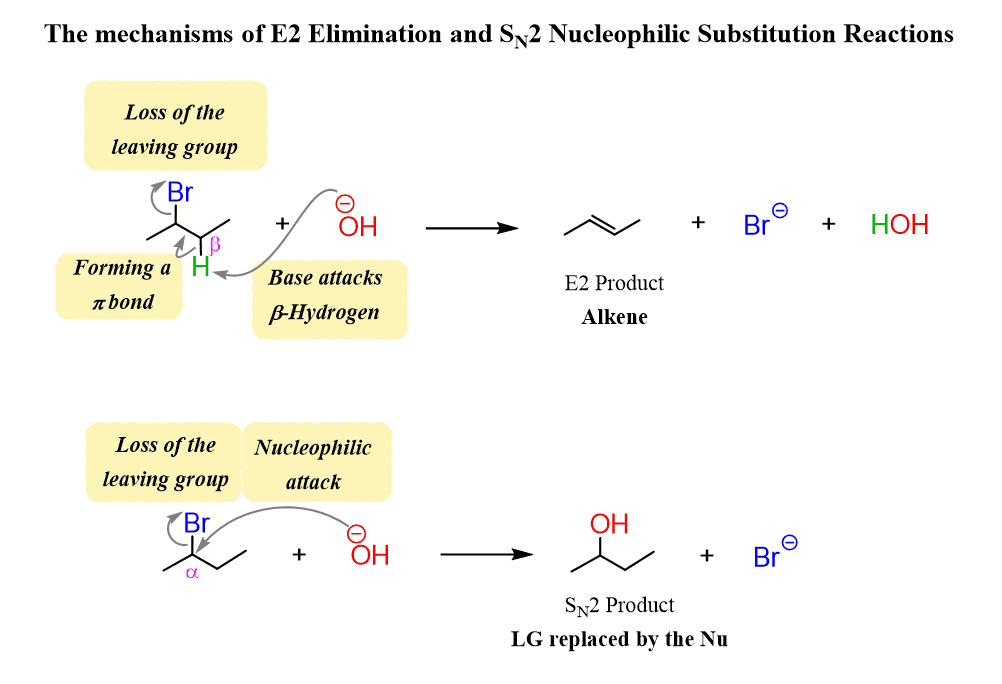
What is the loss of a leaving group?
Arrow originates from the bond between the carbon atom and the leaving group, pointing towards the leaving group, signifying the movement of electrons as the bonds breaks
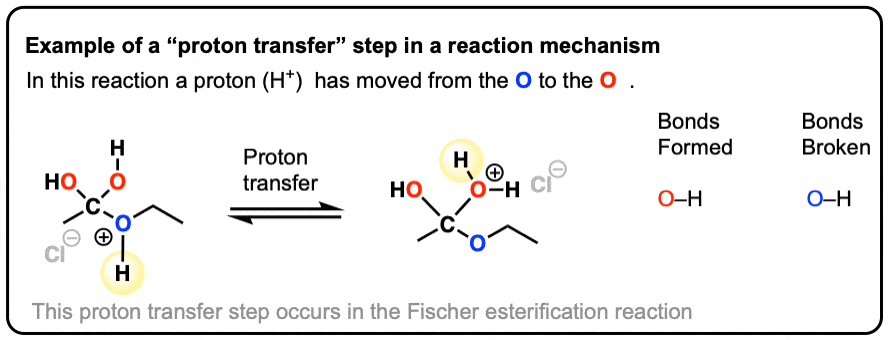
What is a proton transfer?
The movement of electrons from a base (electron-rich site) to a proton (H+) from an acid, forming a new bond between the base and the proton.
First arrow: Base is attacking the proton, forming a new bond between the base and proton
Second arrow: Electrons in the bond between the acid and the proton moving to form a new bond (or lone pair) on the acid
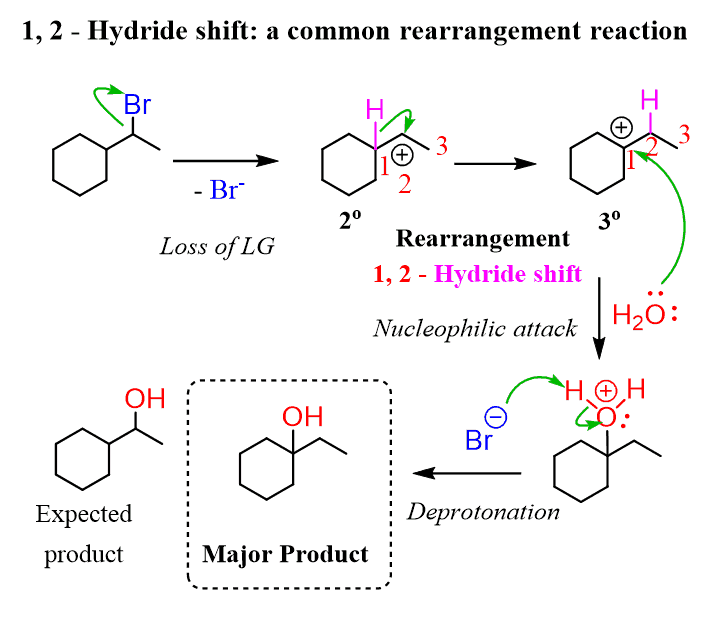
What is a rearrangement?
Reorganization of a molecules structure through the movement of an atom or group resulting in a structural isomer of the original molecule
What are the four characteristics of arrow-pushing patterns?
Nucleophilic attack
Loss of a leaving group
Proton Transfer
Rearrangement
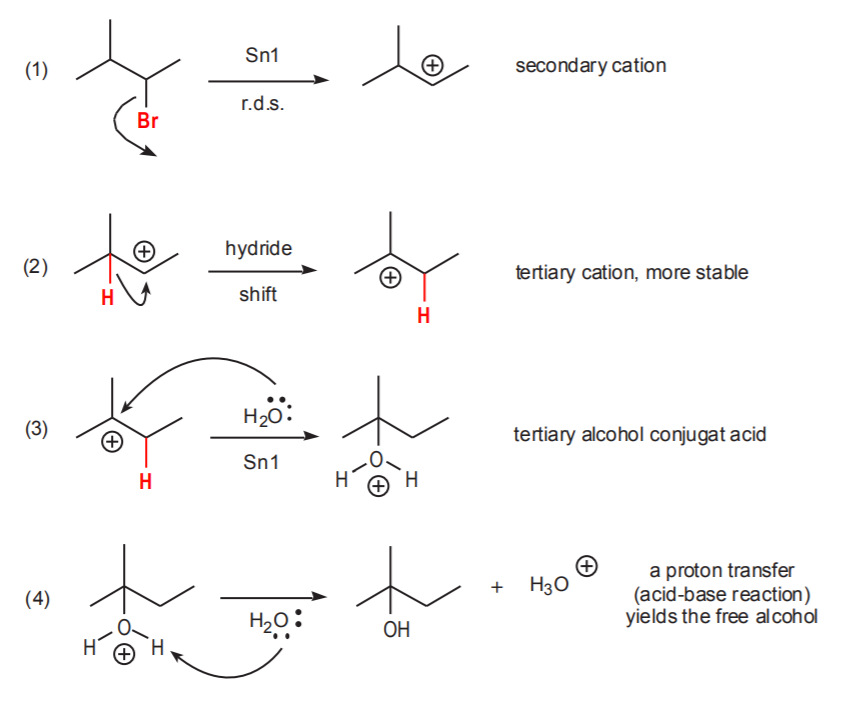
What is a carbocation rearrangement?
A carbocation undergoes a hydride shift or a methyl shift to produce a more stable carbocation
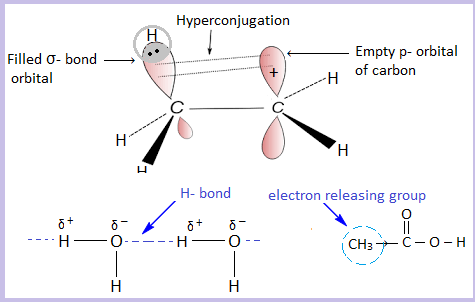
What happens in a result of hyperconjugation?
Tertiary carbocations are more stable than secondary carbocations, which are more stable than primary carbocations
What is a concerted process?
When a single mechanistic step involves two simultaneous arrow-pushing patterns
What do all ionic mechanism have in common regardless of complexity?
All ionic mechanisms are different combinations of the four characteristic arrow-pushing patterns
Where should you NEVER draw an arrow?
Never draw an arrow that gives a 5th orbital to a second row element (C, N, O, F)
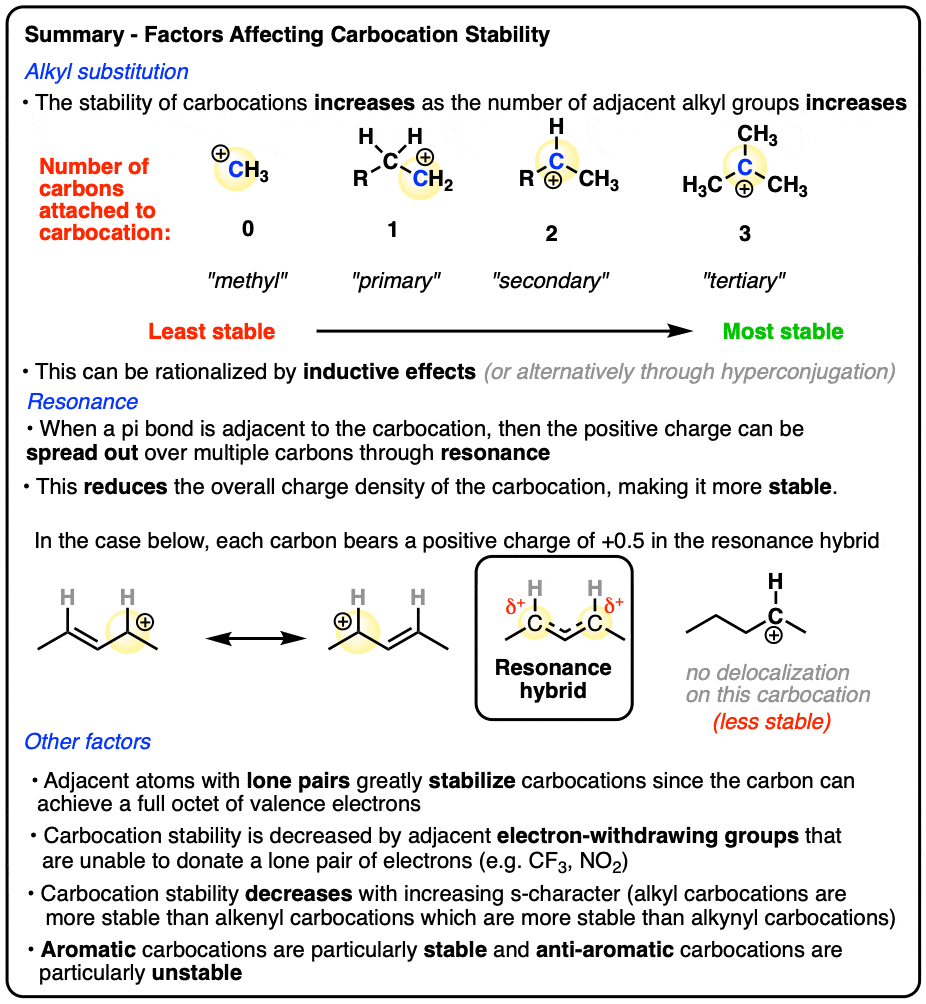
When will a carbocation rearrangement more likely occur?
A carbocation rearrangement will occur if it leads to a more stable carbocation
Tertiary carbocations generally do not rearrange unless?
Tertiary carbocations generally do not rearrange unless a rearrangement will produce a resonance-stabilized carbocation, such as a tertiary allylic carbocation
What is a tertiary allylic carbocation?
A positively charged carbon atom (carbocation) that is both tertiary (attached to 3 other carbon atoms) and allylic (adjacent to a double bond). They are highly stable due to resonance and hyperconjugation effects
What are the circumstances when irreversible reaction arrows are used?
For a nucleophilic attack involving a strong nucleophile that is also a poor leaving group
For a protein transfer step in which there is a bast difference in pKa values between acids on either side of the equilibrium
For a carbocation rearrangement
