Chemical reactions and equations Unit 6
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
20 Terms
Diatomic elements
7 pure elements that exist as pairs in nature — the only pure covalent bonds
How to remember the covalent bonds?
BRINCLEHOF
Conservation of matter
In all chemical and physical changes, matter is neither created or destroyed
The total mass in a chemical reaction remains _______
constant
Chemical Reaction
process in which a new substance with different physical and chemical properties is produced
Reactants
The substance you start with
Products
The substance you end up with
In a chemical reaction…
Atoms are rearranged into new molecules
Arrow
Separates the reactant from the product
(aq)
aqueous solution, dissolved in water

double arrow
indicates a reversible reaction

Can have heat on top of arrow as well
Shows that heat is supplied to the reaction
Any element over arrow
Indicates that a catalyst is supplied
Catalyst
a substance that speeds up a chemical reaction
valence electrons
their arrangement determines whether or not an atom will bond
energy
needed to initiate a chemical reaction
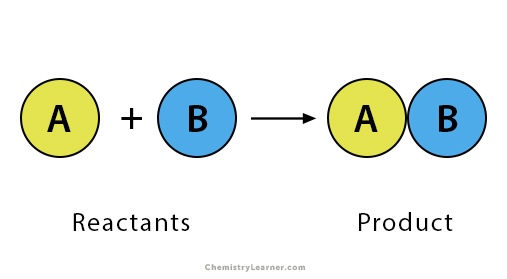
Synthesis
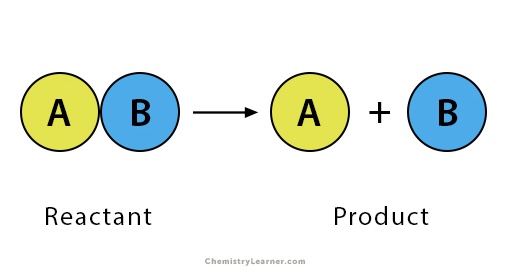
Decomposition
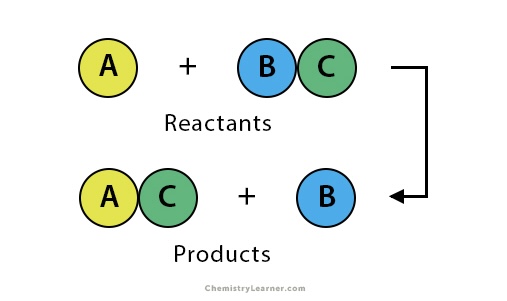
Single replacements
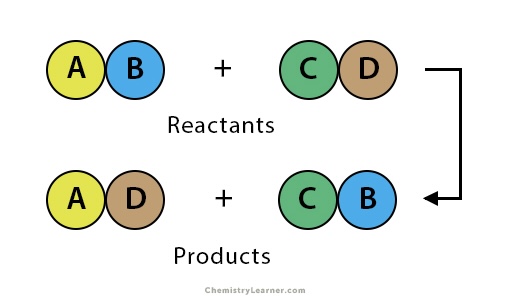
Double replacement
Has h20 and c02