Alkyne Addition Reactions (CH 9)
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
14 Terms
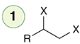
Elimination
Reagent:
1)NaNH2(excess)
2)H2O
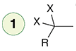
Elimination
Reagent:
1)NaNH2(excess)
2)H2O
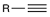
Hydrohalogenation (2 eq)
Reagent: HX (excess)
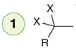
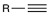
Hydrohalogenation (1 eq)
Reagent: HX (1 eq)
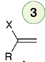
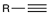
Acid-catalyzed hydration
Reagent:
HgSO4
H2SO4, H2O
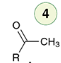
Hydroboration-Oxidation
Reagent:
1) R2BH
2)H2O2, NaOH
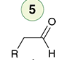
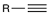
Halogenation (1 eq)
Reagent:
X2 (1 eq)
CCl4
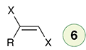
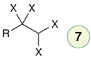
Halogenation (2 eq)
Reagent:
X2 (excess)
CCl4
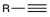
Ozonolysis
Reagent:
1) O3
2) H2O
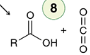
Alkylation
Reagent:
1)NaNH2
2)RX


Dissolving Metal Reduction
Reagent:
Na
NH3 (l)
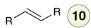

Hydrogenation
Reagent:
H2
Pt/Pd
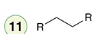

Hydrogenation with a Poisoned Catalyst
Reagent:
H2
Lindlar’s Catalyst
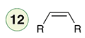
Chapter 9 Splurge
SECTION 9.1
A triple bond is comprised of three separate bonds: one σ bond and two π bonds.
Alkynes exhibit linear geometry and can function as bases or as nucleophiles.
SECTION 9.2
Alkynes are named much like alkanes, with the following additional rules:
The suffix “ane” is replaced with “yne.”
The parent is the longest chain that includes the C≡C bond.
The triple bond should receive the lowest number possible.
The position of the triple bond is indicated with a single locant placed either before the parent or the suffix.
Monosubstituted acetylenes are terminal alkynes, while disubstituted acetylenes are internal alkynes.
SECTION 9.3
The conjugate base of acetylene, called an acetylide ion, is relatively stabilized because the lone pair occupies an sp-hybridized orbital.
The conjugate base of a terminal alkyne is called an alkynide ion, which can only be formed with a sufficiently strong base, such as NaNH2.
SECTION 9.4
Alkynes can be prepared from either geminal or vicinal dihalides via two successive E2 reactions.
SECTION 9.5
Catalytic hydrogenation of an alkyne yields an alkane.
Catalytic hydrogenation in the presence of a poisoned catalyst (Lindlar’s catalyst or Ni2B) yields a cis alkene.
A dissolving metal reduction will convert an internal alkyne into a trans alkene. The reaction involves an intermediate radical anion and employs fishhook arrows, which indicate the movement of only one electron.
SECTION 9.6
Alkynes react with HX via a Markovnikov addition.
One possible mechanism for the hydrohalogenation of alkynes involves a vinylic carbocation, while another possible mechanism is termolecular.
Addition of HX to alkynes probably occurs through a variety of mechanistic pathways all of which are occurring at the same time and competing with each other.
Treatment of a terminal alkyne with HBr and peroxides gives an anti-Markovnikov addition of HBr.
SECTION 9.7
Acid-catalyzed hydration of alkynes is catalyzed by mercuric sulfate (HgSO4) to produce an enol that cannot be isolated because it is rapidly converted into a ketone.
Enols and ketones are tautomers, which are constitutional isomers that rapidly interconvert via the migration of a proton.
The interconversion between an enol and a ketone is called keto-enol tautomerization and is catalyzed by trace amounts of acid or base.
Hydroboration-oxidation of a terminal alkyne proceeds via an anti-Markovnikov addition to produce an enol that is rapidly converted into an aldehyde via tautomerization.
In basic conditions, tautomerization proceeds via a resonance-stabilized anion called an enolate ion.
SECTION 9.8
Alkynes can undergo halogenation to form a tetrahalide.
SECTION 9.9
When treated with ozone followed by water, internal alkynes undergo oxidative cleavage to produce carboxylic acids.
When a terminal alkyne undergoes oxidative cleavage, the terminal side is converted into carbon dioxide.
SECTION 9.10
Alkynide ions undergo alkylation when treated with an alkyl halide (methyl or primary).
Acetylene possesses two terminal protons and can undergo two separate alkylations.
SECTION 9.11
An alkene can be converted into an alkyne via bromination followed by elimination with excess NaNH2.