ochem 18: amines and amides
1/86
Earn XP
Description and Tags
amines, naming, drawing, 1,2,3 amines, heterocyclic, bp, solubility, neurotransmitters, amides, amidation, bp, solubility, hydrolysis, naming, drawing
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
87 Terms
amines and amides are organic compounds that include
nitrogen
amines are derivatives of
ammonia (NH3)
what is a amine
•derivatives of ammonia, NH3, in which one or more hydrogen atoms are replaced with alkyl or aromatic groups.
•contain N attached to one or more alkyl or aromatic groups.
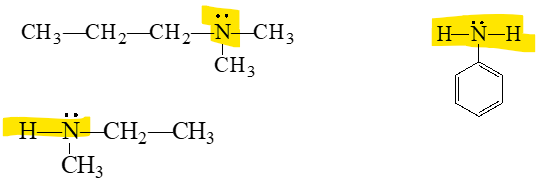
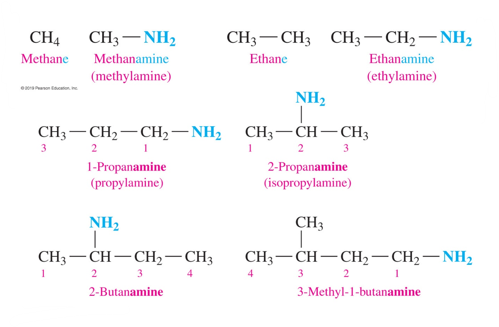
how do we name amines (IUPAC)
-In the IUPAC names for amines, the e in the corresponding alkane name is replaced with amine
- amines with 3 or more carbons are numbered to show the position of the —NH2 group and any other substituents.
-If there is an alkyl group attached to the nitrogen atom, the prefix N and the alkyl name are placed in front of the amine name.
-If there are two alkyl groups bonded to the N atom, the prefix N is used for each, and they are listed alphabetically.
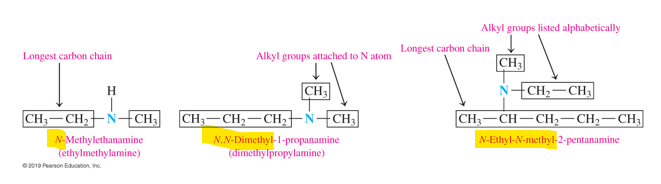
if an amine has an alkyl group or 2 attached to the N atom, how do we name IUPAC
•If there is an alkyl group attached to the nitrogen atom, the prefix N and the alkyl name are placed in front of the amine name.
•If there are two alkyl groups bonded to the N atom, the prefix N is used for each, and they are listed alphabetically.
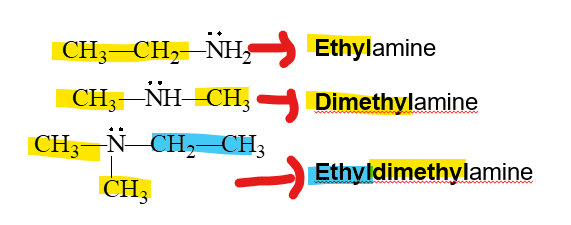
how do we name amines Common name
-used when alkyl groups are not branched
-List the names of the alkyl groups bonded to the N atom in alphabetical order in front of amine.
-Use prefixes di and tri to identify duplicate alkyl substituents.
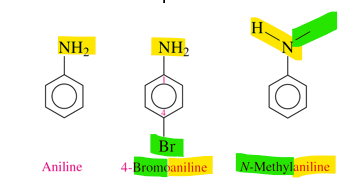
what do we name an aromatic amine (IUPAC)
aniline
how do we name aromatic amines
- amine of benzene is called aniline by IUPAc
-alkyl groups on the N atom use the prefix N- with the alkyl name
how can we classify amines
primary, secondary, and tertiary
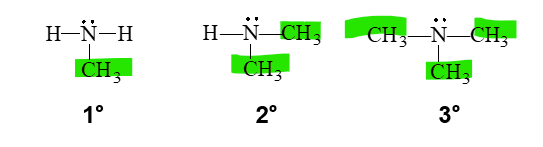
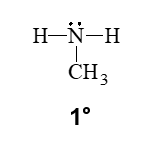
a primary amine
•A primary (1°) amine has one carbon group bonded to the nitrogen atom.
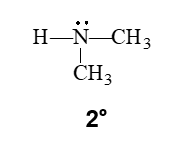
secondary amine
A secondary (2°) amine has two carbon groups bonded to the nitrogen atom.
tertiary amine
•A tertiary (3°) amine has three carbon groups bonded to the nitrogen atom.
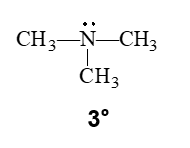
drawing line angle formula for amines
the N is shown
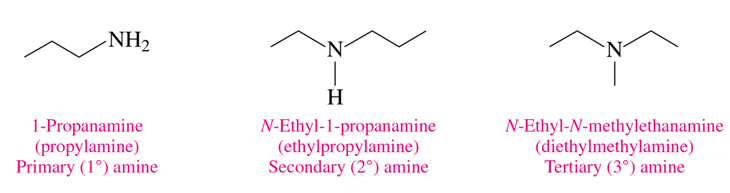
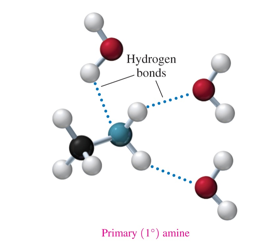
what amines can form hydrogen bonds with water
primary and secondary and tertiary
what amines can form hydrogen bonds with each other
ONLY PRIMARY AND SECONDARY
why cant tertiary amines form hydrogen bonds with each other
no free H
N compared to O (electronegativity)
•Nitrogen is not as electronegative as oxygen, so hydrogen bonds in amines are weaker than the hydrogen bonds in alcohols.
hydrogen bonds of amines compared to alcohols
weaker because N is not as electroneg as O
boiling point of amines
•Amines have boiling points that are higher than those of alkanes but lower than those of alcohols.
•Primary (1°) amines can form more hydrogen bonds and have higher boiling points than secondary (2°) amines of the same mass.
•Tertiary (3°) amines cannot form hydrogen bonds with each other and have lower boiling points than primary or secondary amines of the same mass.
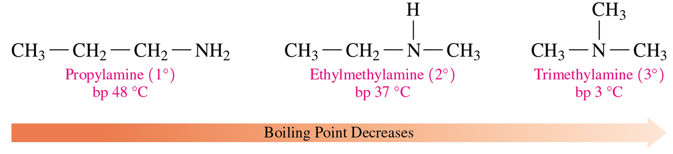
boiling point amines (primary vs secondary)
•Primary (1°) amines can form more hydrogen bonds and have higher boiling points than secondary (2°) amines of the same mass.
tertiary vs primary,secondary amines (bp)
•Tertiary (3°) amines cannot form hydrogen bonds with each other and have lower boiling points than primary or secondary amines of the same mass.
solubility of amines in water
•Amines that contain a polar N—H bond can form hydrogen bonds with water.
•Amines with one to six carbon atoms, including tertiary amines, are soluble in water.
•As the number of carbon atoms in an amine increases in the nonpolar alkyl portions, the effect of hydrogen bonding is diminished.
polarity of N-H bond
polar
allows for H-bonds with water to form
in water, amines react as
bases
how does an amine as a base in water
In amines, the N will accept an H+ from water, to make an ammonium ion (+ charged) and a hydroxide ion (OH-)
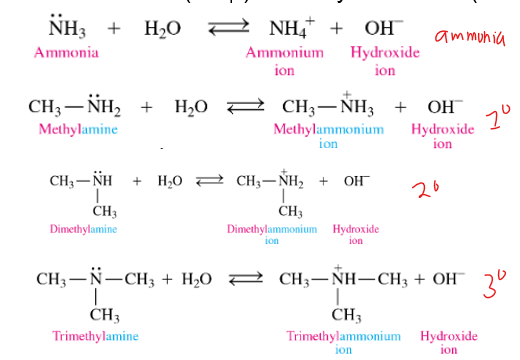
what are the reactants of amines in bases reaction
amine + water
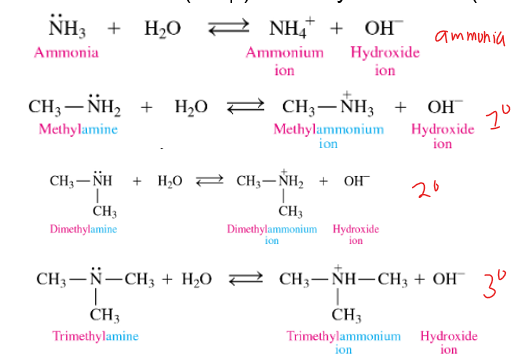
what are the products of amines in water reaction
ammonium ion and hydroxide ion
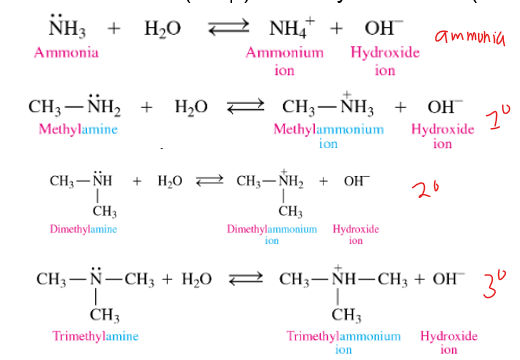
how do we name the amine product when amines are in water
-remove amide and add ammonium ion
-use common name rules for naming the c-chains
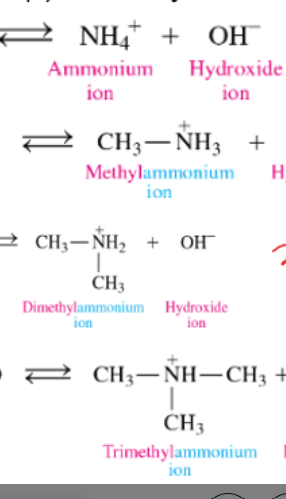
what is an amine salt and how do we name
-formed when amines react as a Brønsted–Lowry base in a neutralization reaction with citric acid.
-named by using its alkylammonium ion name (excluding “ion”) and adding the name of the negative ion at the end
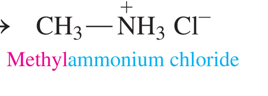
neutralization reaction of amines
-an amine acts as a base and reacts with an acid to form an ammonium salt
-lone pair of electrons from the N atom accepts an H+ from an acid to give ammonium salt
-NO WATER IS FORMED
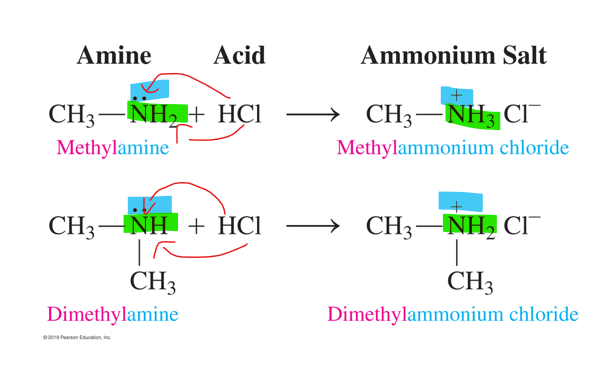
reactants in neutralization of amines
amine + acid (HCl)
products of neutralization of amines
ammonium salt
-NO WATER FORMED
quaternary ammonium salt
•a nitrogen atom is bonded to four carbon groups, which classifies it as a quaternary (4°) amine.
•the nitrogen atom has a positive charge and is not bonded to an H atom.
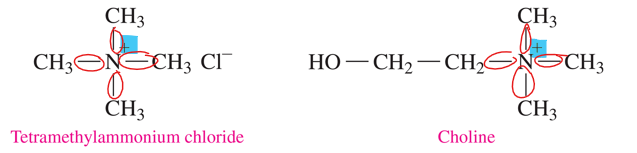
why would a N with 4 bonds be +
it has a lone pair of electron when has 3 bonds, but that is when it is happy. SO adding another bond makes it positive
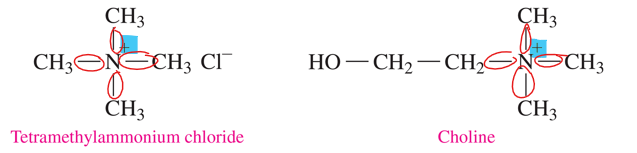
ammonium salt (polarity, state at room temp, odor, solubility)
•ionic compounds with strong attractions between the positively charged ammonium ion and an anion, usually chloride.
•solids at room temperature, odorless, and soluble in water and body fluids.
what is a heterocyclic amine
A heterocyclic amine is a cyclic organic compound that consists of a ring of five or six atoms, of which one or two are nitrogen atoms.
heterocyclic amines are either ___ or ____ rings
5 or 6 atom rings
5 atom rings (heterocyclic amines)
•the simplest one is pyrrolidine, which is a ring of four carbon atoms and one nitrogen atom, all with single bonds.
•pyrrole is a five-atom ring with one nitrogen atom and two double bonds.
•imidazole is a five-atom ring that contains two nitrogen atoms.
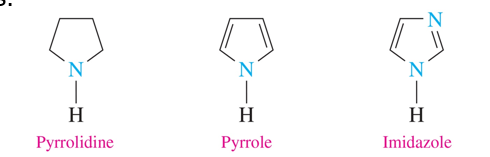
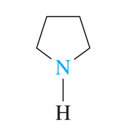
pyrrolidine
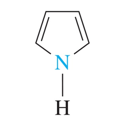
pyrrole
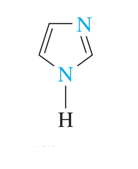
imidazole
6-atom rings (heterocyclic amines)
piperidine, pyridine, pyrimidine, purine
piperidine is found in pepper
purine and pyramidine is found in DNA and RNA
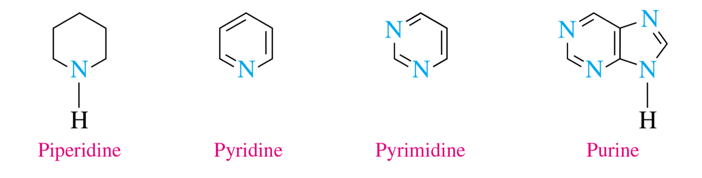
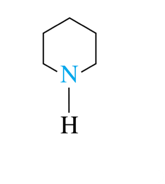
piperidine
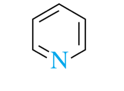
pyridine
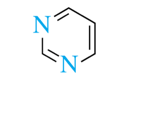
pyrimidine
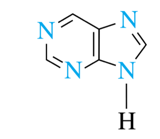
purine (a 6-atom with 5-atom ring)
what are alkaloids
•physiologically active compounds produced by plants that contain heterocyclic amines.
•used in anesthetics, in antidepressants, and as stimulants, and many are habit forming.
Include caffeine, heroin, morphine, codeine
what is a neurotransmitter
A neurotransmitter is a chemical compound that transmits an impulse from a nerve cell to a target cell.
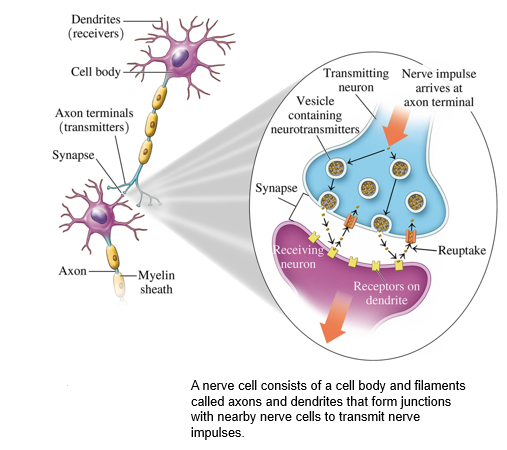
how process works
also known as a nerve cell, consists of
•a cell body and numerous filaments called dendrites at one end.
•an axon that ends at the axon terminal at the opposite end.
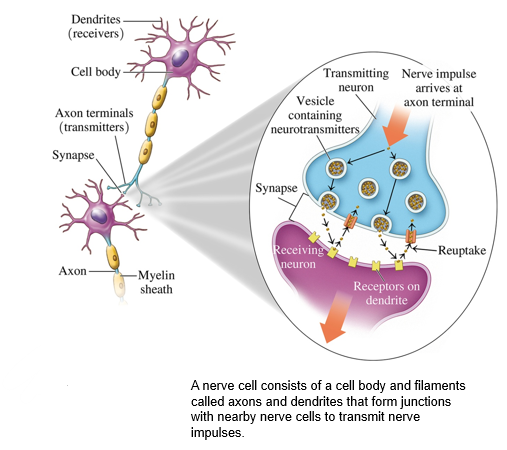
what is a synapse
•junctions called synapses formed between the axon terminals and dendrites of other nerve cells.
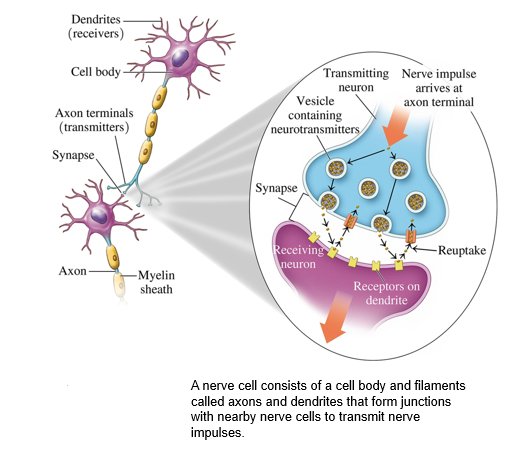
where are neurotransmitters made and stored
Within a nerve cell, neurotransmitters are synthesized and stored in vesicles at the end of the axon terminal.
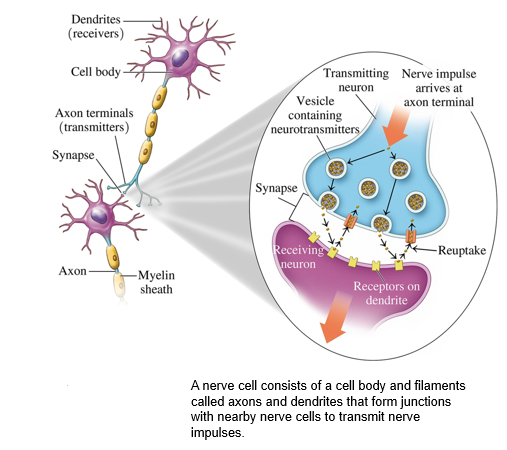
what are the 2 kinds of neurotransmitters
excitatory and inhibitory
what do excitatory neurotransmitters do
stimulate the receptors to send more impulses
what do inhibitory neurotransmitters do
•decrease the activity of the receptors.
when excitatory neurotransmitters bind, what happens to ion channels
The binding of an excitatory neurotransmitter opens ion channels in nearby nerve cells, releasing more neurotransmitters.
neurotransmitters contain what
nitrogen atoms as amines and alkylammonium ions
how are neurotransmitters obtained
•they are synthesized from compounds such as amino acids obtained from our diets.
•their amino groups are usually ionized, forming ammonium cations and carboxylate anions.
what are the important neurotransmitters (8)
acetylcholine, dopamine, norepinephrine (noradrenaline), epinephrine (adrenaline), serotonin, histamine, glutamate, and GABA.
catecholamines are synthesized from
the amino acid tyrosine after it is converted to L-Dopa
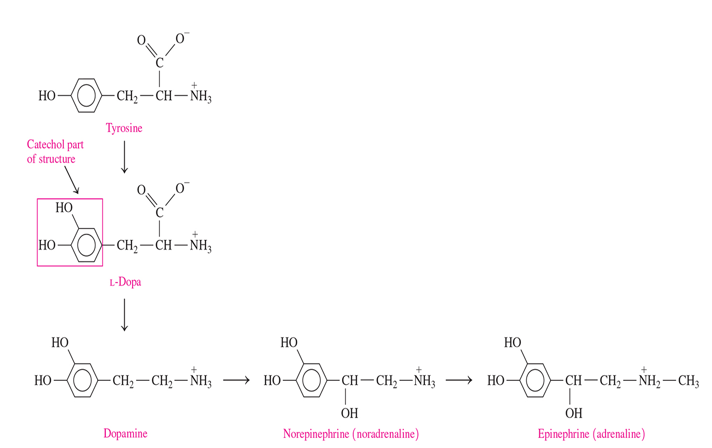
dopamine, norepinephrine and epinephrine made from what
tyrosine—> l dopa —> dopamine —> norepinephrine —> epinephrine
histamine made from what
•synthesized in nerve cells in the hypothalamus from the amino acid histidine.
glutamate
•is the most abundant neurotransmitter in the nervous system.
GABA made from
glutamate, an amino acid
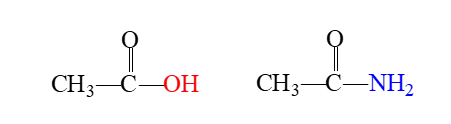
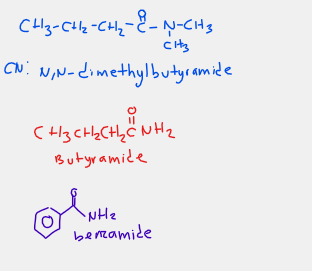
what is an amide
Amides are derivatives of carboxylic acids in which a nitrogen group (—NH2) of a primary or secondary amine replaces the hydroxyl (—OH) group of carboxylic acids.
how are amides made
carboxylic acids where a nitrogen group of a primary or secondary amine replaces the hydroxyl group of the carboxylic acid
-a carboxylic acid + amine
amidation
amides are produced
•in a reaction called amidation or condensation.
•when a carboxylic acid reacts with ammonia or a primary or secondary amine and heat
-Amide production is accompanied by the production of water.
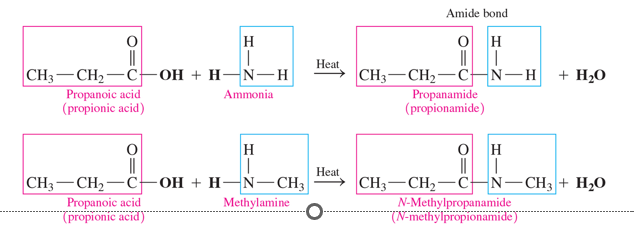
reactants in amidation
carboxylic acid and ammonia or amine
products in amidation
amide and water
how do we name amides (IUPAC)
amides are named by dropping the oic acid of carboxylic acid and adding suffix amide
-Alkyl groups attached to the nitrogen of an amide are named with the prefix N followed by the alkyl name.
how do we name amides (Common name)
amides are names by dropping ic acid of carboxylic acid and adding suffix amide
-Alkyl groups attached to the nitrogen of an amide are named with the prefix N followed by the alkyl name.
amides state in room temp
-methanamide is liquid
-all other amides are solids
amides melting point
•Primary (1°) amides have the highest melting points because the —NH2 group can form the most hydrogen bonds.
•Melting points of the secondary (2°) amides are lower because they form fewer hydrogen bonds.
•Tertiary (3°) amides cannot form hydrogen bonds with each other; they have the lowest melting points
primary amides mp
•Primary (1°) amides have the highest melting points because the —NH2 group can form the most hydrogen bonds.
secondary amide mp
•Melting points of the secondary (2°) amides are lower because they form fewer hydrogen bonds.
tertiary amide mp
•Tertiary (3°) amides cannot form hydrogen bonds; they have the lowest melting points (see Table 18.2).
amides and hydrogen bonds
-primary form the most h-bonds
-secondary form less H-bonds
-tertiary form no H-bonds
primary amides form H-bonds with
-NH2 of other amides
-water
secondary amides can form H bonds with
-less hydrogen bonds with other amides than primary
-form hydrogen bonds with water molecules
tertiary amides form H-bonds with
-only form hydrogen bonds with water
-only 1 H-bond
solubility of amides
-primary soluble up to 5 carbons (H-bonds)
-secondary soluble up to 5 carbons (H-bonds)
-tertiary soluble (only form 1 H bond)
acid hydrolysis of amides
Amides undergo acid hydrolysis with heat to produce a carboxylic acid and an ammonium salt.
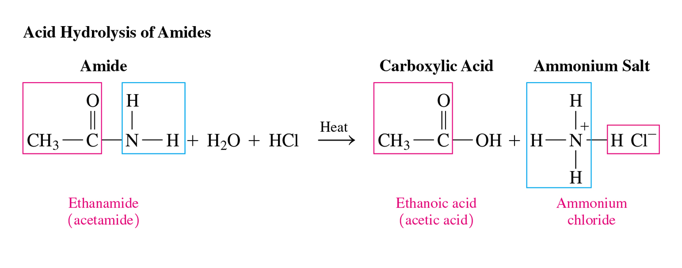
reactants of acid hydrolysis of amides
amide and water and acid
products of acid hydrolysis of amides
carboxylic acid and ammonium salt
base hydrolysis of amides
Amides undergo base hydrolysis with heat to produce a carboxylate salt and an amine or ammonia.
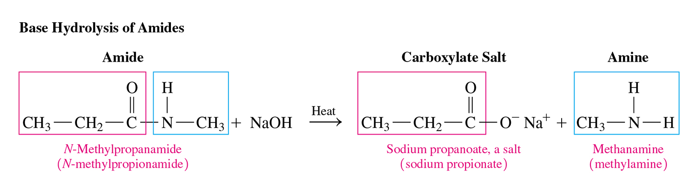
reactants of base hydrolysis
amide and strong base
products of base hydrolysis
carboxylate salt and amine
amine and amide reaction summary
AMINES
-dissociation in water: amine + water—> ammonium ion + hydroxide ion
-neutralization: amine + strong acid (HCl)—> ammonium salt
AMIDES
-amidation: carboxylic acid + amine → amide + water
-acid hydrolysis: amide + water + acid (HCl)→ carboxylic acid + ammonium salt
-base hydrolysis: amide + strong base (NaOH or KOH) → carboxylate salt + amine (or ammonia)