Some Families of Organic Compounds
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
When does tetrahedral geometry only occur?
Saturated compounds with four single bonds
Name two types of compounds that are tetrahedral?
Chloroalkanes. Alcohols
Chloroalkane (definition)
A compound in which one or more of the hydrogen atoms in an alkane have been replaced by chlorine atoms
Draw chloromethane
…
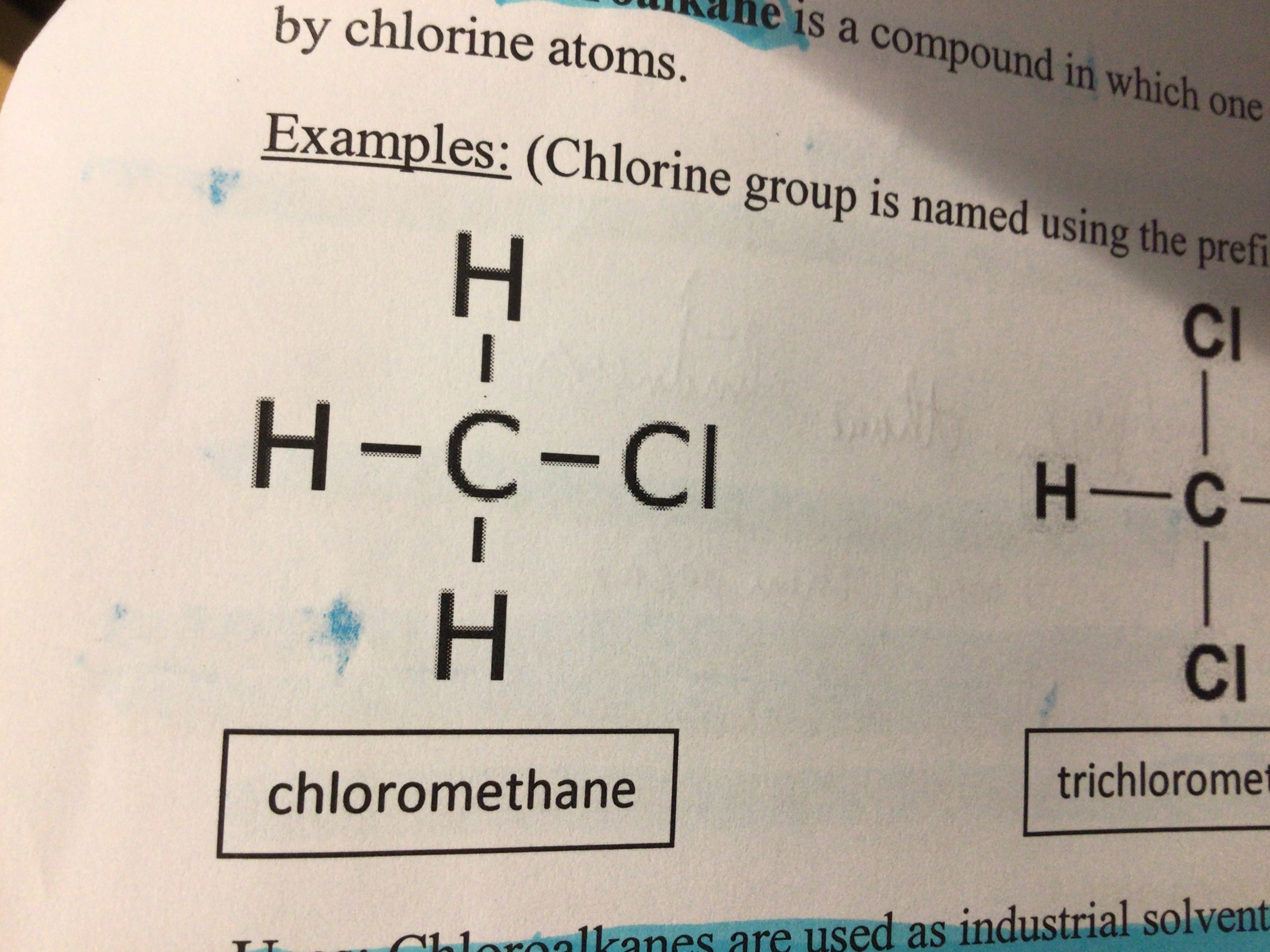
Draw trichloromethane
…
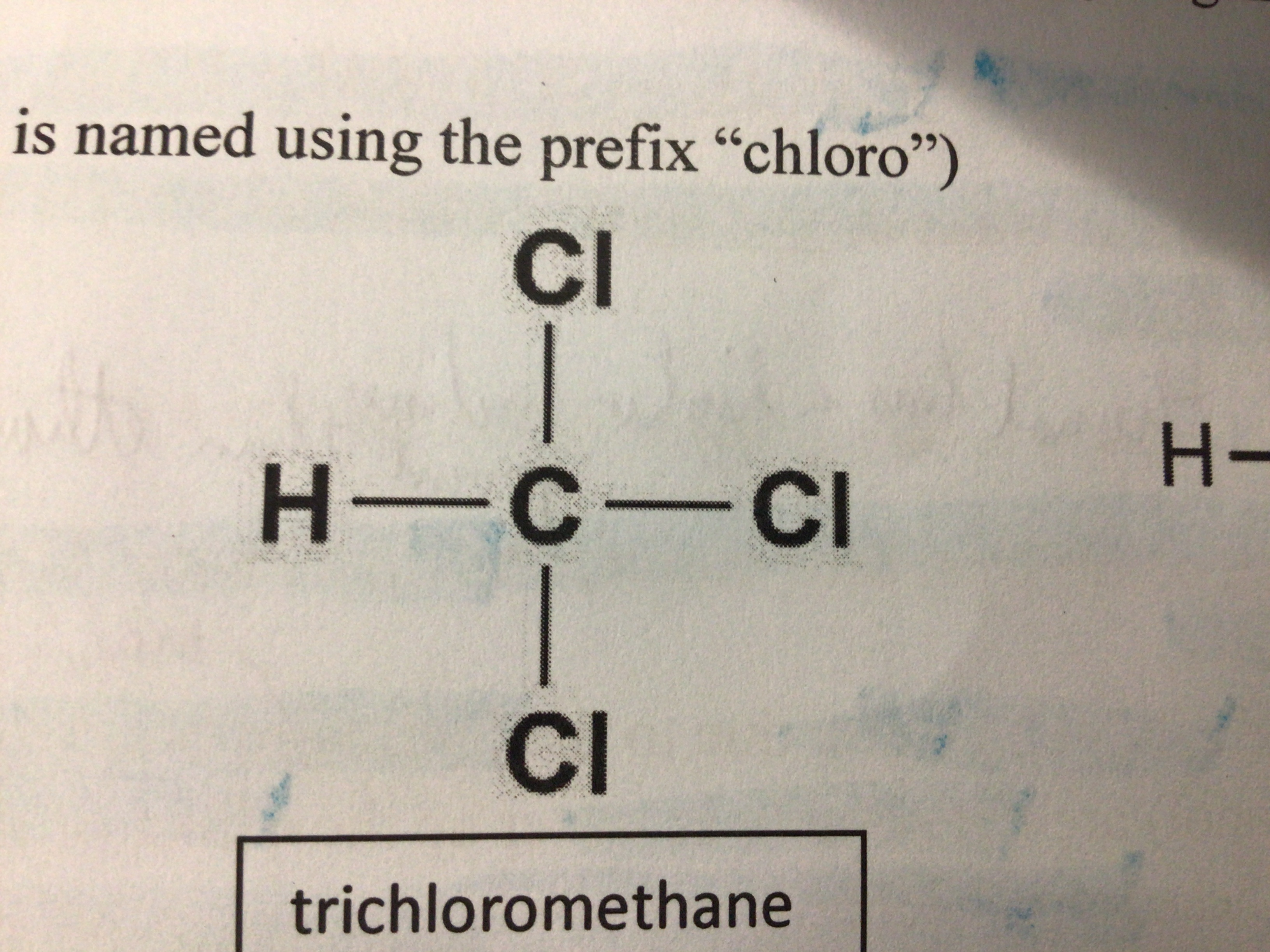
Draw 1,2-dichloropropane
…
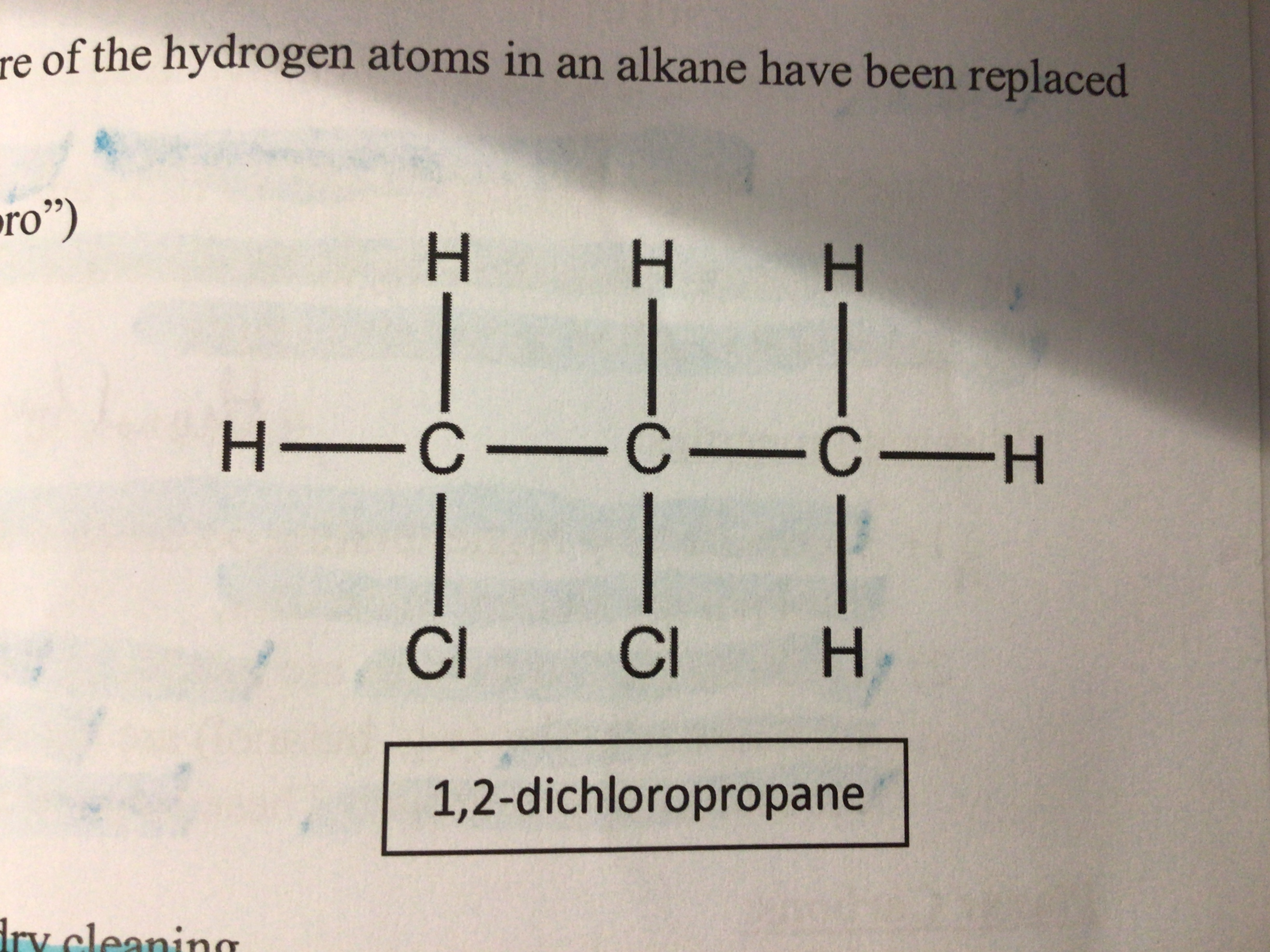
Uses of chloroalkanes
Industrial solvents e.g. dry cleaning
In what solvents are chloroalkanes soluble in?
Non polar solvents
What form are chloroalkanes in at room temperature?
Liquid
Functional group (definition)
An atom or group of atoms which are responsible for the characteristic properties of an organic compound or a series of organic compounds
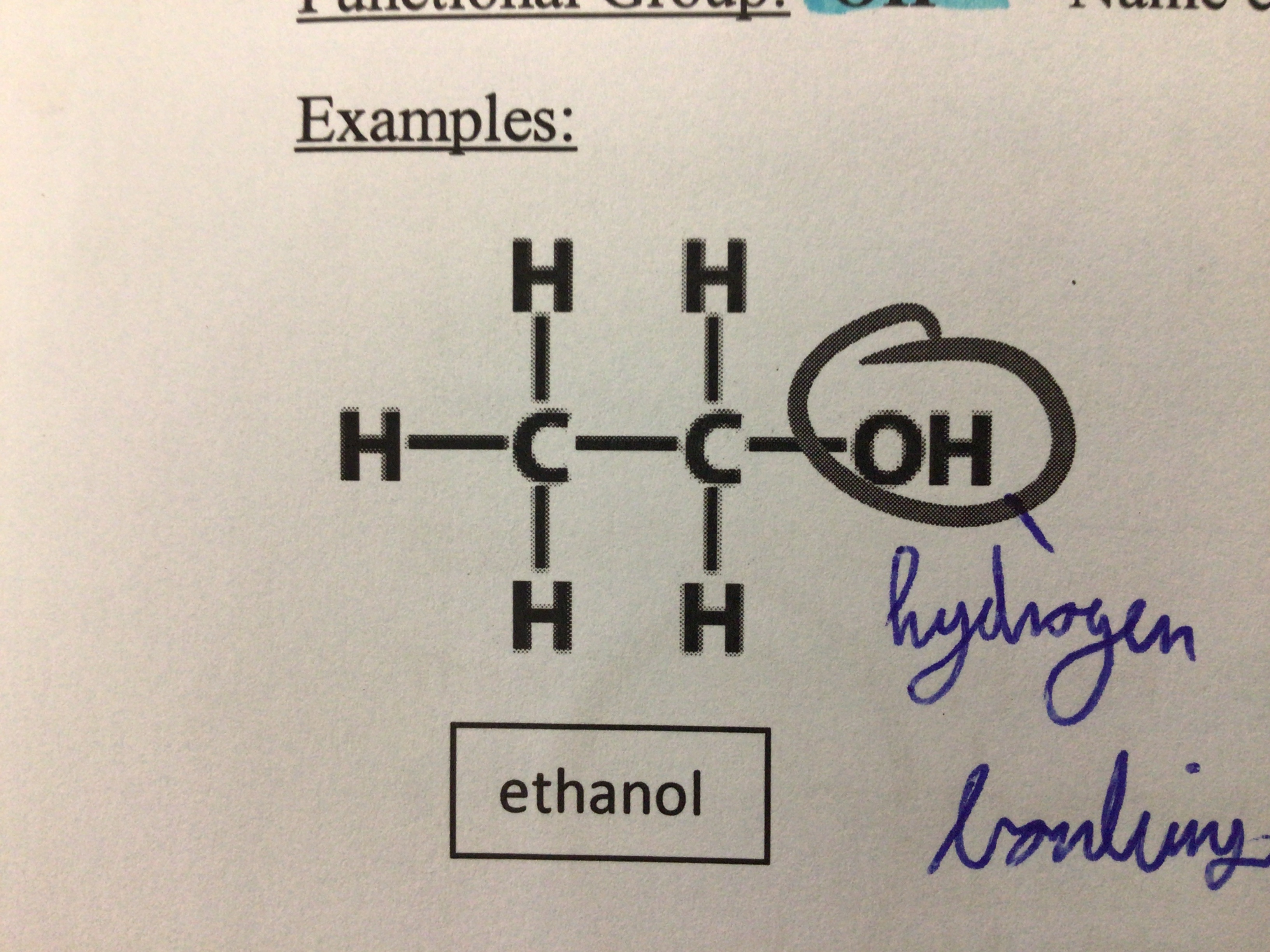
Functional group of alcohols
OH
What kind of bonding is between O and H?
Hydrogen bonding
Draw ethanol
…
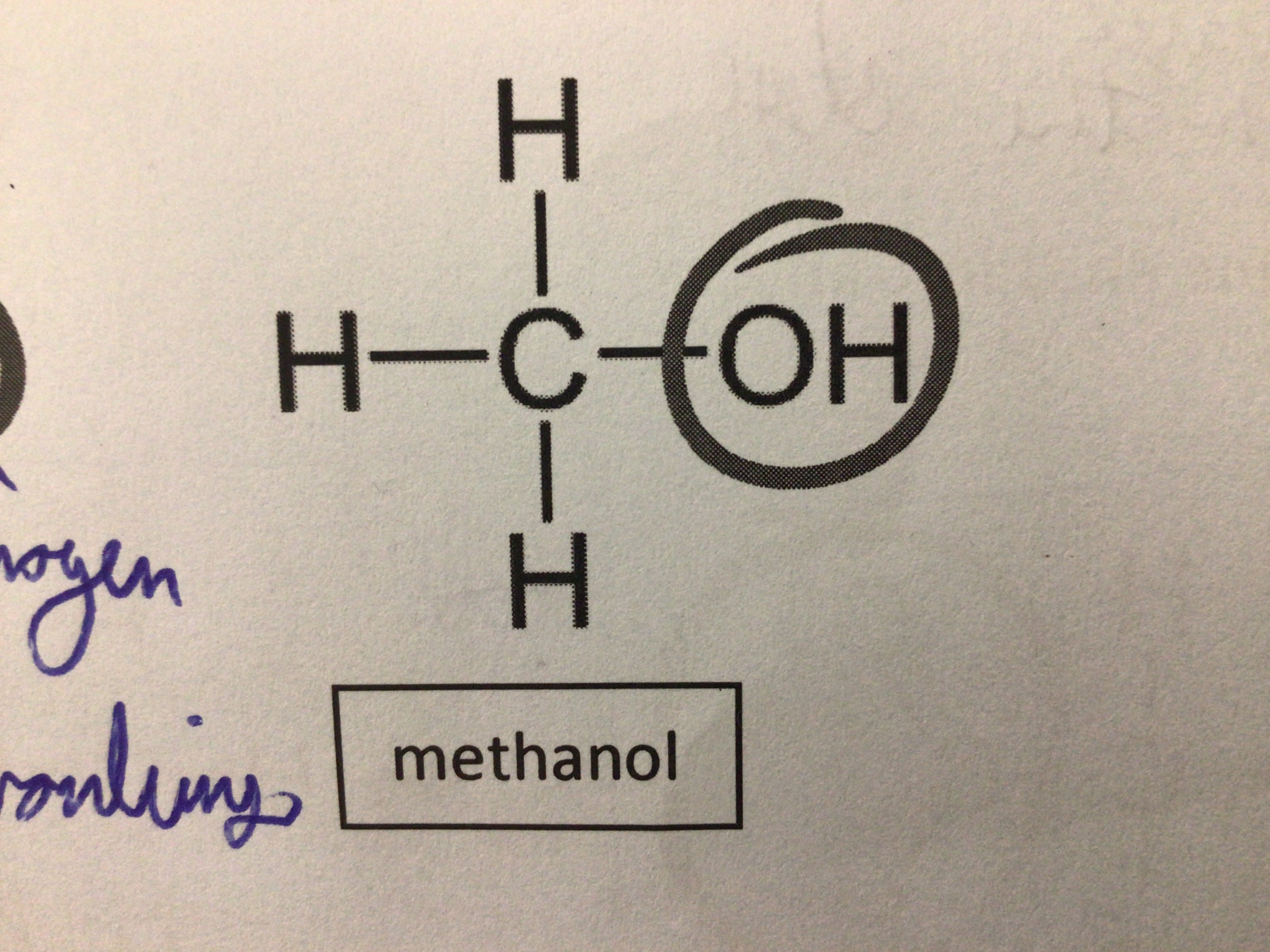
Draw methanol
…
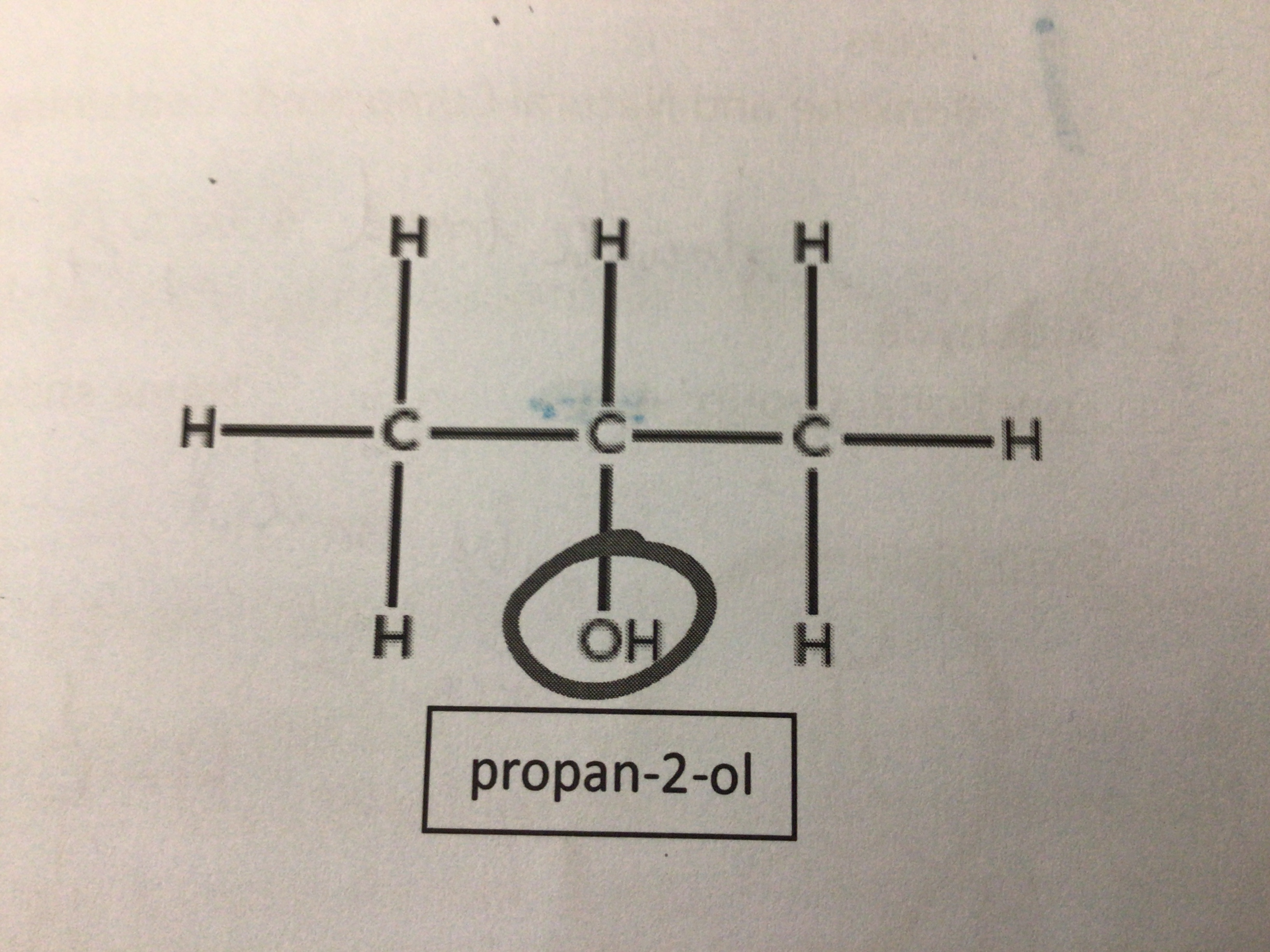
Draw propan-2-ol
…
Primary alcohol (definition)
An alcohol where the carbon atom bonded to the OH group is bonded to 1 other carbon
Draw a primary alcohol
…
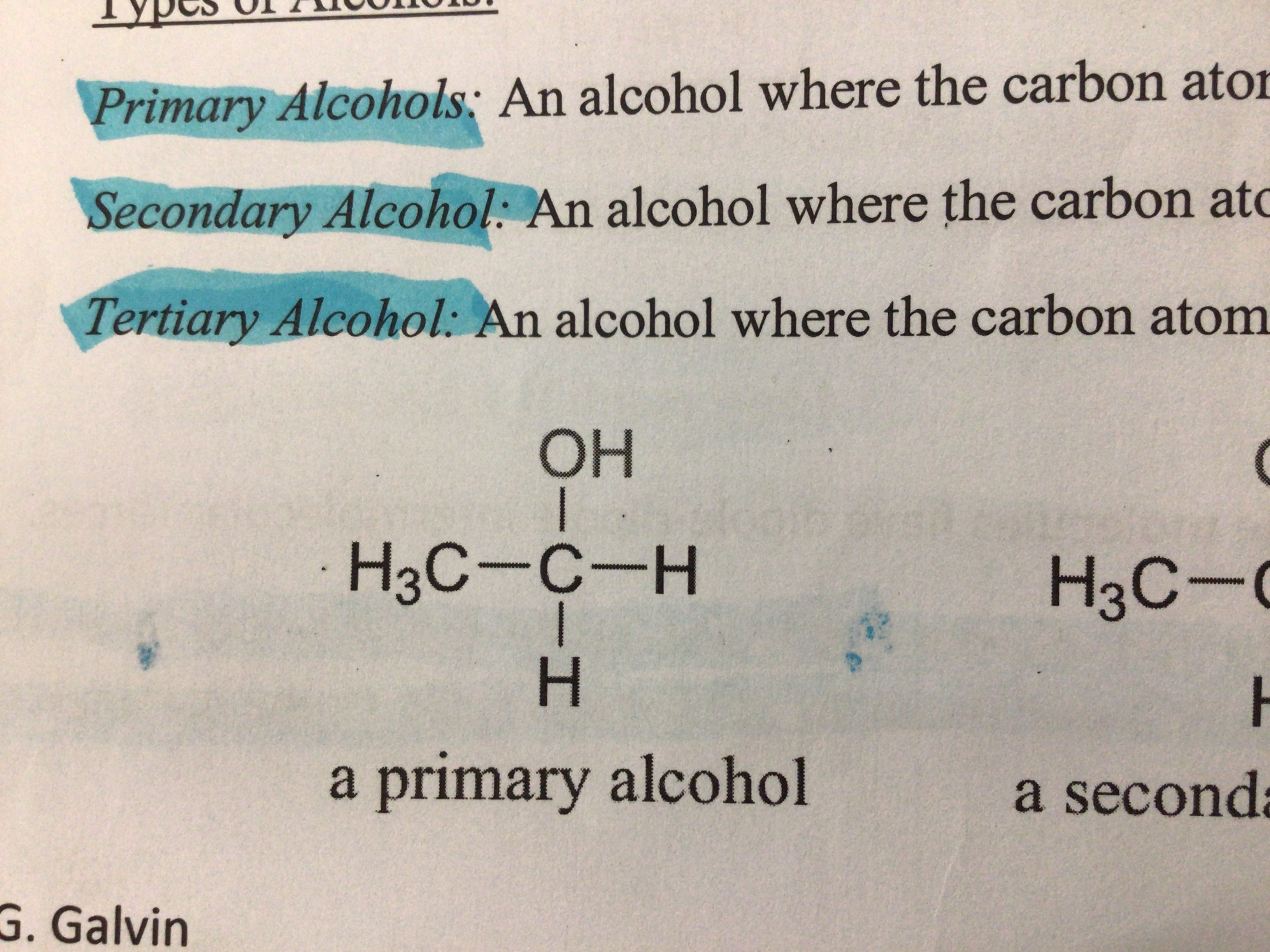
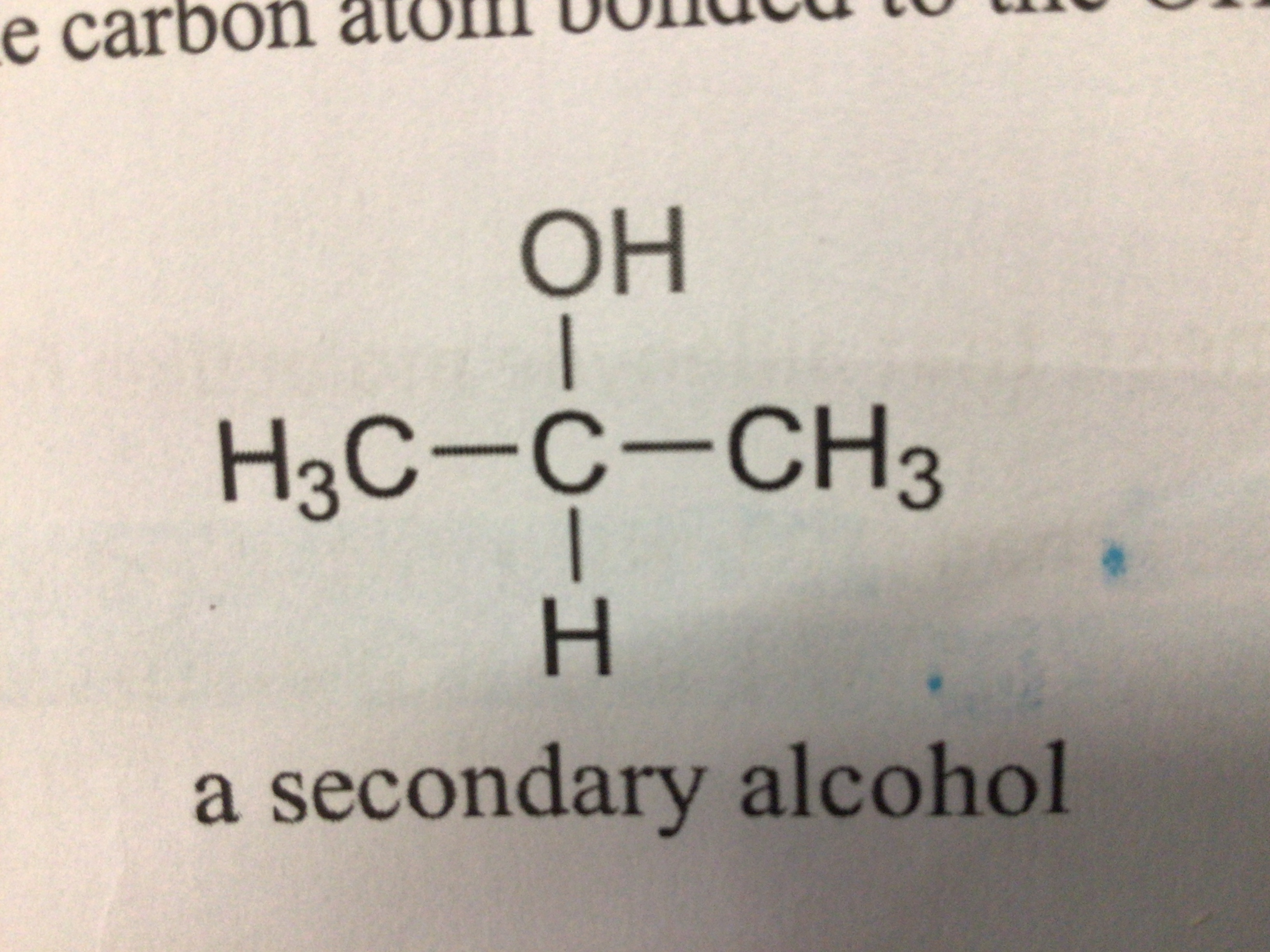
Secondary alcohol (definition)
An alcohol where the carbon atom bonded to the OH group is bonded to 2 other carbons
Draw a secondary alcohol
…
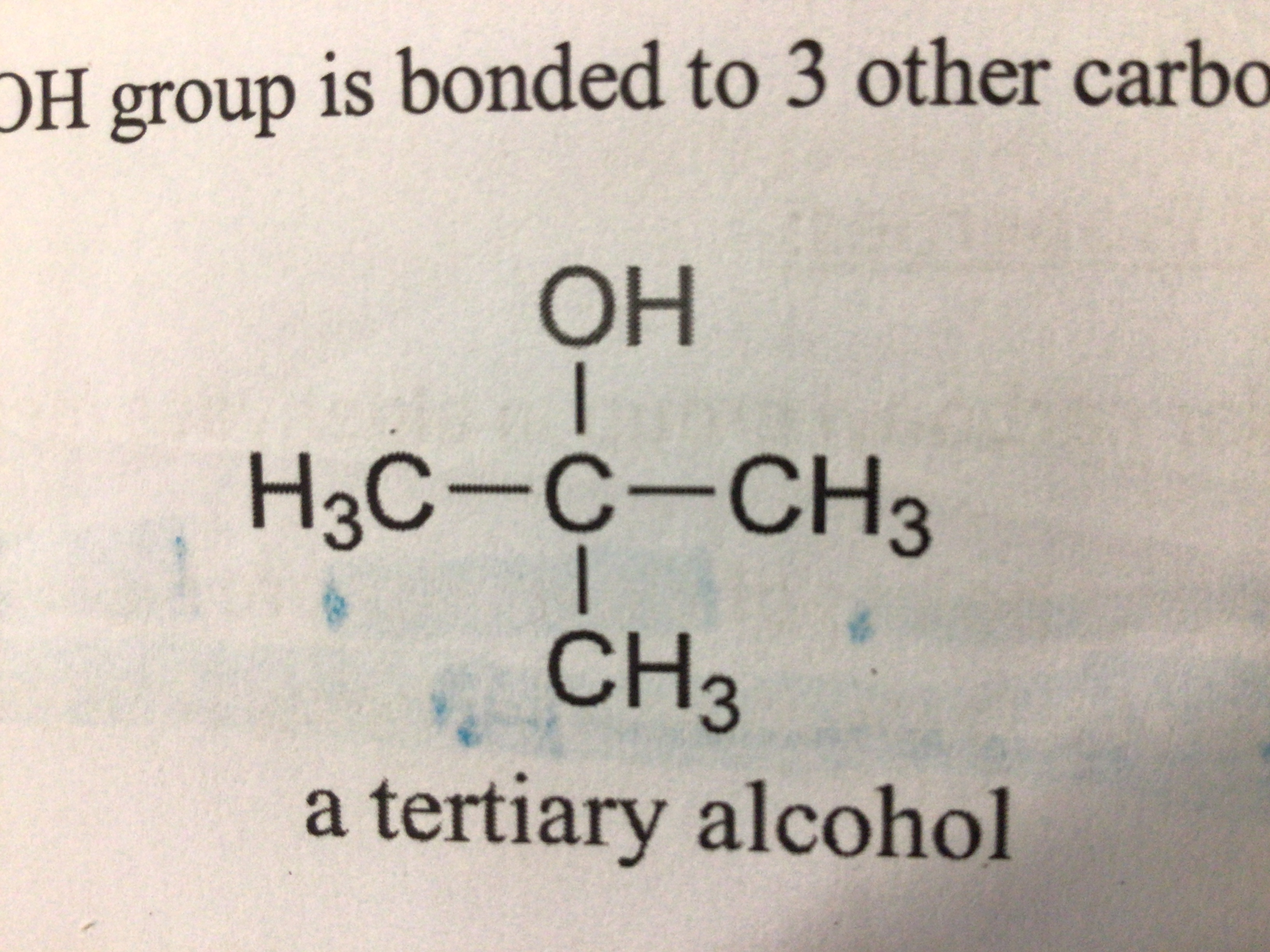
Tertiary alcohol (definition)
An alcohol where the carbon atom bonded to the OH group is bonded to 3 other carbons
Draw a tertiary alcohol
…
Use of ethanol/alcohols
Found in alcoholic drinks
How is ethanol made?
Fermenting glucose using yeast
Equation for fermentation
C6H12O6 → 2CH5OH +2CO2
Do alcohols have higher or lower boiling points than their corresponding alkanes e.g. ethanol and ethane?
Higher
Why do alcohols have higher boiling points than their corresponding alkanes?
Due to the hydrogen bonding between the O and the H
What is added to prevent people from drinking industrial ethanol?
Methanol
Why does methanol prevent people from drinking industrial ethanol?
Methanol is toxic
What happens to ethanol after methanol is added and what’s it now called?
Becomes denatured and is known as methylated spirits
Are alcohols soluble in water?
Only small alcohol molecules. Larger ones are soluble on non-polar solve ts
Why are only small alcohol molecules soluble in water?
Hydrogen bonding between O and H. Effect of hydrogen bonding decreases as molecule gets larger
What is a planar carbon?
A carbon with planar geometry
In what type of molecules does planar geometry occur in?
Unsaturated
Name 5 types of planar compunds
Aldehydes. Ketones. Carboxylique acids. Esters. Benzene/natural compounds with benzene
Functional groups of aldehydes
CHO
What name ending are given to the compounds of Aldehydes?
“Al”
Where are double bonds located in aldehydes?
The edge
What part of aldehydes is polar
The double CO bond
What shape is the rest of the aldehyde (C-H)?
Tetrahedral
What is a carbonyl group?
C = O
Draw methanal
…
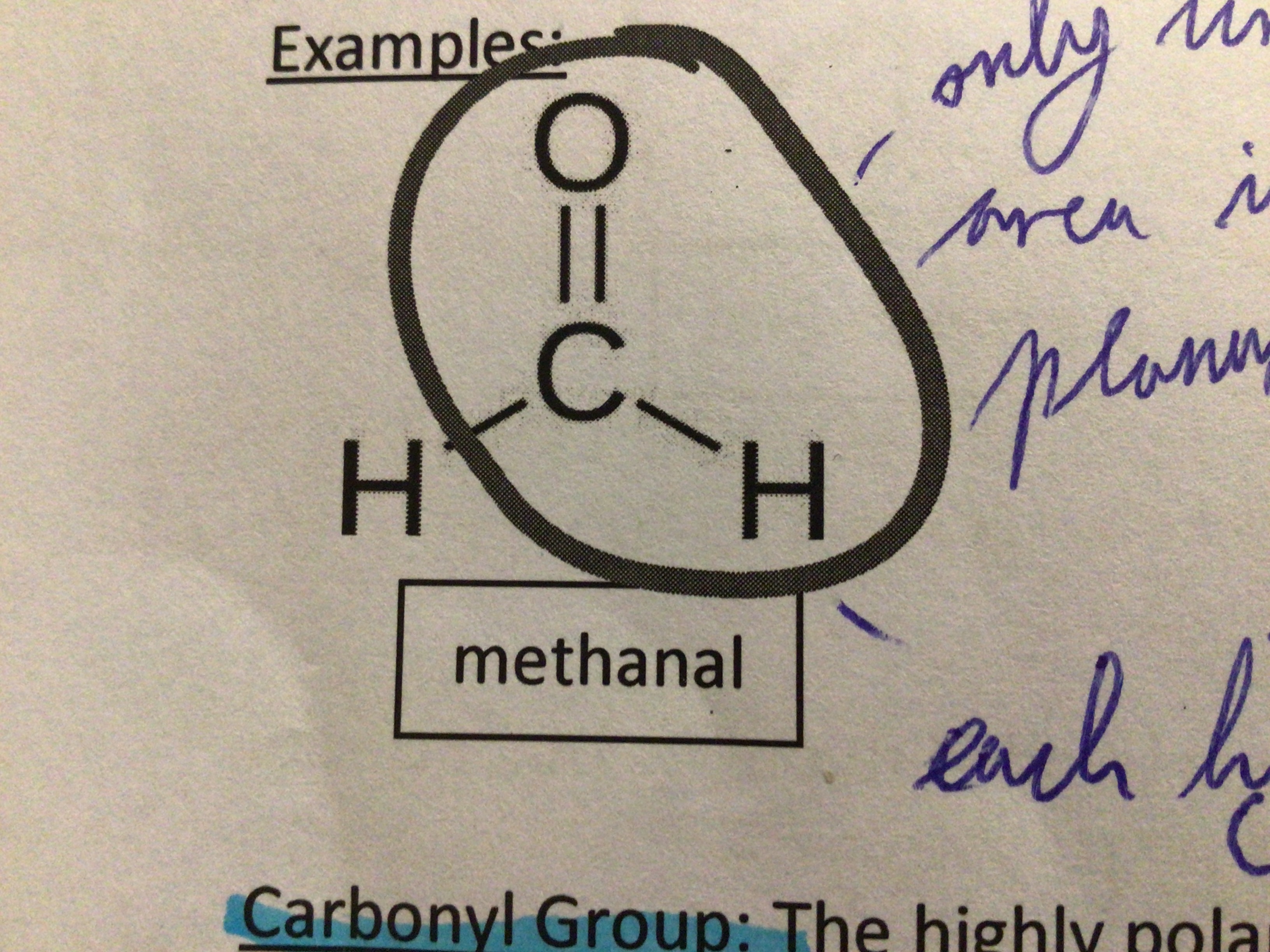
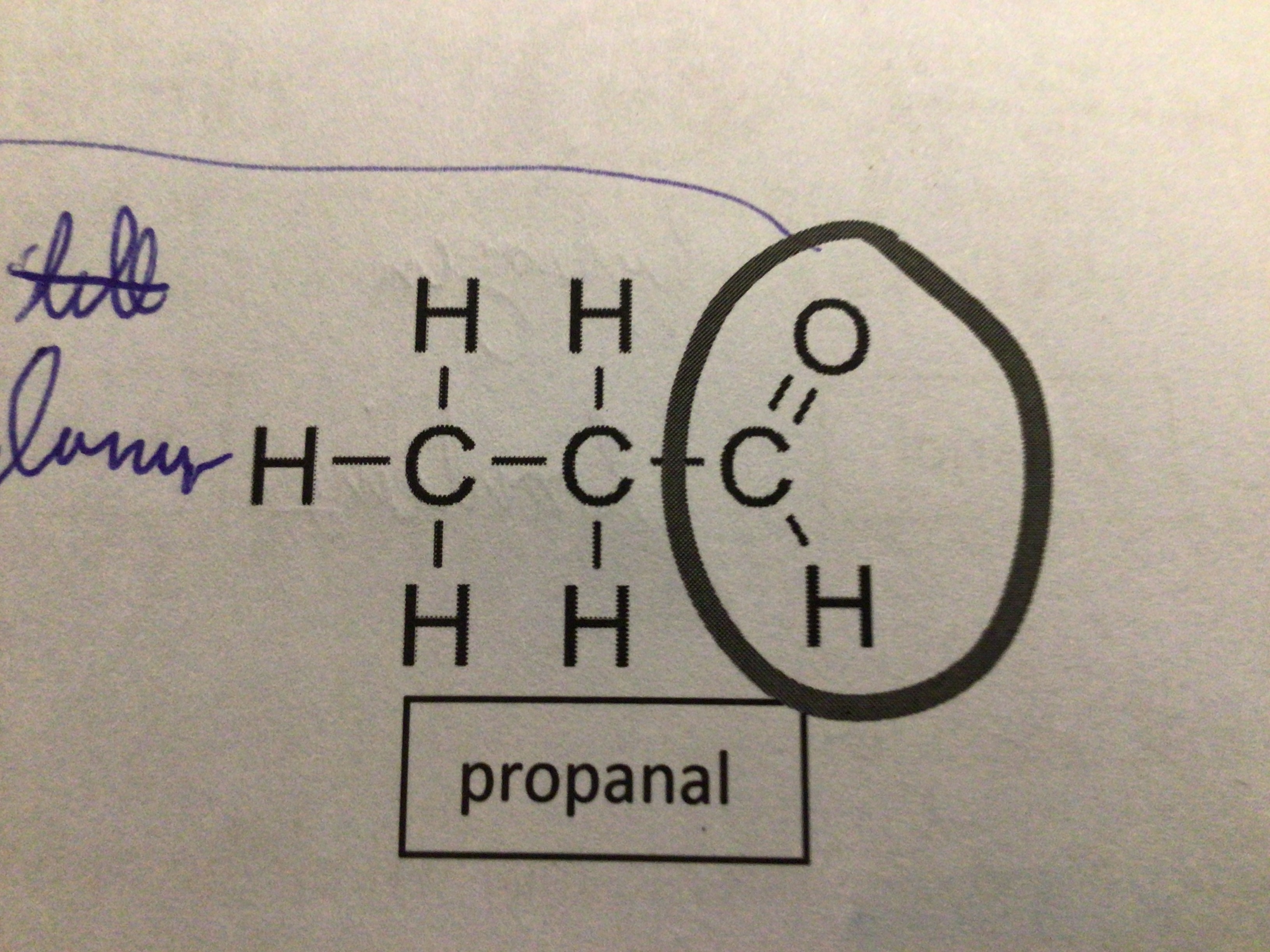
Draw propanal
…
Boiling points of aldehydes compared to alkanes
Boiling points of aldehydes are higher than the respective alkanes
Why do aldehydes have higher boiling points than their respective alkanes?
Dipole-dipole forces between the aldehyde molecules are stronger than weaker Van-der-Waals forces between alkane molecules
Boiling points of aldehydes compared to corresponding alcohols
Boiling points of aldehydes are lower than their corresponding alcohols
Why do aldehydes have higher boiling points than their respective alcohols?
Dipole-dipole forces in aldehydes are weaker than hydrogen bonding in alcohols
Are aldehydes soluble in water? Why?
Only small molecules are soluble because of the polar carbonyl group
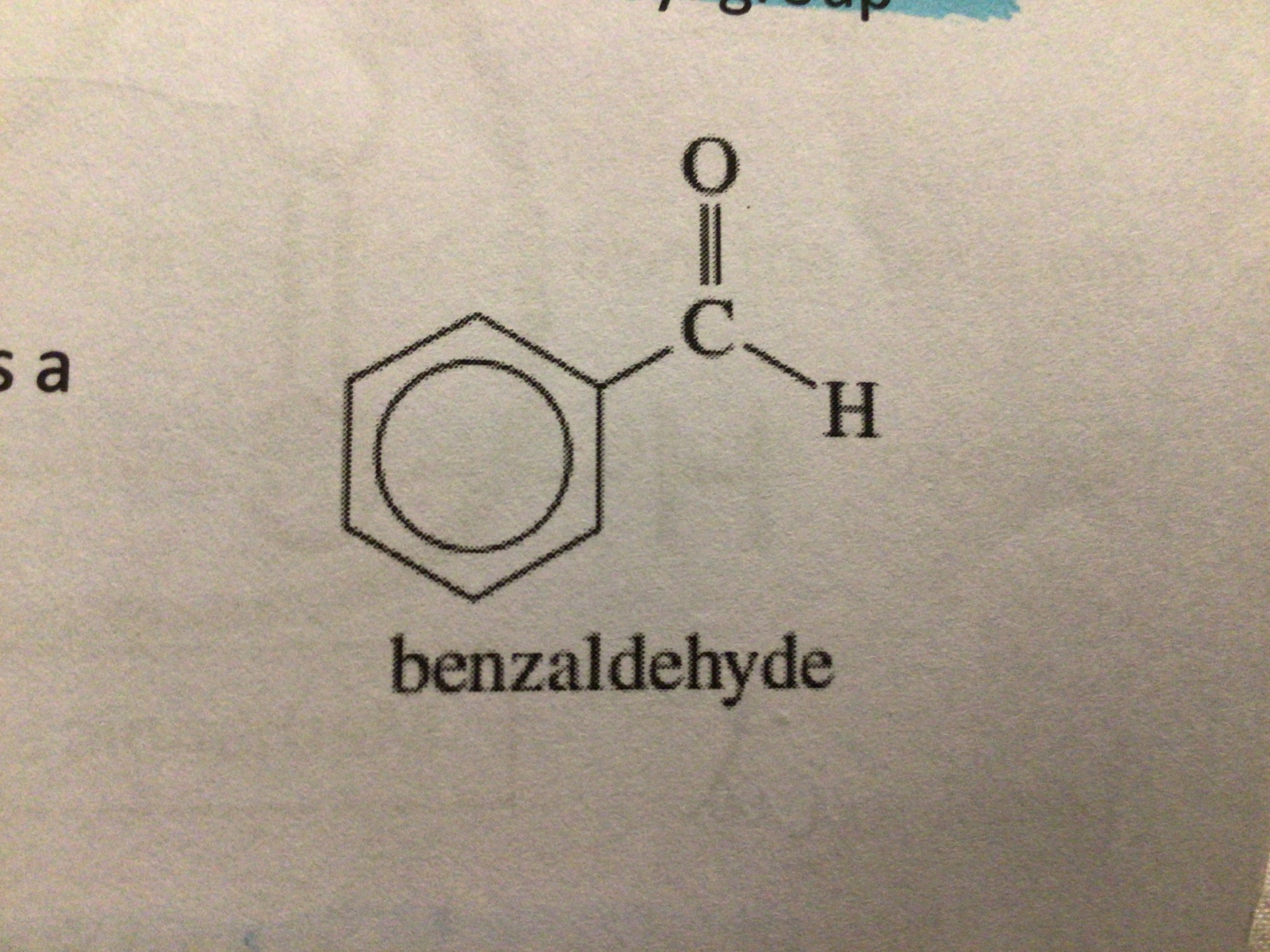
Name an aromatic aldehyde
Benzaldehyde
Draw benzaldehyde
…
What does benzaldehyde smell like?
Almonds
Where is benzaldehyde found?
Almond kernels
What is benzaldehyde used as?
Flavouring agent
Functional group of ketones
CO
Name ending for ketones
One
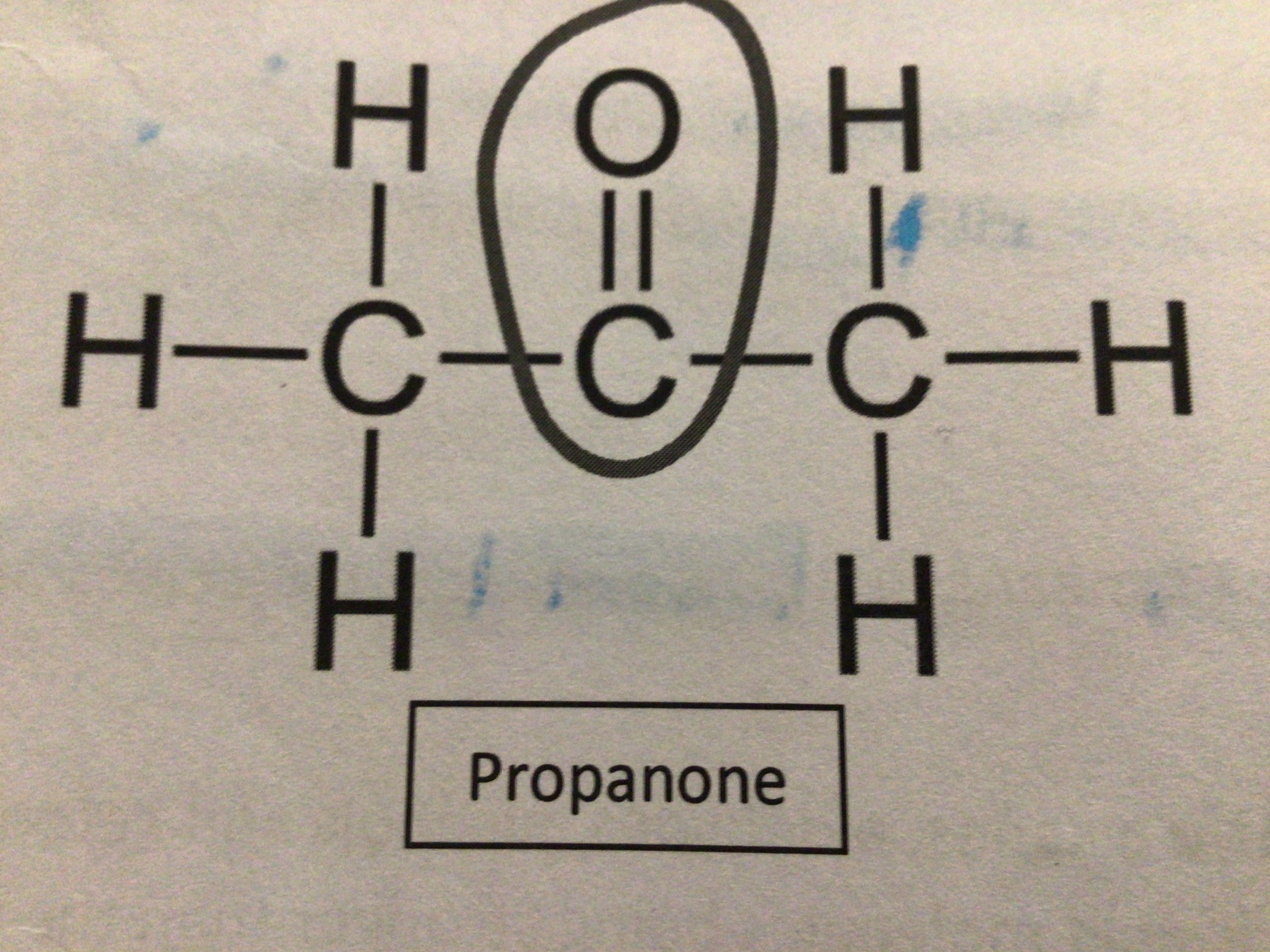
Draw propanone
…
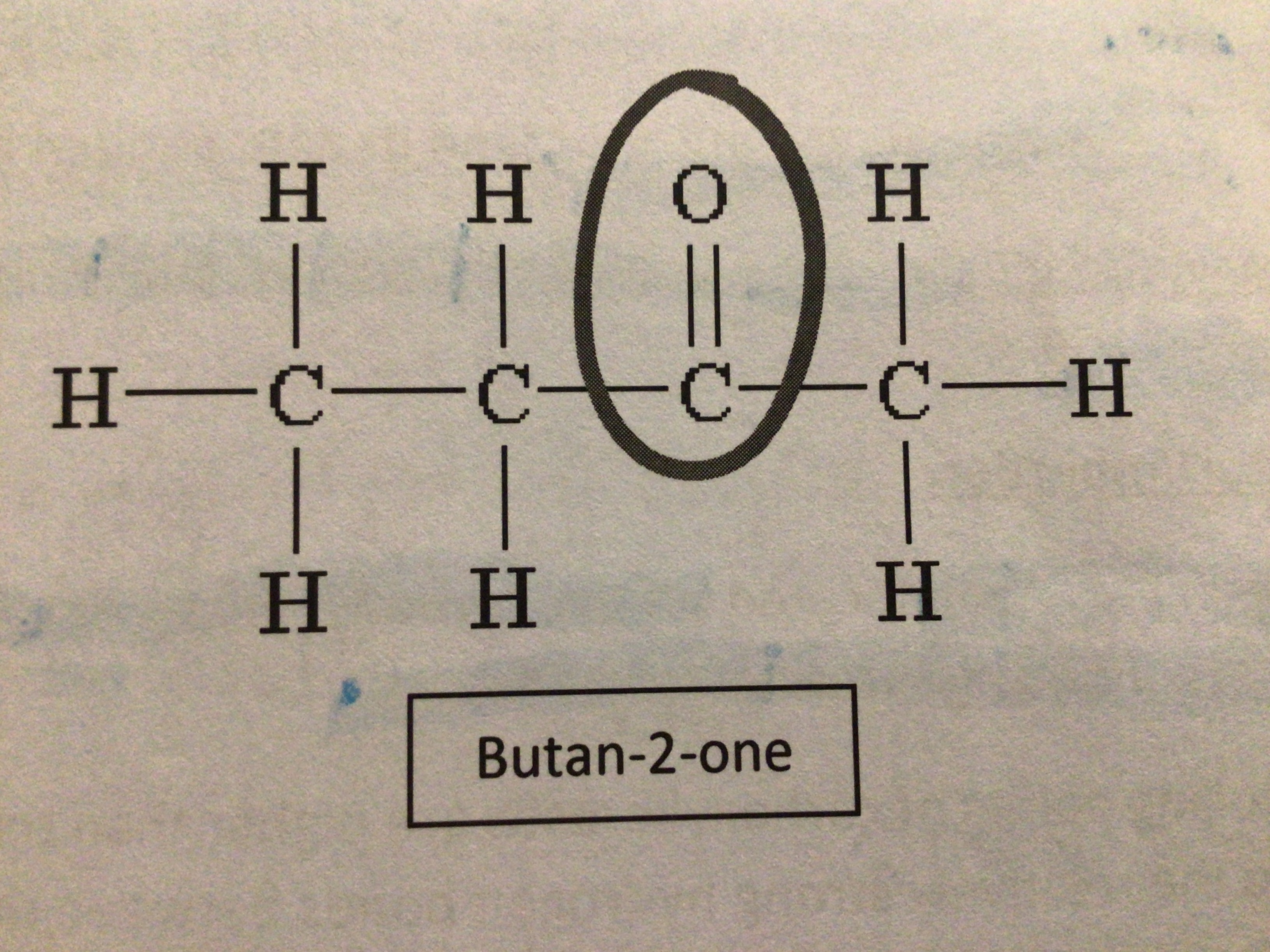
Draw butan-2-one
…
What is propanone also known as?
Acetone
What is propanone used for?
Nail polish remover
Where is the carbonyl group located on ketones?
On a central carbon
What kind of intermolecular forces do ketones have?
Dipole-dipole
Boiling points of ketones are … than their respective alkanes
Higher
Why are the boiling points of ketones higher than their corresponding alkanes?
Dipole-dipole forces (ketones) are stronger than the Van-der-Waals forces (alkanes)
Boiling points of ketones are … than their respective alcohols
Lower
Why are the boiling points of ketones lower than their corresponding alcohols?
Dipole-dipole forces (ketones) are weaker than hydrogen bonding (alcohols)
Are ketones soluble in water? Why?
Small molecules are soluble in water due to the polar carbonyl group. Larger the ketone molecule the less soluble due to the effect of the carbonyl group decreasing
Corresponding aldehydes and ketones are … (e.g. propanol and propanone)
Structural isomers
Carboxylic acid functional group
COOH
Name ending of carboxylic acids
oic acid
Where is the COOH located on carboxylic acids?
End of the molecule at the same carbon
Draw ethanoic acid?
…
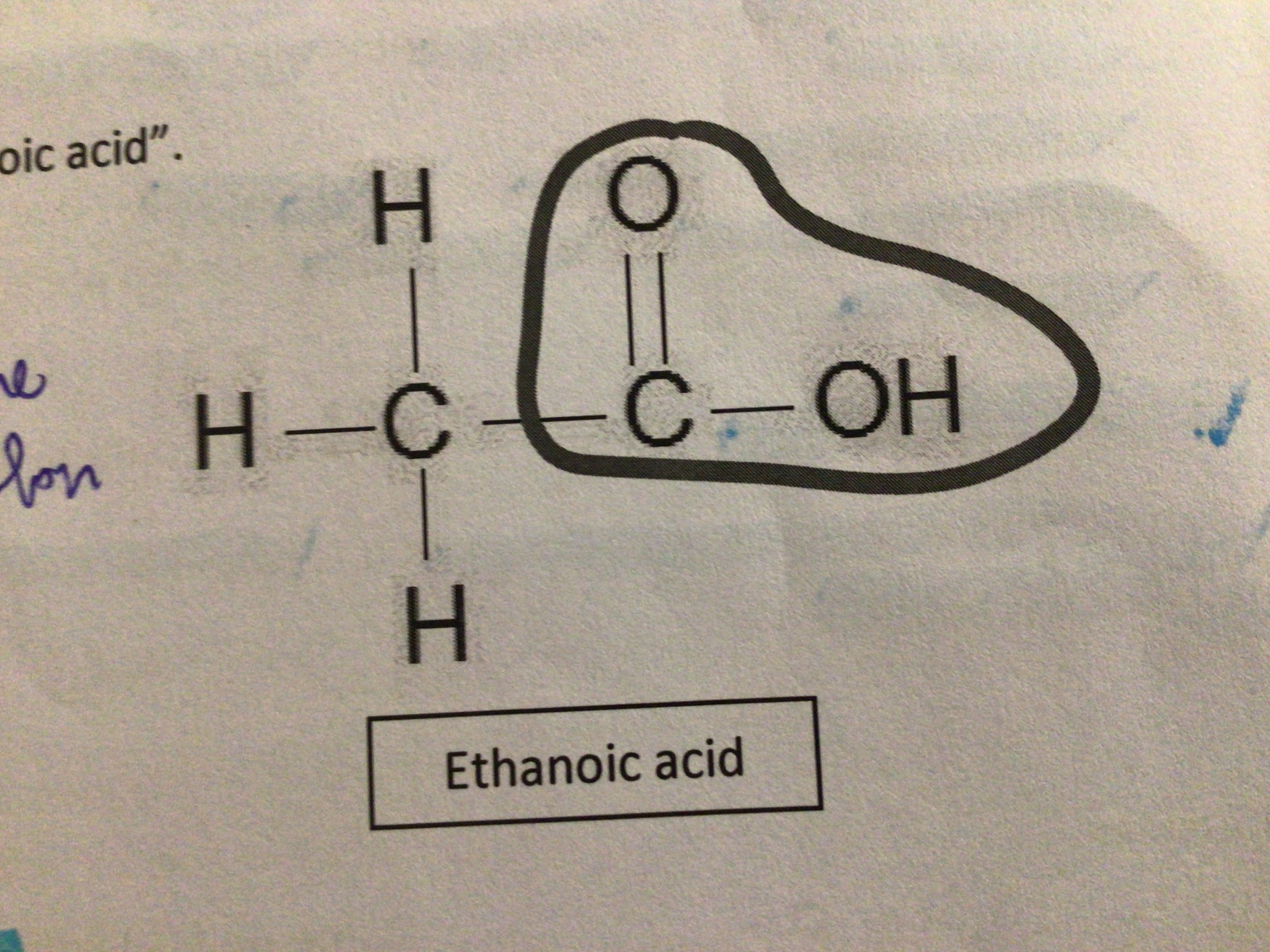
What acid is found in nettle and ant stings?
Methanoic/formic acid
What is ethanoic/acetic acid used for (2)?
Flavouring agent in vinegar. Makes cellulose acetate (camera film)
Use of propanone acid and benzoic acid?
Preservatives in food
What kind of bonding do carboxylic acid molecules have?
Hydrogen bonding
Boiling points off carboxylic acid are … than their respective alkanes, aldehydes, alcohols and ketones. Why?
Higher. Strong hydrogen bonding between the carboxylic acid molecules are stronger than the Van-der-Waals and Dipole-Dipole forces between the others
Are carboxylic acid molecules soluble in water? Why?
Small carboxylic molecules are soluble. Longer the carbon chain, the less soluble the carboxylic acid is in water due to the effect of the hydrogen bonding decreasing
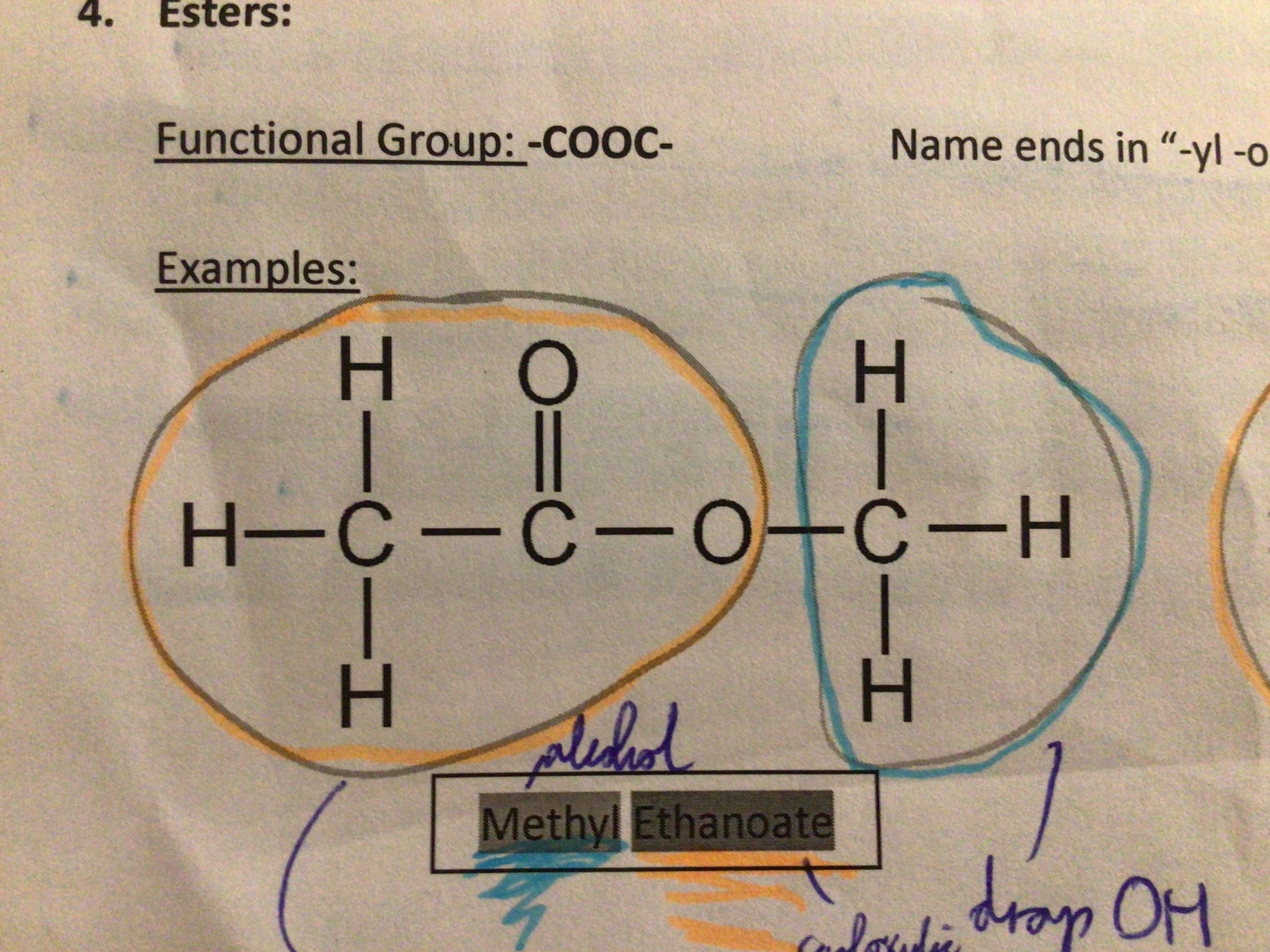
Functional group of esters
COOC
Name ending for esters
“-yl -oate”
How to get the chemical formula of an ester from an alcohol and a carboxylic acid?
Drop H from carboxylic acid. Drop OH from alcohol
How are esters formed?
Alcohol and carboxylic acid are reacted with sulphuric acid used as a catalyst
What kind of reaction is the formation of an ester?
Condensation reaction
Why is the formation of esters called a condensation reaction?
Loss of a water molecule
Process of naming an ester
Identify which part is from the carboxylic acid and which is from the alcohol. Name the section from the alcohol first. Name carboxylic acid and change the “oic acid” to ”oate”. Combine names.
What are esters used as?
Flavourings and fragrances because they have sweet, fruity scents
What kind of organic compounds are fats and oils?
Esters
What kind of intermolecular forces do esters have? Why?
Dipole-dipole due to the carbonyl group
Are esters soluble in water? Why?
Small esters are soluble because of the Dipole-Dipole forces. The longer the carbon chain, the less soluble due to a decrease of the effect of the C = O.
Are fats and oils soluble in water? Why?
Insoluble as they are large esters
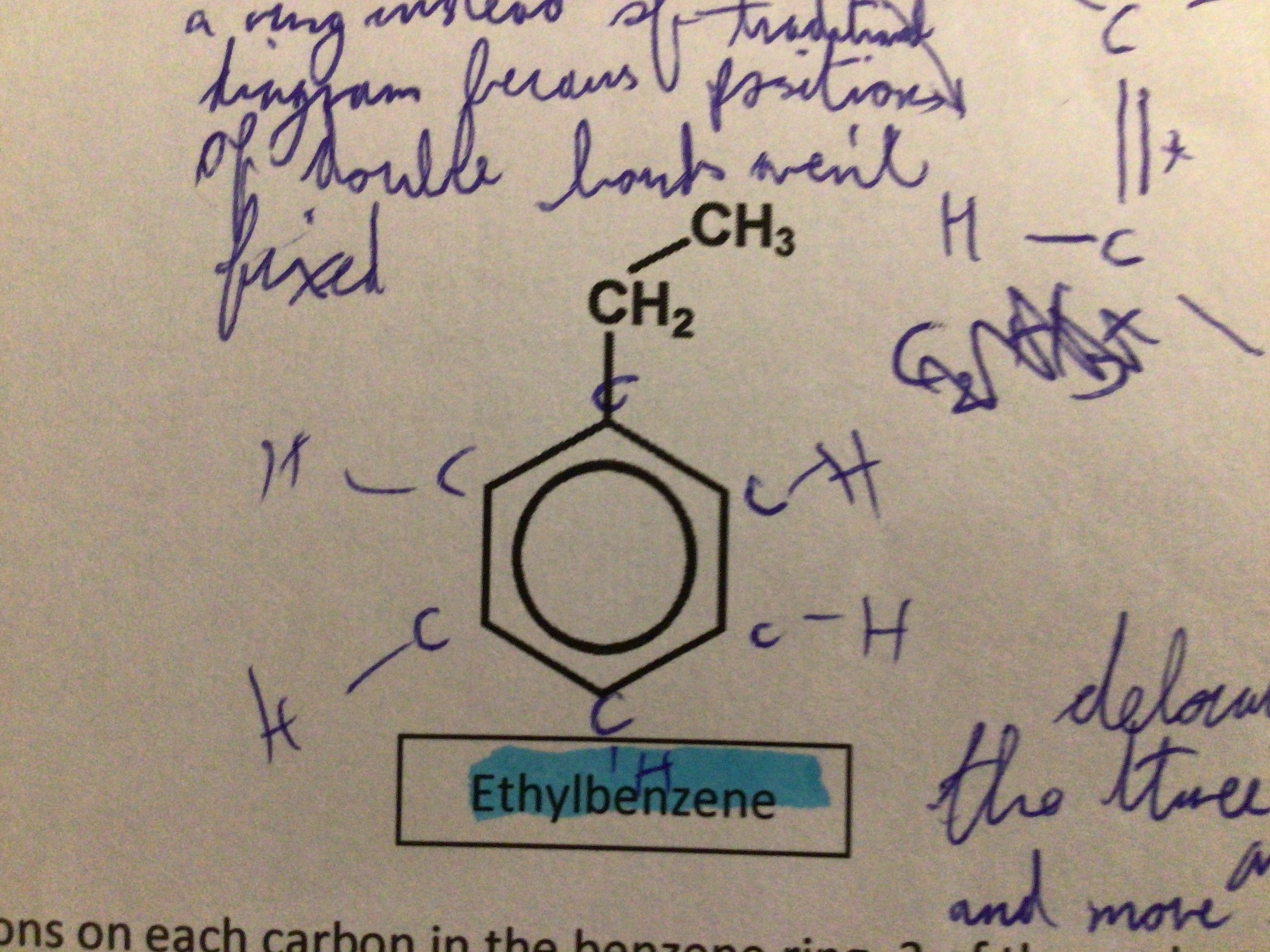
Why is there a ring in aromatic compounds?
Positions of double bonds aren’t fixed
Why are the electrons in a benzene ring delocalised?
The three pi bonds of the three double bonds in the benzene ring are shared and move from carbon to carbon
Evidence for aromatic compounds being stable?
Bond lengths between carbon atoms are identical
What do aromatic compounds contain?
Benzene ring
What are organic natural products?
Compounds made in nature that contain benzene rings
Examples of organic natural products
Cocaine (lab). Paracetamol (lab). Eugenol (clove oil) (natural)
Draw ethylbenzene
…
Draw methyl ethanoate
…
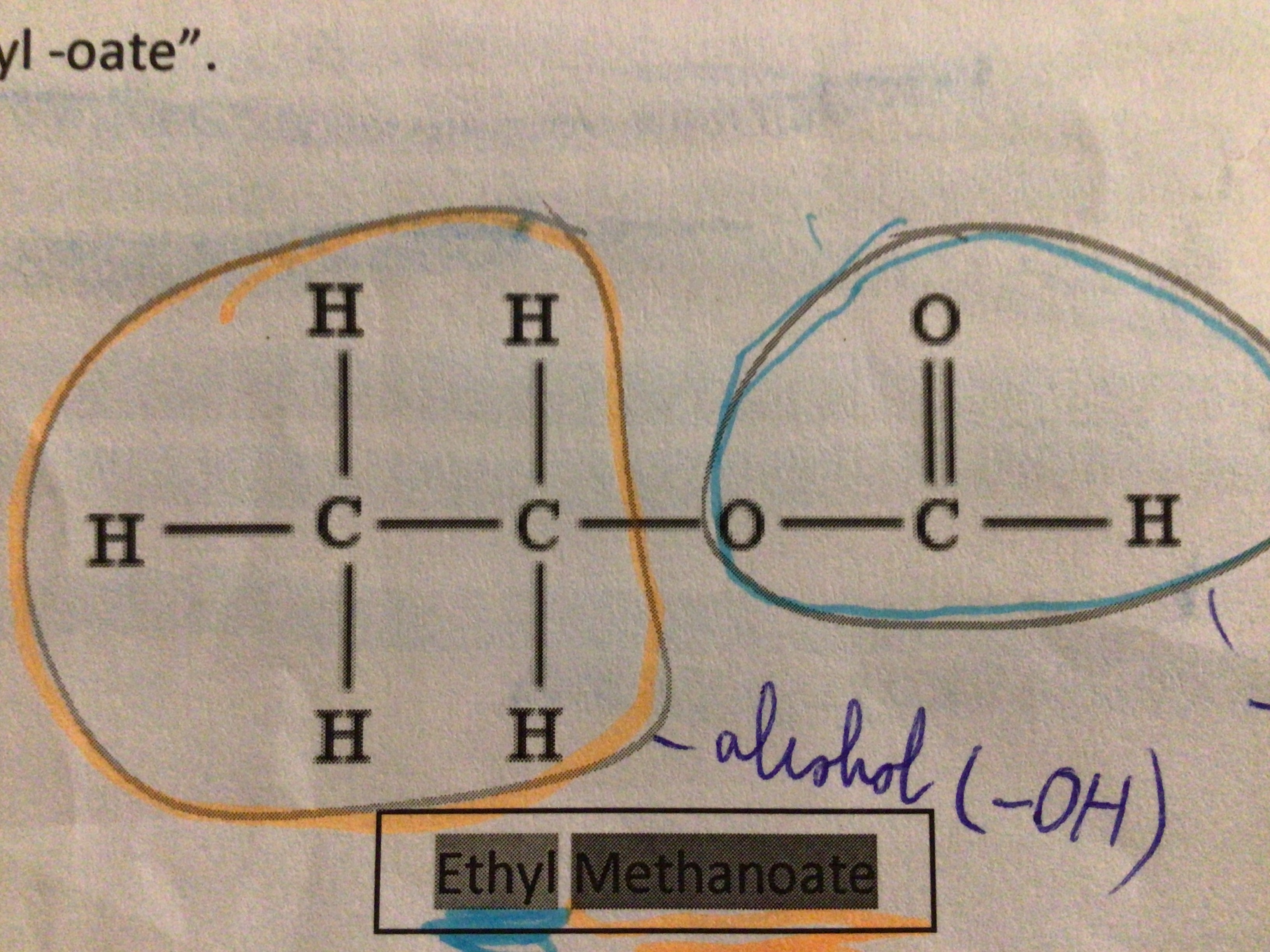
Draw ethyl methanoate
…
Why is only a small amount of benzene used in petrol despite it improving petrol’s octane number?
Carcinogenic
How many electrons does a benzene molecule have?
42
How many electrons in a benzene molecule are involved in hydrogen bonding?
12