Chemistry-Atomz
1/29
Earn XP
Description and Tags
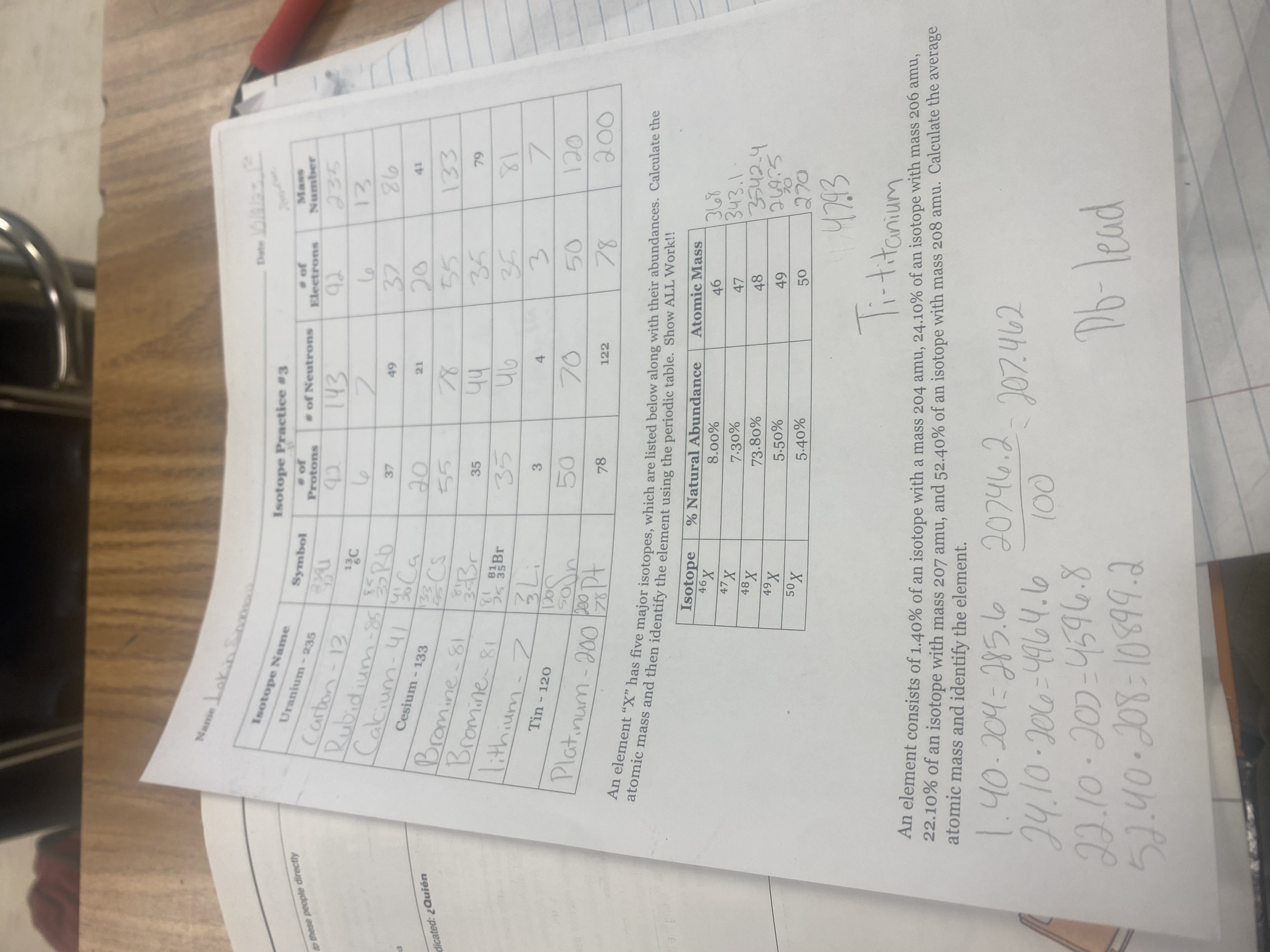
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms

The name of this particle means indivisible and it’s the building block of matter
Atom

The mass of an atom is wut ni-
Protons+Neutrons
Which particle has negative charge???
Electrone

Which particle has no charge
Neutrons

Where are the electrons located
In the spaces around the nucleus
T or F - Protons and Neutrons have around tha same mass
True
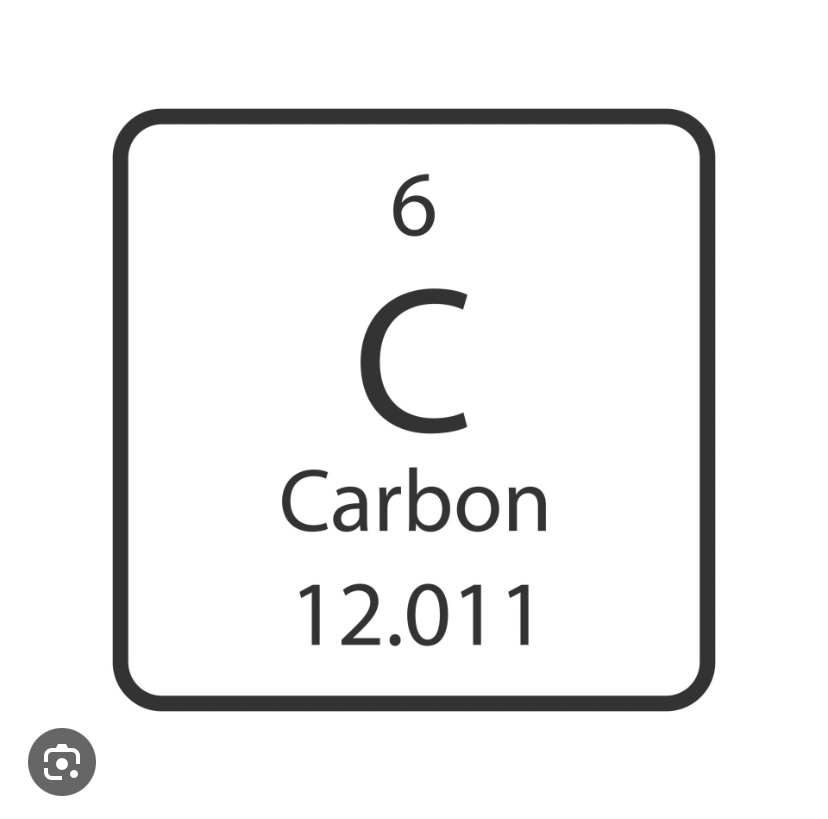
What does the 6 represent
Atomic number
In a neutral atom
Protons=Electrons

What is the atomic mass of this atom
7
Which particle has a positive charge
Proton
Value representing
Atomic number
How is the number of neutrons in the nucleus of an atom calculated
Subtract numbers of protons from the mass number
What is the mass number
The number of protons and neutrons
Elements in groups 3-12 of the modern periodic table and are further divided into transitional metals
Transition element
Which family does Uranium belong 2
Actinide series
Vertical columns of elements (increasing atomic number)
Group
Bro demonstrated a connection between atomic mass and the properties of elements
Dmitri Mendeleev
Elements from groups 1,2 and 13-18 are what
Representative elements
Which family does Calcium belong 2
Alkaline Earth metal (she went over this sh for legit 2 mins 🤦🏽♂️)
Which family does sodium belong 2
Alkali Metal
Which family does bromine belong 2
Halogen
Which family does europium belong 2
Lanthanide series
Which elements r similar to iodine
Chlorine
Which family does silver belong 2
Transition metal
Which family does xenon belong 2
Noble ass
Who found that you should arrange the elements by their atomic number not by atomic mass
Henry Moseley
Which counts as a representative element rmb it’s groups 1 and 2 13-17
Lead (Pb)
States that when the elements are arranged by increasing atomic number
Periodic Law
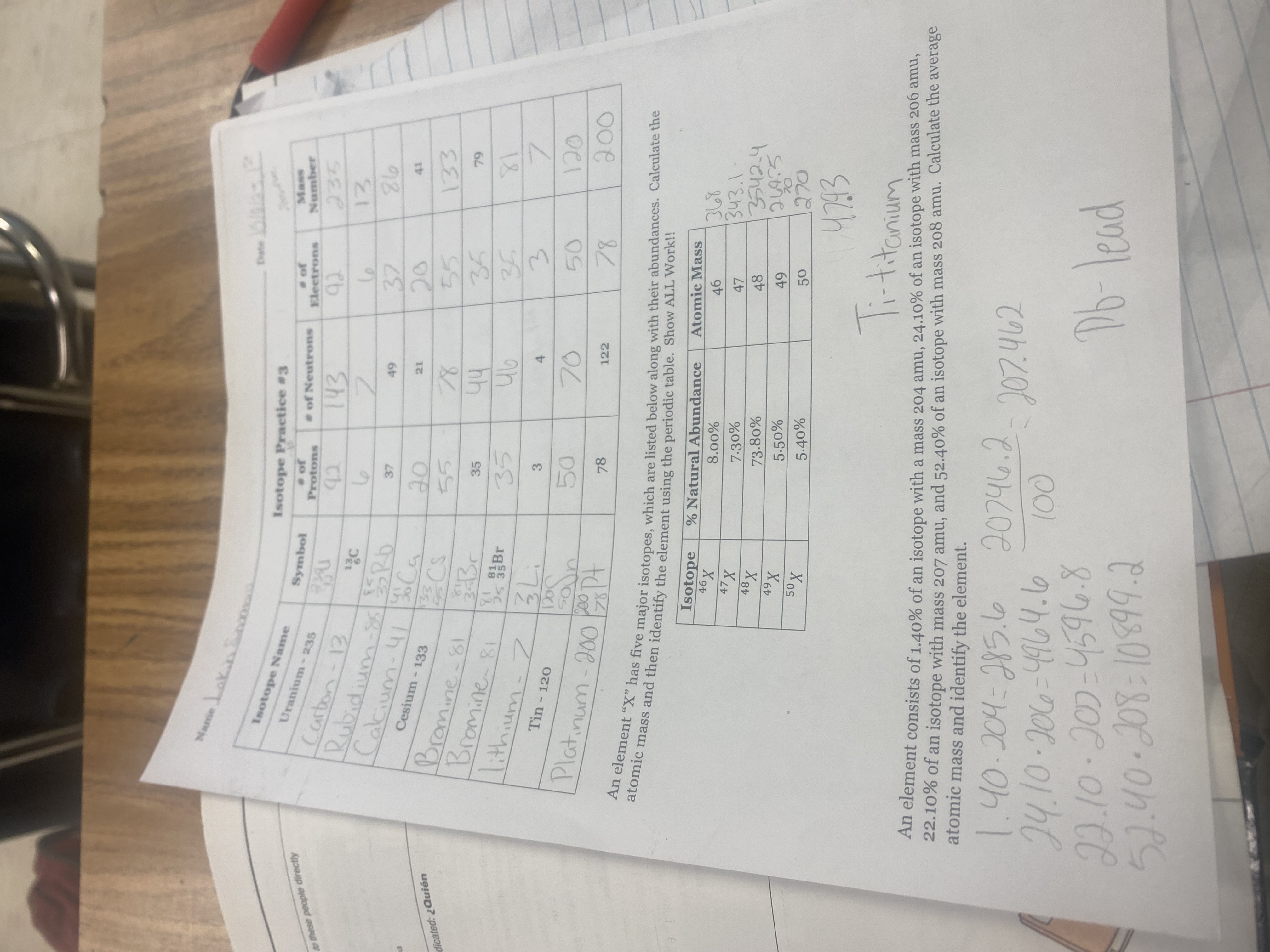
at the bottom what is amu being multiplied
1.40%
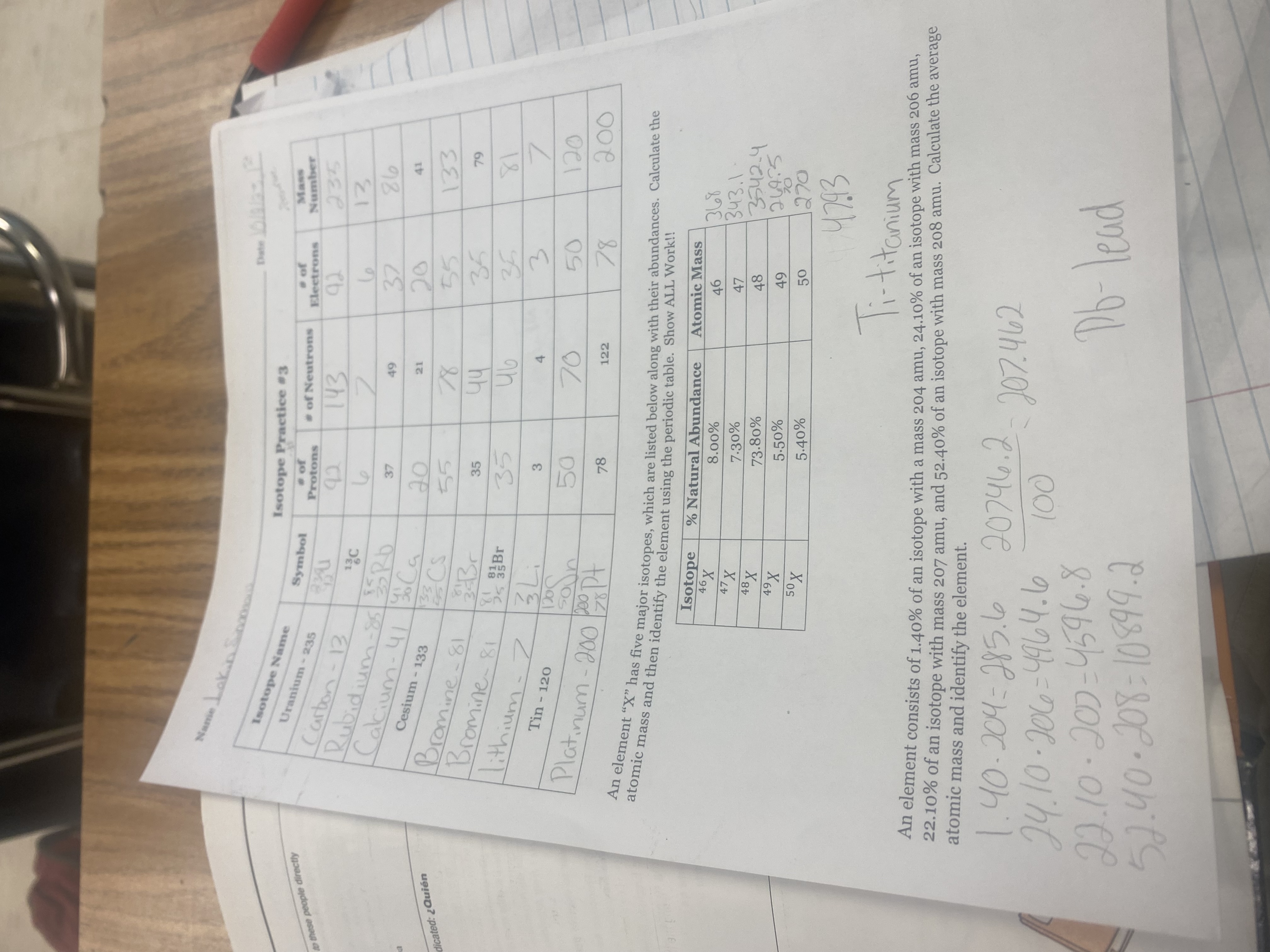
What are the percentages and so forth
24. 10, 22.10,52.40% multiply by the Amus add them up then divide by 100= Answer