some basic concepts of chemistry
1/18
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
19 Terms
atomic mass
mass of 1 atom of any element
mass of an element / (1/12) of 1-C12 atom
molecular mass
mass of 1 molecular
mass of a molecule / (1/12) mass of 1-C12 atom
gram molecular mass
mass of 1 mole atom of any element
molar mass
mass of NA number of particles (calculate in grams)
number of moles with weight
n= given Wt / molecular wt
no. of moles with molecules
no. of particals / 6.02 × 1023
no. of moles with volume
Volume at stp / 22.4 l
percentage composition
weight of the element / total molecular weights x 100
density absolute (g/ml) in case of solids and liquid
mass / volume
density absolute (g/l) in case of gas
d = PM / RT
specific gravity (unitless)
density of substance / density of water at 4 C
relative density (unitless)
d1/d2 = M1 / M2
minimum molecular mass (condition)
for a molecular mass to be minimum, then, no. of atom should be 1
vapour density
M1 /Mh2 = M1 / 2 ( density of the gas with respect to hydrogen gas at constant T,P
avarage atomic weight for isotopes

molecular formula
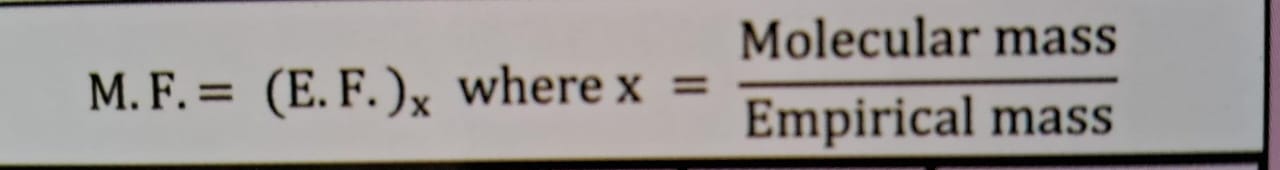
mean molar mass

mole fraction
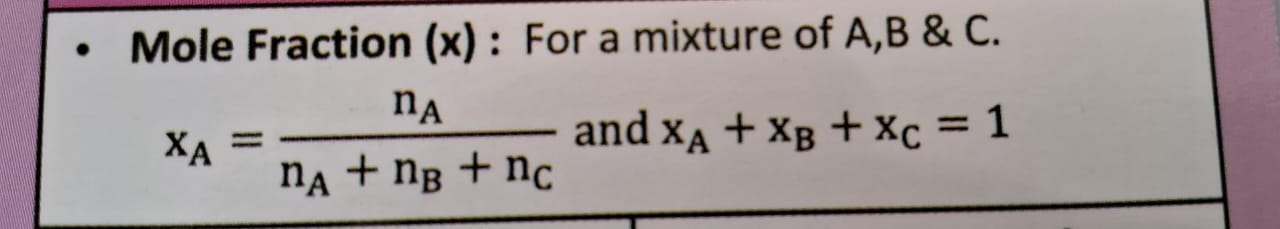
percentage by mass
