Lecture 15 - Cancer Models and Assays
1/98
Earn XP
Description and Tags
ONCOL 335 - Radiobiology. University of Alberta
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
what type of research is E.Coli and S.cervisiae used for?
DNA repair mechanisms
what tyoe of research is C. elegans use for?
apoptosis
what type of research are D. melanogaster used for?
deregulated signaling pathways
what type of research are x. laevis used for
egg extracts great for biochemistry
what type of research is D. rerio used for
developmental studies
why are CHO cells often used for in cytogenetics?
easy to culture and manipulate as 2n = 22
What are spheroids in cell culture?
Spherical, heterogeneous aggregates of proliferating, quiescent, and necrotic cells that retain three-dimensional architecture and tissue-specific functions.
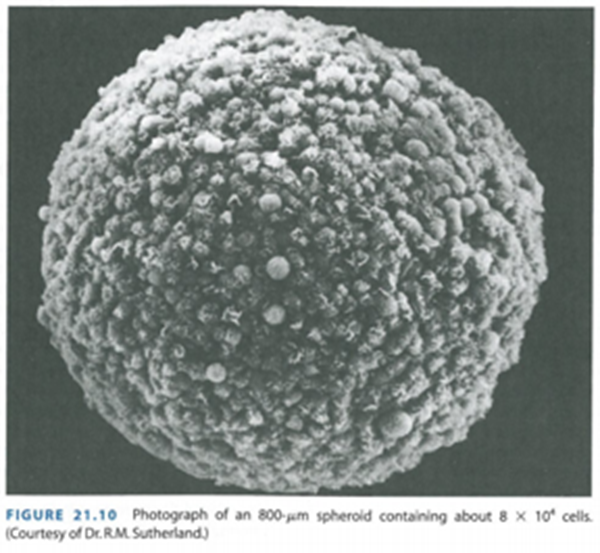
What do spheroids represent in research?
They represent a model for tumor growth and tissue organization, mimicking the in vivo environment more closely than traditional two-dimensional cultures.
What is a characteristic trait of cultured tumor cells from solid tumors?
the ability to form spheroids
What can be formed when a single stem cell of mammary origin is seeded in culture?
a mammosphere
What is the role of animal models in cancer research?
to discover the underlying basis of how and why cancers arise, why primary tumors metastasize, and how various interventions and treatments can stop cancer progression.
What can animal models help us understand regarding tumors?
they help understand how tumors grow, spread, and how preventive interventions or treatments can stop their progression and prolong patient lives.
Why are similarities in animal models studied?
To discover and test new therapies, find better ways to detect cancers early, assess approaches to cancer prevention, and determine factors influencing individual cancer risk and therapy response.
How can studying animal models help improve patient outcomes?
By discovering and testing new therapies that improve patient outcomes.
What factors related to cancer risk and therapy can be determined through animal model studies?
Why some individuals are at higher risk for developing cancer, are less responsive to therapy, or are more likely to develop therapy-limiting toxicity.
Why is it important to study differences between cancer in animal models and humans?
It provides insights that may be unobserved if only similarities are examined.
ex: Mice have different lifestyle and metabolism than us humans, So they get cancer way earlier than us as they only live for two years
4 types of animal models for cancer
Animals in which cancers occur spontaneously without any alteration of the animal’s genes or initiation of cancer by chemical treatments.
Animals whose genes are altered so that they develop spontaneous tumors of the same types and with similar properties as the tumors which those altered genes cause in humans.
Animals that develop spontaneous tumors if they are exposed to environmental factors, such as chemicals or radiation.
Animals whose natural, unaltered genetic makeup permits researchers to identify the genes that generate susceptibility to cancer development.
What factors determine the utility of animal cancer models?
The species, available research reagents and tools, and similarity to the human situation.
What are the most commonly used animal cancer models?
Rodents, particularly mice and rats.
What are the advantages of using mice c.f rats in cancer research?
Mice are smaller, less expensive, have a short lifespan, and their genetic makeup is well understood, making them ideal for genetic studies.
disavantages of using mice in cancer reasearch
Mice have a more limited size and physiological differences compared to humans, which may not fully replicate human tumor progression and treatment responses.
rats are larger and easier to do surgical procedures on
4 types of mouse models
Genetically Engineered Mouse Models (GEM)
inbred mice
transplantation models
carcinogen-induced and spontaneous models
Carcinogen Induce and Spontaneous Mouse Models
certain mouse strains are prone to developing certain cancers
Some mice have been genetically engineered to have pre-disposing mutations that can be triggered to evade normal cellular control systems.
What is the digestive chemically induced mouse model?
A model where mice are exposed to specific chemicals to induce digestive tract cancers, allowing researchers to study tumor development and treatment responses.
Why do scientists use inbred strains of animals in cancer research?
Inbred strains provide genetic consistency, allowing researchers to study cancer in a controlled genetic background.
How does the genetic background of laboratory animals affect cancer research?
The genetic background is a significant component of organ-specific carcinogenesis, similar to how genetic factors affect cancer in human populations.
What is a key strength of the laboratory mouse in cancer research?
The availability of hundreds of inbred strains that harbor different characteristics, including those prone to specific types of cancers.
How does the genetic background of laboratory mice benefit cancer research?
All mice have an identical genetic background, which allows for standardized responses to experimental therapies and treatments.
How do standardized collections of laboratory mice help cancer research?
They allow researchers to investigate virtually any phenotype of interest and take advantage of the accumulated global knowledge on these model systems.
What types of models have been used in breast cancer studies?
Spontaneous and induced models have long been used in breast cancer studies.
What is one way in which some inbred and genetically engineered mice develop mammary cancers?
They harbor mouse mammary tumor virus (MMTV) or other oncogenic viruses and mutations, which trigger mammary cancers.
How can chemicals or hormones be used in breast cancer models?
Chemicals or hormones can initiate cellular alterations that lead to breast cancer in model systems.
How was one brain tumor model created in transgenic mice?
By delivering a viral oncogene called SV40 T-antigen into mouse eggs.
How was the mammary tumor model created in transgenic mice?
By delivering a mutant human oncogene called c-Myc using MMTV, a mouse virus that infects mouse mammary tissue.
What do the mammary tumors in mice resemble?
The mammary tumors in mice resemble human breast tumors.
What is homologous recombination in mouse embryonic stem cells used for in cancer research?
It allows researchers to remove a gene (knock out), replace it (knock in), or perform other genetic alterations to replicate mutations associated with human tumors.
What is the role of CRISPR-Cas9 gene editing in cancer research?
CRISPR-Cas9 allows for precise editing of the genome, making it a powerful tool for altering genes in cancer models.
types of altered mice we can make using CRISPR
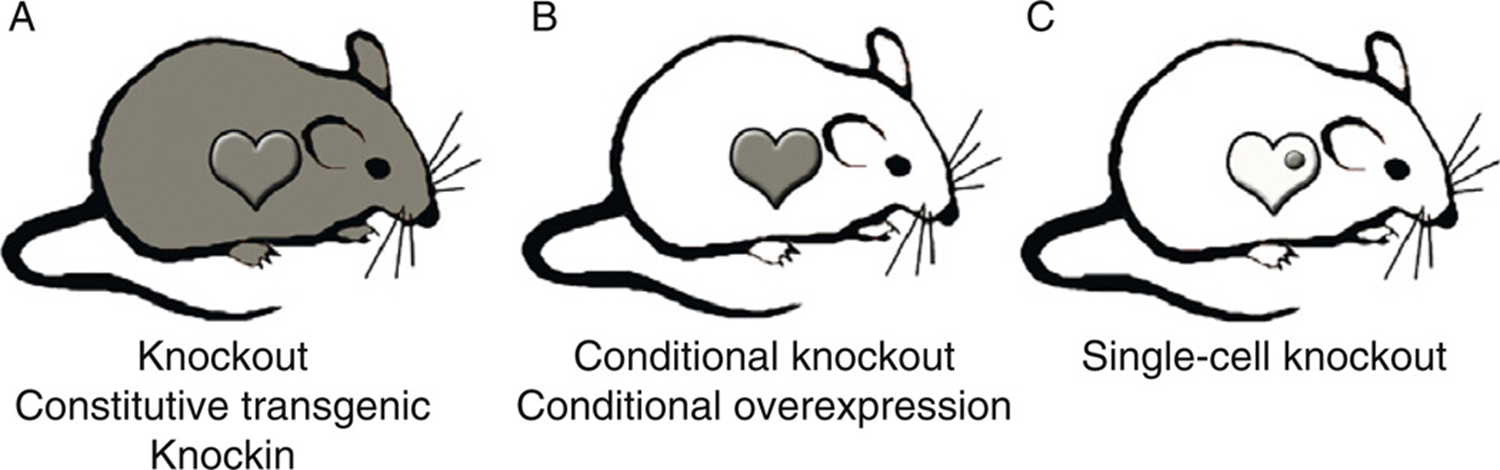
What is the key characteristic of tumor tissues used in allograft (syngenic) transplantation models?
the tumor tissues are derived from the same genetic background as the given mouse strain.
How are cancerous cells or solid tumors used in allograft transplantation models?
Cancerous cells or solid tumors are transplanted into a host mouse.
Why is the transplant not rejected in allograft transplantation models?
Because the cancer tissues and the recipient share the same genetic ancestry, the transplant is not rejected by the host’s immune system.
What are xenograft transplantation models?
Xenograft models involve transplanting actual human cancer cells or solid tumors into a host mouse.
Why don't the host mice reject the foreign cancer cells in xenograft transplantation models?
The host mice have impaired immune systems, preventing rejection of the foreign cells.
What are the two types of transplant placement in xenograft models?
orthotopic
placed in the site where tumor would naturally arise
subcautaneouly
placed just under host’s skin
What is an advantage of xenografting in cancer research?
Xenografting uses real human cancer tissue, which may be more representative of the properties and mutations of human cancer.
What is a challenge of using xenograft models in cancer research?
Due to changes in the host immune system, the model may not fully mimic the situation in actual human patients.
what are the 4 different ways cells can lose their proliferative abilities?
Quiescence
senescnce
terminal differentriation
death
what is quiescence
a reversible property of stem cells where apoptosis and differentiation are inhibited
high free radical scavenger levels
what do we mean by cell survival for differentiated cells?
retention of function
what do we means as cell survival for proliferating cells
the retention of ability to reproduce and divide
what does the type of cell death depend on?
the function of cell cycle checkpoints
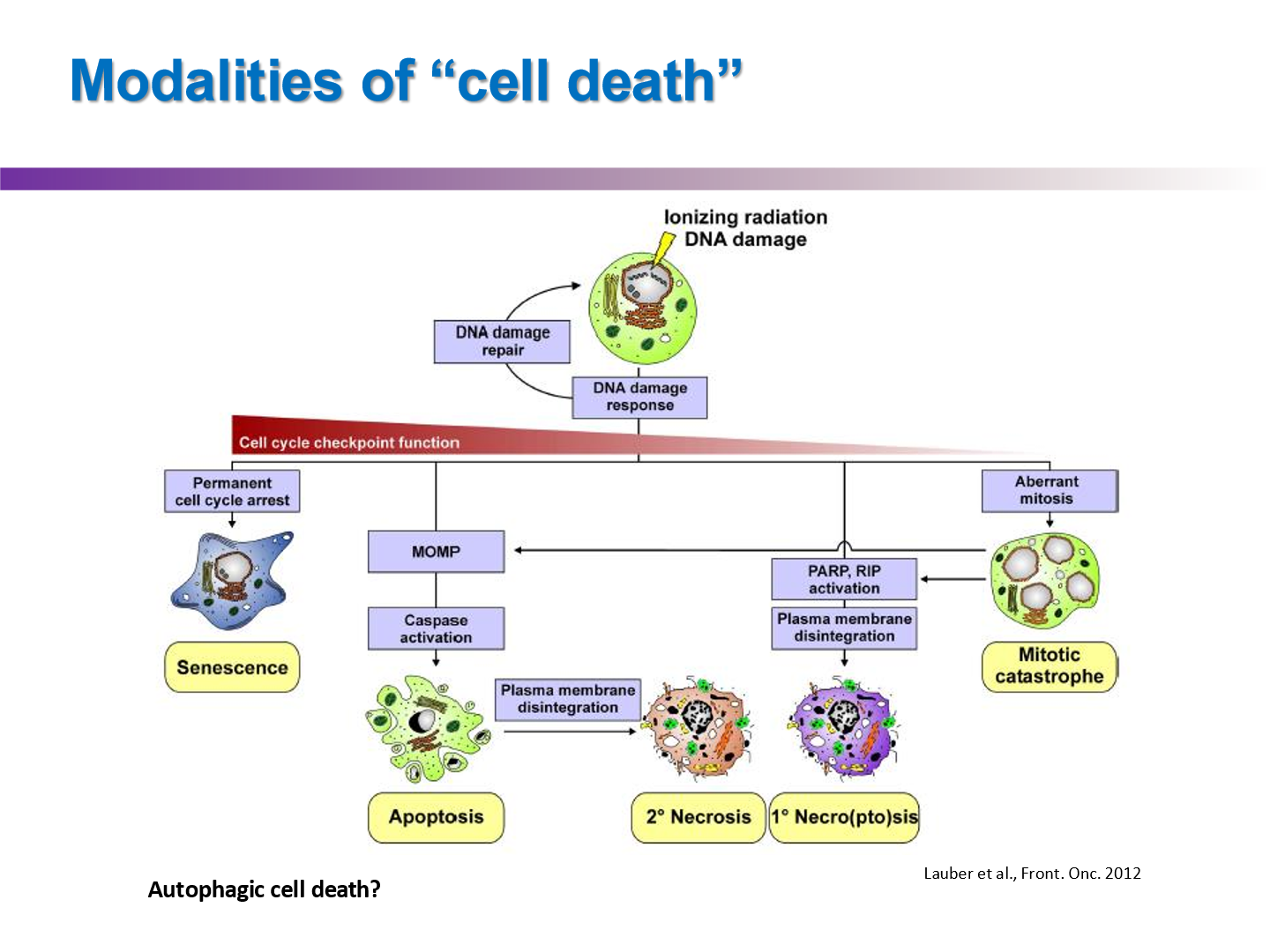
what are the main modalities of cell death after radiation exposure?
Apoptosis
Mitotic catastrophe
Senescence
Necrosis
Autophagy
what three factors do tissue kinetics depend upon?
cell cycle
growth fraction
cell loss factor
What does the growth fraction (G.F.) represent?
The growth fraction is the proportion of proliferating cells.
How is the growth fraction (G.F.) calculated?
G.F. = P / (P + Q), where P = proliferating cells and Q = non-proliferating cells (quiescent, senescent, or differentiated cells).
What is the cell loss factor (Φ)?
the rate of cell loss (due to death) relative to cell division
What happens if the cell loss factor (Φ) equals 0?
If Φ = 0, the actual volume doubling time (Td) equals the potential volume doubling time (Tpot), meaning there is no cell loss.
How is the cell loss factor (Φ) calculated?
Φ = 1 - Tpot / Td,
What does it mean if the growth fraction (G.F.) equals 1?
If G.F. = 1, the potential volume doubling time (Tpot) equals the cell cycle time (Tc).
what does a cell loss factor (Φ) = 1 signify
there is complete cell loss, indicating no cells are contributing to growth
tumor is staying same size
What is the relationship between cell proliferation and cell loss in steady-state conditions?
Under steady-state conditions, a constant cell number is maintained by the balance between cell proliferation and cell loss, i.e., Φ = 1.0.
How does the cell loss factor (Φ) behave in tumors and embryos?
In tumors and embryos, Φ is less than 1.0, indicating that cell loss is not balanced by proliferation.
rate of tumor growth and regression are determined largely by ___
cell loss factor
cell loss factor varies with ….
tumor type and TME
do metastasis have longer or shorter volume doubling times compared to primary tumors
shorter
they proliferate quicker
what three things is tumor growth and regression determined by
rate of cell loss (Φ)
G.F
cell cycle kinetics
How do slow-growing tumors behave after treatment?
Slow-growing tumors may regress rapidly.
How do rapidly growing tumors behave after treatment?
Rapidly growing tumors are expected to regress and regrow rapidly.
is slow regression a indicator of treatment failure?
no
Is the rate of tumor regression after treatment generally prognostic?
no
what factors in the TME can effect tumor regresion
oxygenation, pH, growth factors, cytokines etc
What is one of the simplest endpoints for measuring tumor growth?
Tumors are grown to a specific size, then treated.
What are the two methods used to measure tumor growth?
growth delay and time to reach a certain size compared to the control.
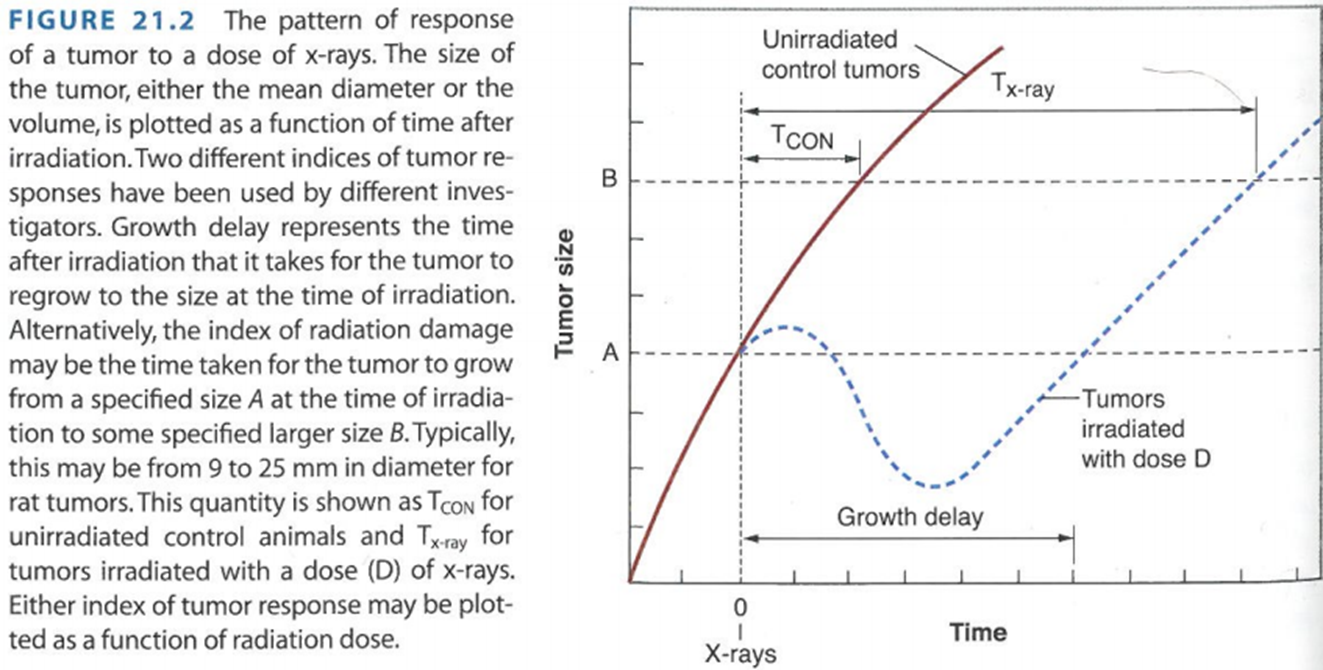
describe this graph
An untreated tumor proliferates as indicated by the red solid line. At time t = 0, the tumor is treated, leading to a growth delay. After some time, the tumor will resume growth with similar kinetics to before. The goal is to determine how long it takes for the tumor to reach the size it was before treatment.
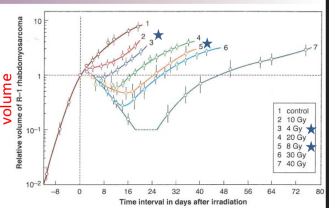
what is dose dependent growth delay
as a function of dose, you get bigger tumor growth delay
how does changing LET effect killing
kills more effectively, as seen in RBE calculations
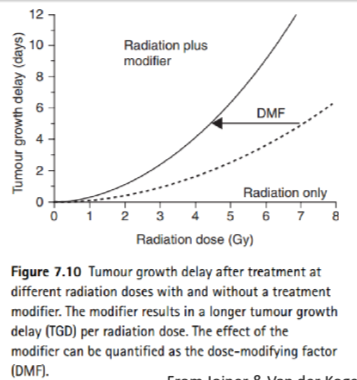
What is a Dose Modifying Factor (DMF) in radiation therapy?
A Dose Modifying Factor (DMF) adjusts the radiation dose based on factors like chemotherapy, medications, age, health status, tumor type, and pre-existing conditions to optimize treatment effectiveness and minimize side effects.
types of imaging techniques used for measuing tumor growth
CT, MRI, PET, BLI (bioluminescence, not used in humans), SPECT, ultrasound
Pros and Cons of CT
Pros:
Fast and widely available
High-resolution imaging of bone and soft tissue
Can be used for a variety of conditions (trauma, cancer, etc.)
Cons:
Exposure to ionizing radiation
Limited soft tissue contrast compared to MRI
Not ideal for pregnant patients due to radiation risks
Pros and Cons of MRI
Pros:
No ionizing radiation
Excellent soft tissue contrast
Non-invasive and versatile (brain, joints, organs)
Cons:
Expensive and less available
Not suitable for patients with metal implants
Can be time-consuming
how does PET work
PET works by injecting a radiotracer (a radioactive substance) into the body. The radiotracer emits positrons, which collide with electrons in the body, producing gamma rays. A scanner detects these gamma rays to create detailed images of metabolic activity, often used for detecting cancer, evaluating heart function, or studying brain activity.
Pros and Cons of PET
Pros:
High sensitivity for detecting cancer and metabolic activity
Can visualize function and activity, not just structure
Useful in oncology, cardiology, and neurology
Cons:
Exposure to radiation (low levels)
Expensive and less widely available
Requires radiopharmaceutical injection
What is BLI
BLI is a non-invasive imaging technique that detects light emitted by bioluminescent organisms or cells. It uses luciferase enzymes and their substrates to generate light, which is captured by sensitive cameras. BLI is primarily used in preclinical research to study biological processes in live animals.
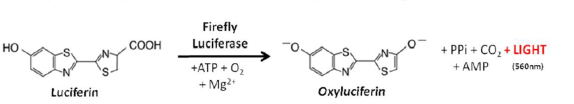
Pros and Cons of BLI
Pros:
Non-invasive, real-time imaging of biological processes
High sensitivity in small animal models
No ionizing radiation
Cons:
Limited to preclinical (animal) studies, not for humans
Requires genetically modified organisms expressing luciferase
Depth penetration is limited to superficial tissues
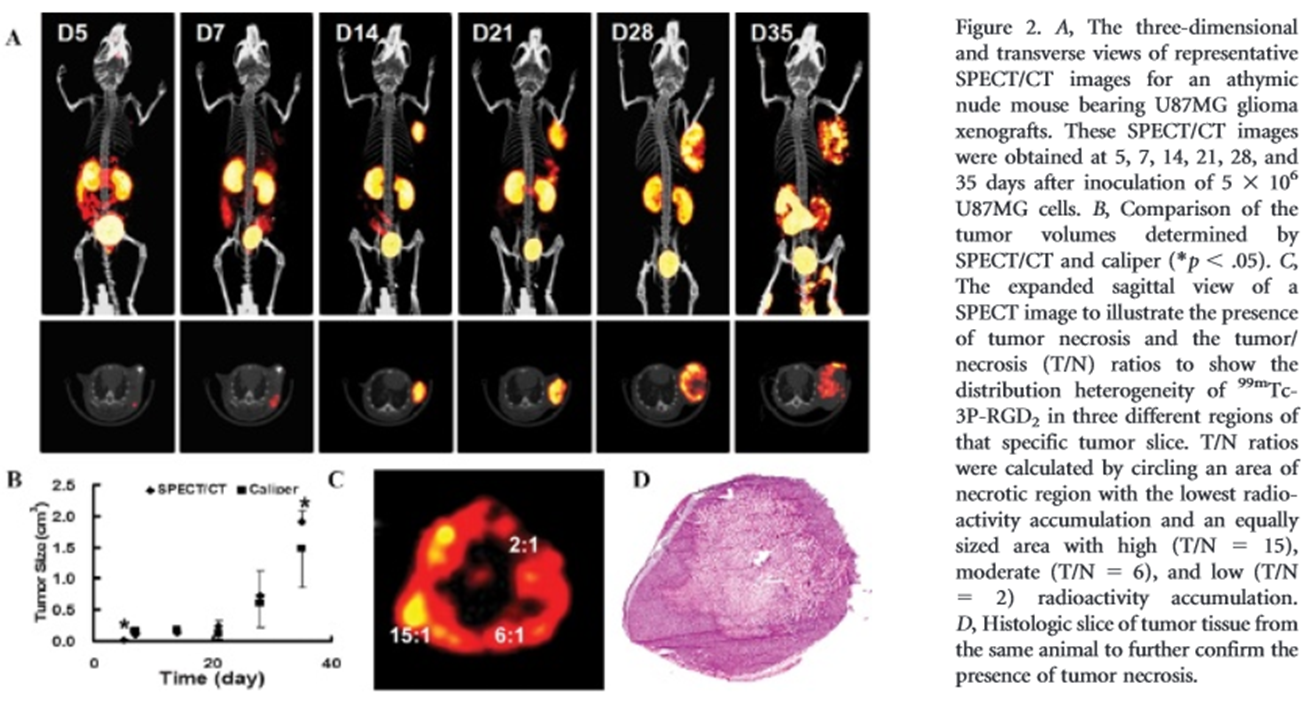
What are the pros and cons of SPECT (Single Photon Emission Computed Tomography)?
Pros:
Functional imaging (e.g., blood flow, metabolism)
Useful in oncology, cardiology, and neurology
Lower cost than PET
Cons:
Exposure to ionizing radiation
Lower resolution compared to CT or MRI
Limited availability
Pros and Cons of ultrasound
Pros:
No ionizing radiation
Real-time imaging, portable, and relatively inexpensive
Safe for use in pregnant patients and children
Cons:
Limited depth penetration and resolution
Dependent on operator skill
Can be difficult to use for imaging structures behind bone or air
What is tumor control probability?
A measure of the likelihood that a given treatment will lead to the eradication of a tumor, often used in radiotherapy to assess treatment effectiveness.
the probability that all of the clonogenic cell is killed
describe the steps to a tumur cure/control assay (AKA TCD50)
Inject mice with tumor
Wait until tumor reaches certain size
Must control: larger tumor = more clonogenic cells, so you have to kill more cells to get tumor control
irradiate mice with varying amounts of dose
plot dose of radiation to cure 50% of mice
creates sigmoidal curve
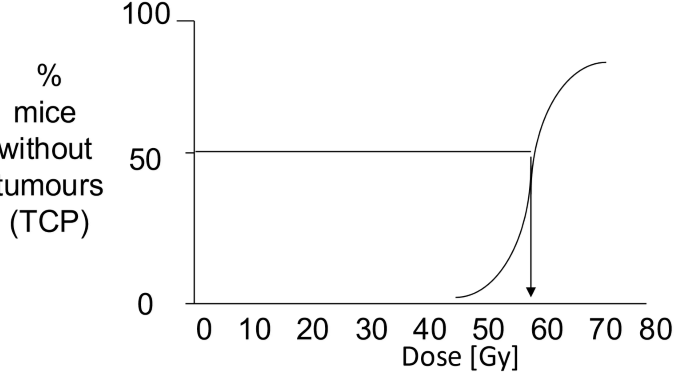
what is the TCD50 of this graph
~ 58 Gy
although the TCD50 is the most clinically relevant assay, what three reasons make it the most difficult
it is expensive
it is labour intensive
we kill a lot of mice
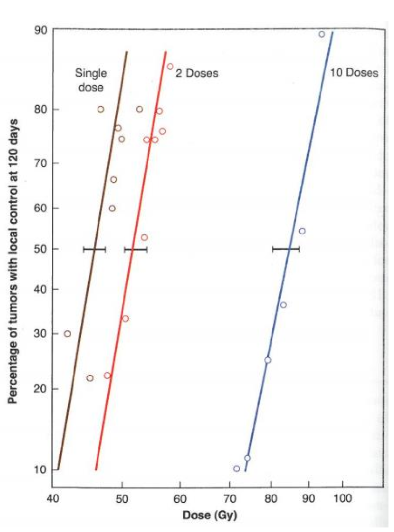
why would we do a TCD50 assay test with different fractionation protocols?
to calculate the alpha/beta of the tumor
why would we do a TCD50 assay test with split dose experiment?
to estimate how long it would take for a tumor cell to repair
and to see if we can use hypo/hyperfractionation regiment
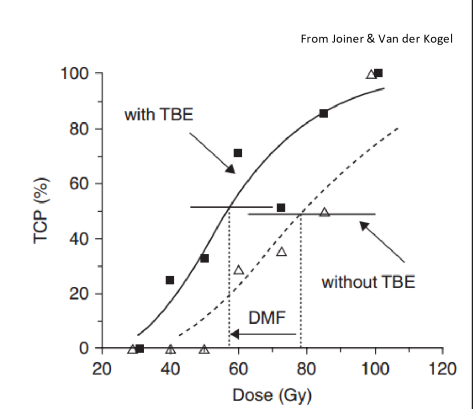
what type of chemo radiosensitizer is used with radiation to make a dose modifying factor?
cisplatin
shifts curve to left
formula for dose modifying factor

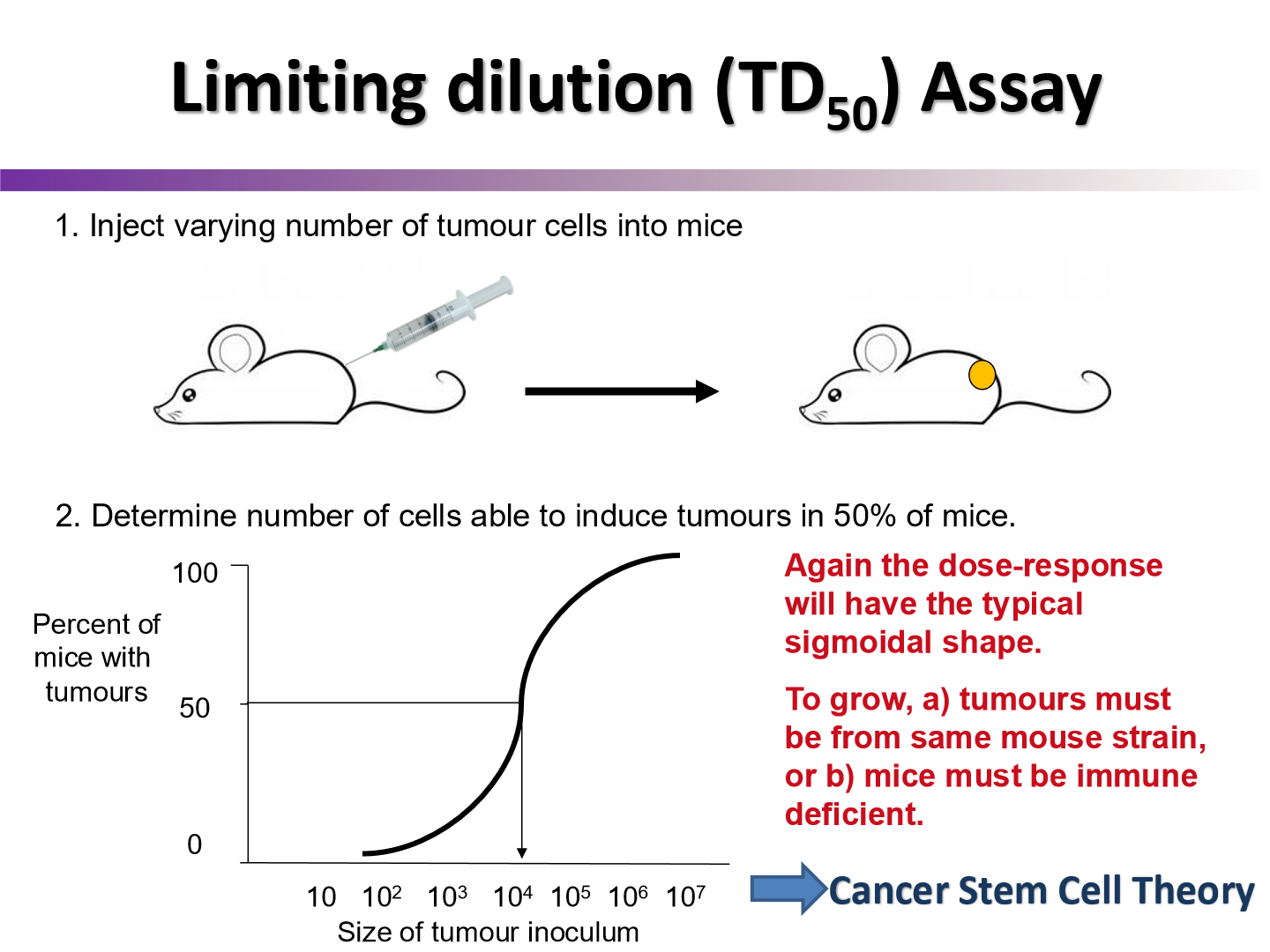
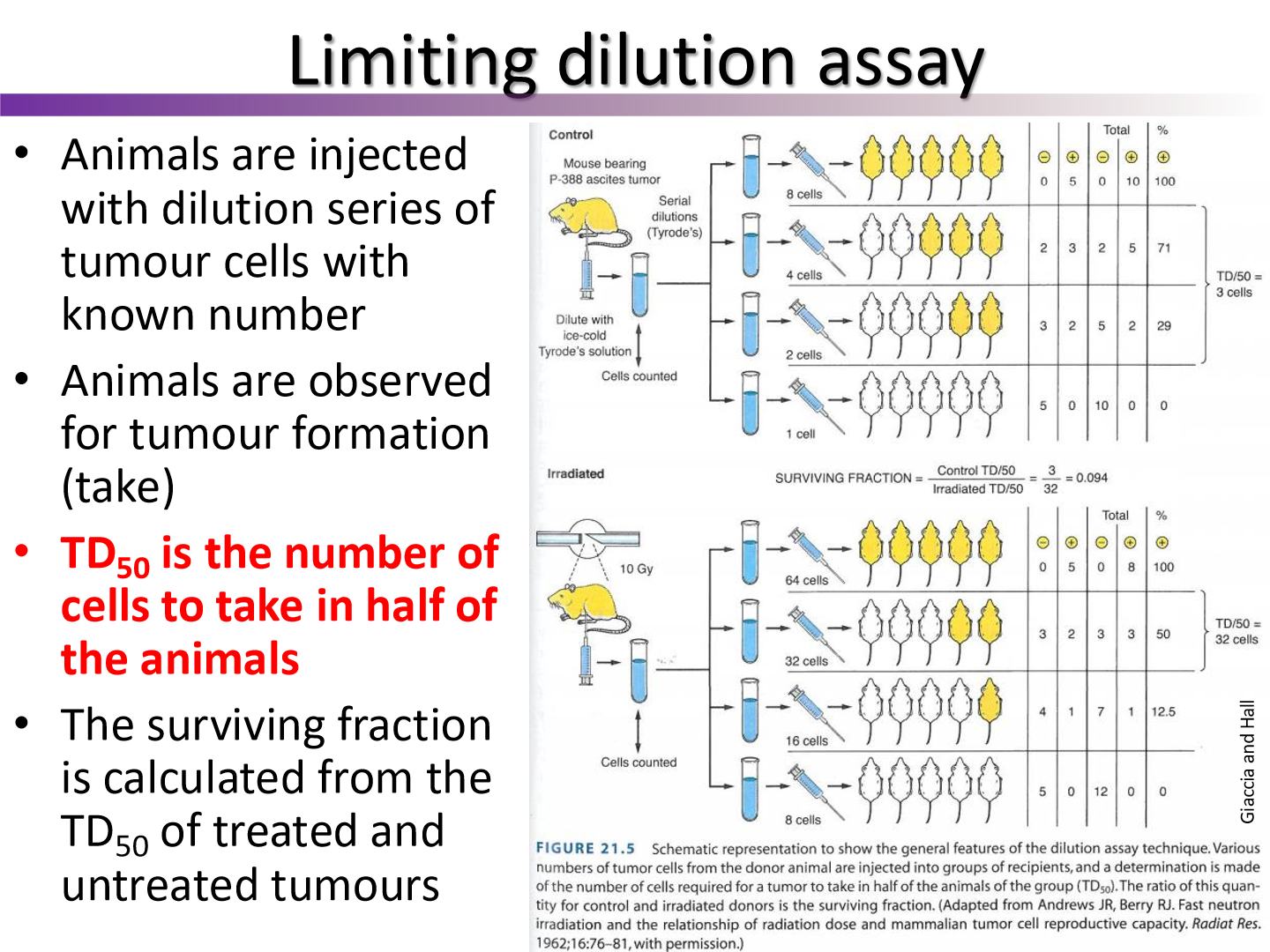
describe the steps of a limiting dilution (TD50) Assay
inject varying number of tumor cells into the mice
look at the amount of mice who get tumors and plot to how many cells they are injected with
what sort of modification can we add to the limiting dilutation assay
we can irradiate the mouse with the tumor with various doses before removing it and then transplant it to other mice
see how TD50 changes
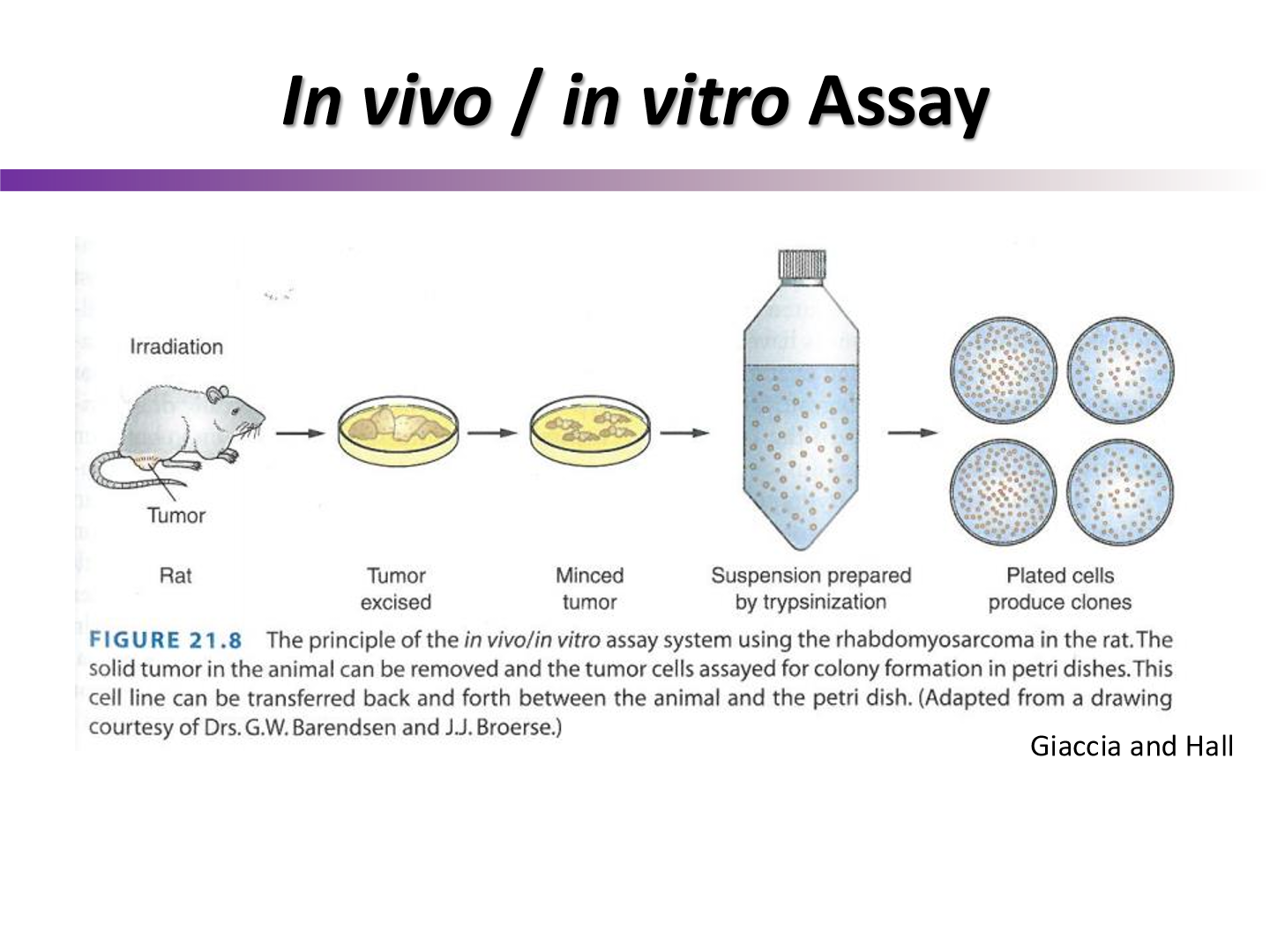
describe the steps to an in vivo/in vitro assay
irradiate mouse with tumor
remove tumor from mouse
plate the tumor and see how many colonies are formed
describe the steps of a lung colony assay
irradiate mouse with tumor with varying amounts of dose
excise tumor and plate colonies
implant the tumor back into healthy mice
open up lungs and count any many lung colonies/lesions were created
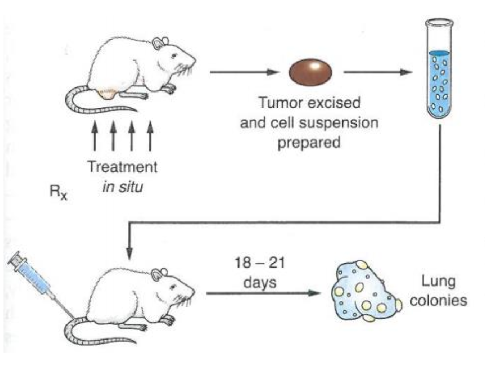
why are the lungs checked for cancer lesions?
many tumors will metastasize in the lung since the travel through the blood