10/11 Equilibrium I + II
1/12
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
What is Kc?
The equilibrium constant for a given temperature.
What is Kc for the generic equation aA + bB → cC + dD?

What phases are not included in Kc for heterogenous Equilibria?
Solids or pure liquids as their concentrations stay constant throughout the reaction.
0.2 moles of phosphorus chloride (PCl₅) decompose at 600K in a vessel of 5dm³. The equilibrium mixture is found to contain 0.08 moles of chlorine and PCl₃ is also produced. Write an expression for Kc and calculate its value.

What is the total pressure of a gas mixture?
The sum of all its partial pressures
What is a mole fraction?
The proportion of a gas mixture that is a particular gas.
What is the formula for mole fraction of a gas in a mixture?
No of moles of gas/total no of moles in mixture
What is the equation for partial pressure of a gas?
Mole fraction x total pressure of mixture
What is the equation for Kp for the generic gaseous equation aA + bB → cC + dD?
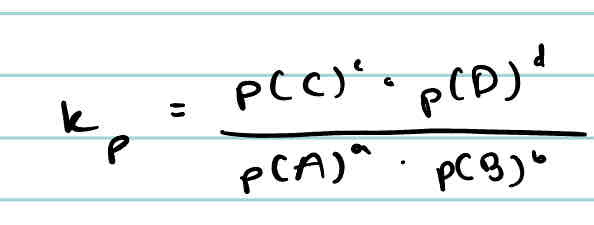
How does the size of the equilibrium constant tell you where the equilibrium lies?
Greater the Kc or Kp, the further to the right that equilibrium is.
Why doesn’t concentration or pressure affect the equilibrium constant?
Equilibrium shifts to the left or right to counteract the change. Eg. More reactant = shift to right to get rid of this increase, more product is produced and Kc stays the same. The ratio of reactants to products stays the same.
Why does temperature affect the equilibrium constant?
Changes in temperature alter the amounts of products and reactants present, changing the ratio of reactants:products, and therefore the equilibrium constants change.
Why don’t catalysts affect the equilibrium constant?
They have no effect on the position of equilibrium. They only mean that equilibrium is reached faster.