The 7 O-chem topics
1/16
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Allotropes of Carbon
Diamond, Graphite, Buckminister Fullerine
Allotropes
The same ELEMENT, but arranged differently
Diamonds
Very hard substance, composed of covalently bonded Carbon
Graphite
Pencil Lead! —> sheets of carbon
Buckminister Fullerene
C_60 in a dome-like pattern (kind of like a soccer ball)
Alcohols
Hydrocarbons with at least one hydroxyl group (—OH)
These are all able to hydrogen bond because of the hydroxyl group (Hydrogen atom is bonded to Oxygen).
Isopropyl Alcohol
aka - 2-propanol
rubbing alcohol
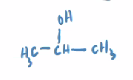
Ethanol
The alcohol that we consume (in drinks etc.)
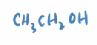
Octane Boosters
additives that increase the octane rating of fuels.
prevent fuel knocking when engines are under high temp. and pressure
Fuel knocking
The premature combustion of fuel-oxygen mixtures in engines. (think swing analogy)
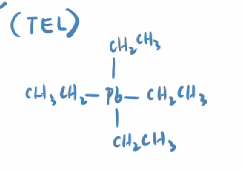
Tetra-Ethyl Lead
TEL:
One Pb atom with 4 ethyl groups attached
we stopped using this because we learned its neurotoxic effects and how it corroded engines
“Gasohol”
Ethanol is now also added to gasoline to boost octane rating
E-10 means 10% ethanol and 90% gasoline
“Flex-Fuel“ vehicles can use gasoline from E-10 up to E-85!
Oxygenator
These are added to increase fuel efficiency by introducing more Oxygen
ethanol is also a common oxygenator because of the hydroxyl group (adds oxygen)
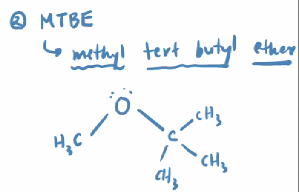
Methyl Tert Butyl Ether
MTBE
It was a nice oxygenator (the ether adds oxygen) until we realized its danger
Contaminates groundwater still today
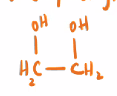
Ethylene Glycol
1,2 - ethanediol
used as antifreeze in coolants for cars
has a “sweet“ taste that animals try and eat (they die tho cuz its bad)
“better” girl makes things “bitter“
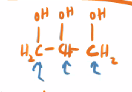
Glycerin
1,2,3 - propanetriol
Also called Glycerol
used as a humectant (a moisturizer just like hyaluronic acid 🔥 )
a lubricant
Keeps hand sanitizer from evaporating
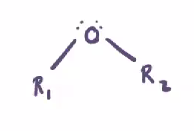
Ether
Oxygen bonded between two hydrocarbons
cannot undergo hydrogen bonding because the oxygen is not bonded with hydrogens
Only dipole-dipole bonding and London dispersion
Weak intermolecular force —> volatile (lower boiling point)
we love coc