Chapter 14: Vesicular Traffic, Secretion, and Endocytosis
1/59
Earn XP
Description and Tags
Highlighted Terms from Slides Defined
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
60 Terms
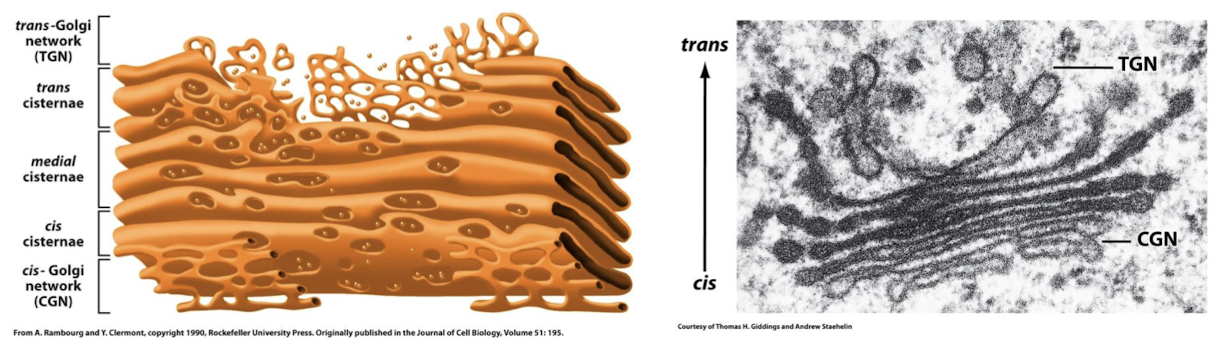
Cisternae
Flattened membrane-bound compartment, as found in the Golgi complex and endoplasmic reticulum.
Not continuous with the Er or one another
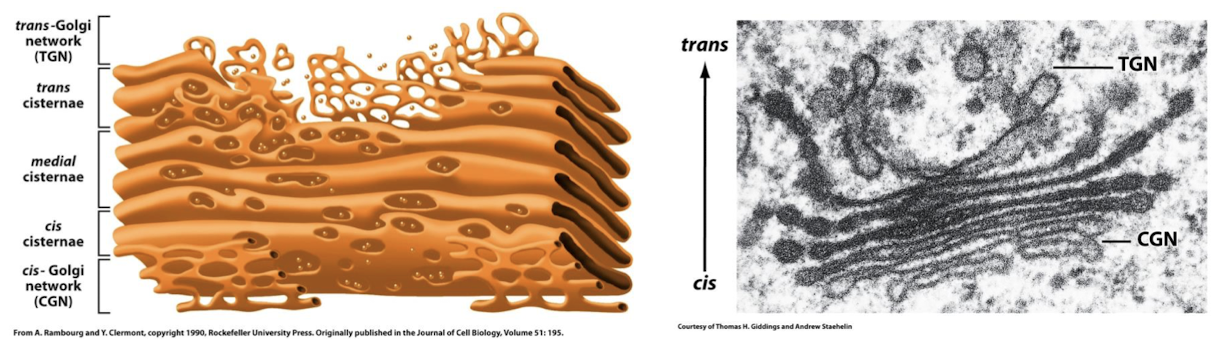
Cis-Golgi Network
The cis compartment faces the ER/nucleus, it recieves proteins and lipids from ER via transport vesicles.
When a new cis-Cisterna is generated it displaces all other cisternae in the trans direction
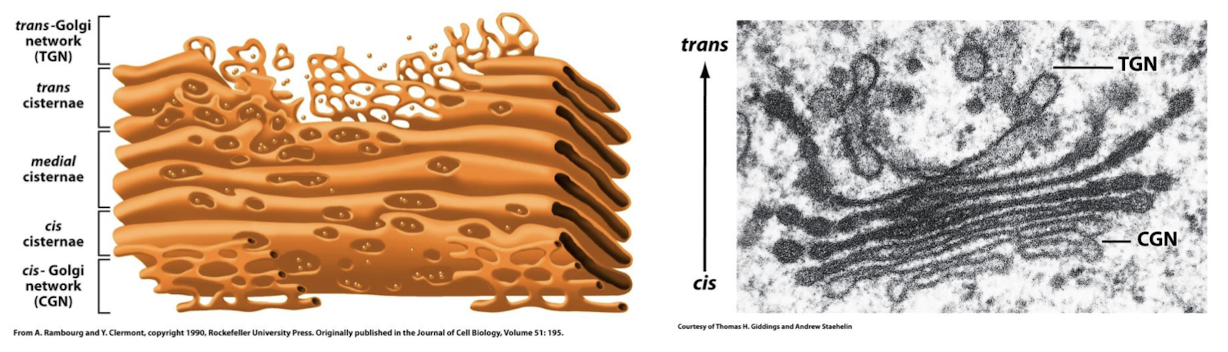
Medial-Golgi Network
Molecules are modified as they pass through this medial-Golgi network from the cis-Golgi network to the trans-Golgi network.
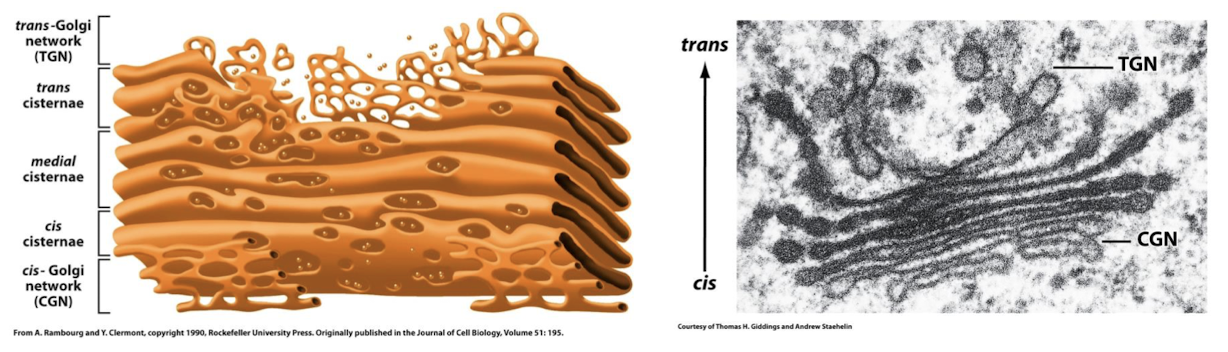
Trans-Golgi Network
Packages proteins and lipids from medial-Golgi network into transport vesicles for delivery to the plasma membrane and lysosomes.
Terminal sorting station where signals on cargo molecules are interpreted and proteins and lipids are packaged in transport vesicles for delivery to:
Integral membrane proteins of the plasma membrane
Lysosomes
resident Er proteins are returned to the Er via retrograde transport vesibles from the cis-Golgi Network
Golgi Complex Overview of Synthesis, Modification, Sorting, and Delivery
Modifies core N-linked sugars of ER derived proteins
Completes synthesis of sphingomyelin
O-linked glycosylation of certain proteins
attached to -OH of serine or threonine residues of select proteins
Covalent attachment of oligosaccharides to lipids
Synthesis of most cellular complex polysaccharaides
Polysaccharides of extracellular matrix of animal cells
Polysaccharides of cell walls of plants except cellulose
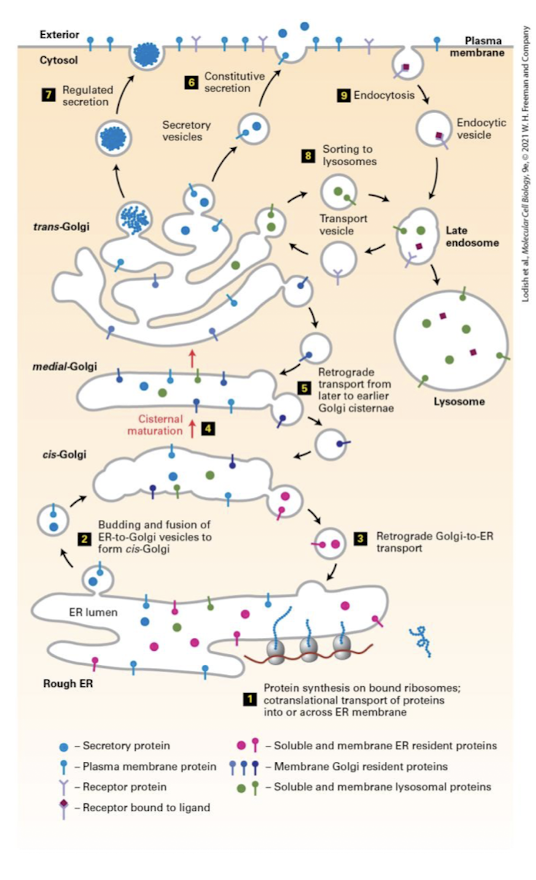
Reads sorting signals of Er derived proteins and packages them into transport vesicles for delivery to appropriate destinations
Transport Vesicles
A small, membrane-bounded compartment that carries soluble and membrane “cargo” proteins in the forward or reverse direction in the secretory pathway. Vesicles form by budding off from the donor organelle and release their contents by fusion with the target membrane.
What ER synthesized lipids and translated proteins that passes quality control are packaged into.
ER Exit Sites (ERES)
Where transport vesicles bud from the ER at these specialized ER zones for transport.
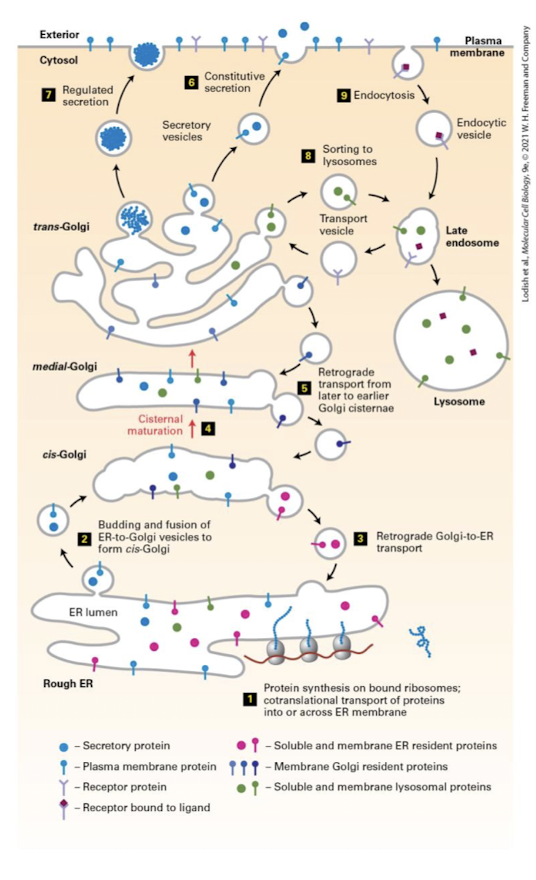
ER to Golgi Intermediate Compartment (ERGIC)
Vesicles move through the action of microtubule motor proteins and fuse into a sorting station called the ER to Golgi Intermediate Compartment (ERGIC)
Also receives transport vesicles from the Golgi for transport back to the ER.
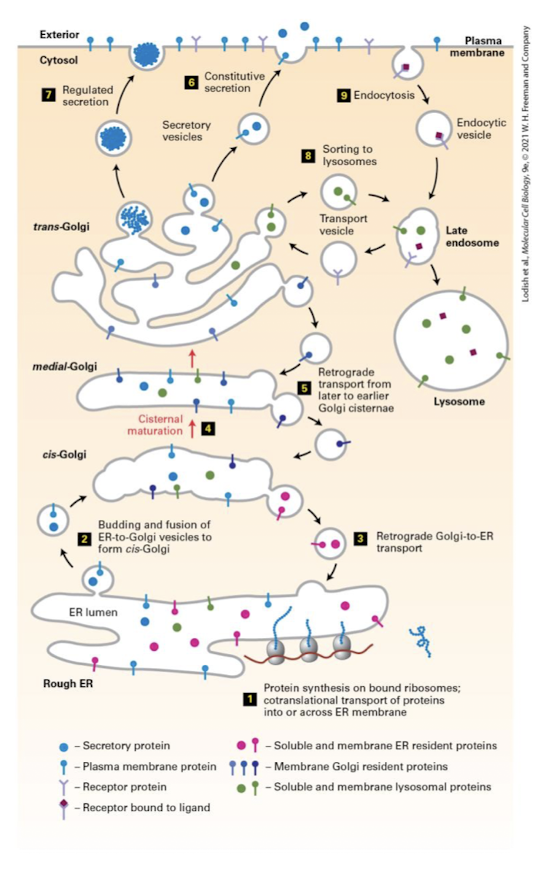
Anterograde
Vesicular trafficking in the direction of ER to plasma membrane.
ER→ Golgi → Lysosome or Plasma Membrane
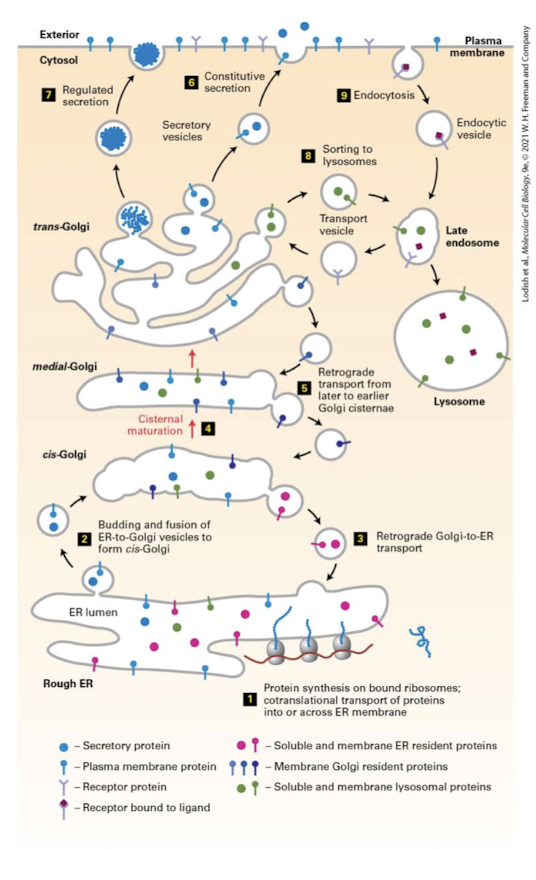
Retrograde
Backward-moving transport vesicle that retrieves proteins from the cis-Golgi cisterna and returns them to the ER.
Plasma Membrane → Endosome → Lysosome → Golgi → ER
Used to maintain resident proteins in their appropriate cis- medial- and trans- locations.
Cisternal Maturation Model
The process by which most proteins traverse the Golgi by sequential maturation of cisternal compartments.
Collective process of new cis-Cisternae formation via anterograde movement and recycling proteins via retrograde movement.
Medial-Golgi Enzymes
Complete the synthesis of some lipids like sphingolipids
Modify N-linked glycosylation- glucosidases and glycosyltransferases remove and add sugar in an organelle-specific and cell-type-specific manner
Generate O-linked glycosylation
Constitutive Secretion
The process in which in all cel types at least some proteins are secreted continuously.
Some proteins of certain cells are continuously secreted.
Regulated Secretion
Proteins are stored inside the cell in specialized vesicles known as secretory granules until a signal for exocytosis causes them to be released.
Other proteins are only released when a cell receives or activates a go-ahead signal.
Secretory Granules
Proteins released by regulated secretion are stored in these specialized vesicles just beneath the plasma membrane until a signal promotes fusion of the vesicles with the membrane and the controlled release of the proteins.
Endocytosis
General term for uptake of extracellular material by invagination of the plasma membrane; includes receptor mediated endocytosis, phagocytosis, and pinocytosis.
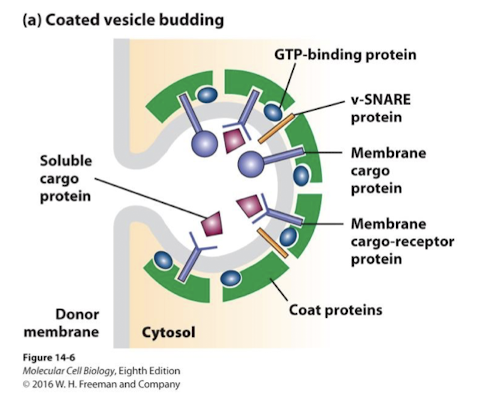
Coat Proteins
Transport Vesicles are formed through the action of distinct coat proteins that serve several functions:
Select the appropriate cargo proteins and lipids
Curve the membrane of origin, forming the spherical vesicles
Detach the vesicles from membrane of origin
Select targeting and docking proteins that facilitate fusion with the appropriate target membrane
COPII-Coated Vesicles
A class of proteins that coat transport vesicles in the secretory pathway. COPII-coated vesicles move proteins from the endoplasmic reticulum to the Golgi.
Form from the ER and move anterograde to the ERGIC and Golgi complex.

COPI-Coated Vesicles
A class of proteins that coat transport vesicles in the secretory pathway. COPI-coated vesicles move proteins from the Golgi to the endoplasmic reticulum and from later to earlier Golgi cisternae.
Move retrograde from ERGIC and Golgi to Er, or from the trans-Golgi to the cis-Golgi cisternae.

Clathrin-Coated Vesicles
A fibrous protein that with the aid of assembly proteins polymerizes into a lattice-like network at specific regions on the cytosolic side of a membrane, thereby forming a clathrin-coated pit that buds off to form a vesicle.
Move materials from TGN to endosomes, lysosomes and become secretory vesicles/ granules desined for the plasma membrane- anterograde.
Also drive endocytosis, importing cargo from the plasma membrane- retrograde.
Vesicles Formation Overview
Specific Small G-proteins, unique to each type of coat, are recruited to sites of vesicle formation and initiate membrane curvature.
G-protein SAR 1 promotes formation of COPII coated vesicles
G-protein ARF promotes formation of COPI and Clathrin coated vesicles.
Coat proteins binds specific sorting signals in:
The cytosolic domain of transmembrane cargo proteins
Receptor proteins bound to soluble cargo in the lumen of the forming vesicle.
Coat proteins also recruit proteins that direct appropriate docking and fusion of vesicles to target membrane
Rabs: G-proteins that promote vesicle docking to appropriate target membranes
SNAREs: promote fusion of vesicle and target membranes
Specialized microdomains in the Er membrane called ERES (ER Exit Sites) are enriched with the G proteins and their activating GEFs
these domains accept recently synthesized proteins, and when threshold concentrations are reached, the GEF’s activate the G proteins- GDP is exchanged for GTP
This changes conformation of the G protein- projecting a hydrophobic alpha-helix that spontaneously inserts into the outer leaflet of the ERES.
G-proteins (aka GTPase switch protein)
Group of intracellular switch proteins that cycle between an inactive state with bound GDP and an active state with bound GTP. Includes the Galpha subunit of trimeric (large) G proteins, monomeric (small) G proteins (e.g., Ras, Rab, Ran, and Rac) and certain elongation factors used in protein synthesis.
Diverse family of small regulatory proteins that control function/ activity of many proteins/ enzymes.
In the GTP bound state they adopt an active conformation and can bind to and thereby allosterically regulate other proteins activities.
In the GDP bound state they adopt and inactivate conformation and cannot interact with target proteins.
Activated by GEFs, inactivated by GAPs, kept inactive by GDIs (guanine nucleotide dissociation inhibitors.)
Vesicle Fusion Overview
Once a vesicle is free of the membrane of origin the protein coat disassembles- this allow the targeting proteins to do their jobs
Vesicle transport is driven by associated motor proteins that move along cytoskeleton
Rabs and SNAREs then promote docking and fusion respectively.
Rab Proteins
A family of small GTPases that regulate the tethering and targeting of transport vesicles.
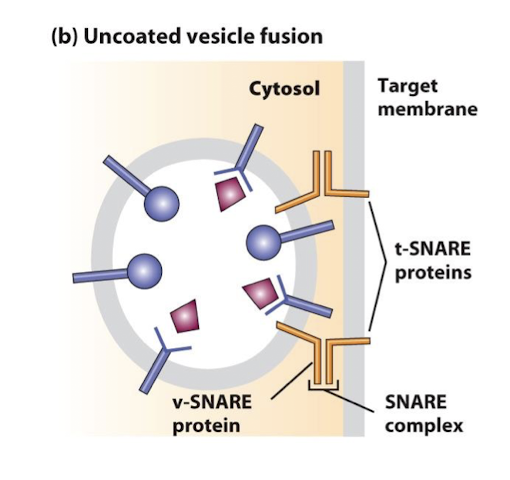
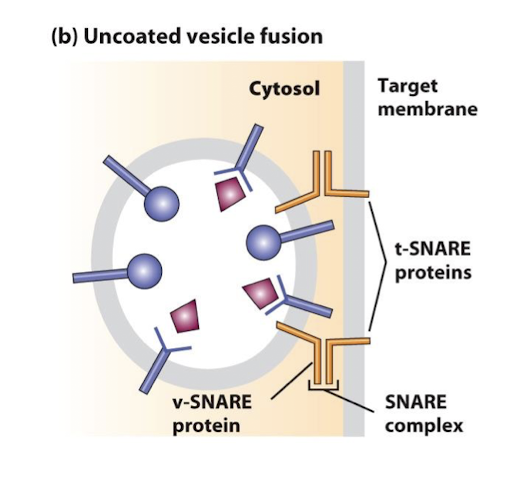
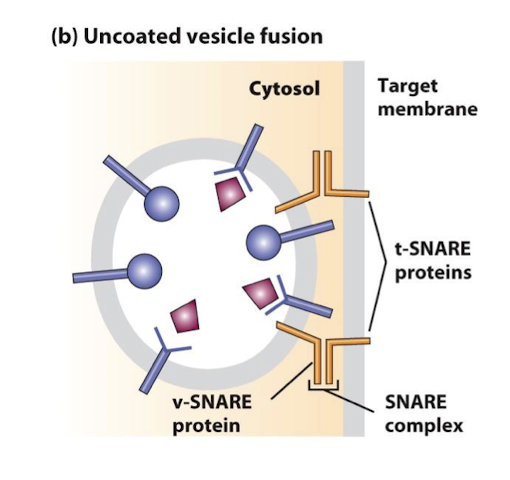
SNAREs
Cytosolic and integral membrane proteins that promote fusion of vesicles with target membranes. Interaction of v-SNAREs on a vesicles with cognate t-SNAREs on a target membrane forms very stable complexes, bringing the vesicle and target membranes into close apposition.
In the formation of COPII coated vesicles: motor proteins deliver the vesicles toward the cis-Golgi network via microtubules where they fuse in a SNARE dependent process into a short-lived transition state, the ER-Golgi intermediate compartment, which then processes proteins for transport to the cis-cisterna or back to the ER
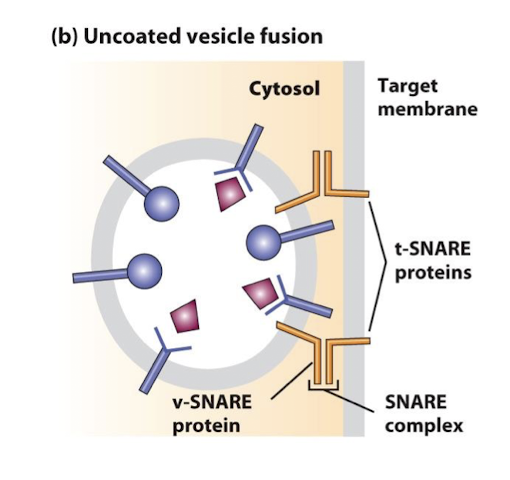
v-SNAREs
A family of small proteins that reside in the membrane of transport vesicles, and through their specific association with similar proteins in the target membrane bring about vesicle membrane fusion.
Sar1
Sar1 promotes the formation of COPII coated vesicles
Activating GEF Sec12
Activated Sar-GTP recruits the adapter proteins sec23 and 24
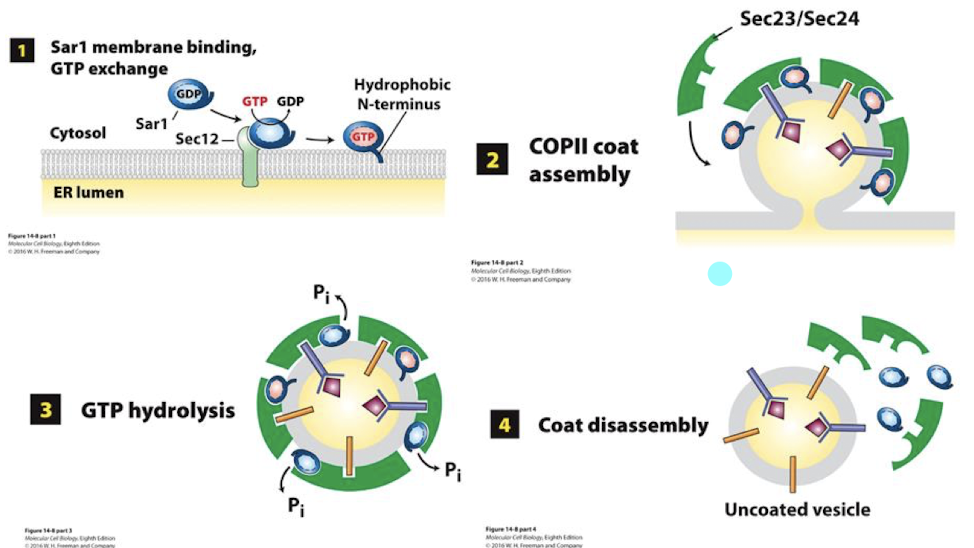
Sec12
Sar1’s activating GEF in formation of COPII coated vesicles
Sar1-GTP
Binds the Er outer membrane and inserts its alpha-helix into the outer leaflet. This expands the leaflet and initiates curvature.
In formation of COPII coated vesicles
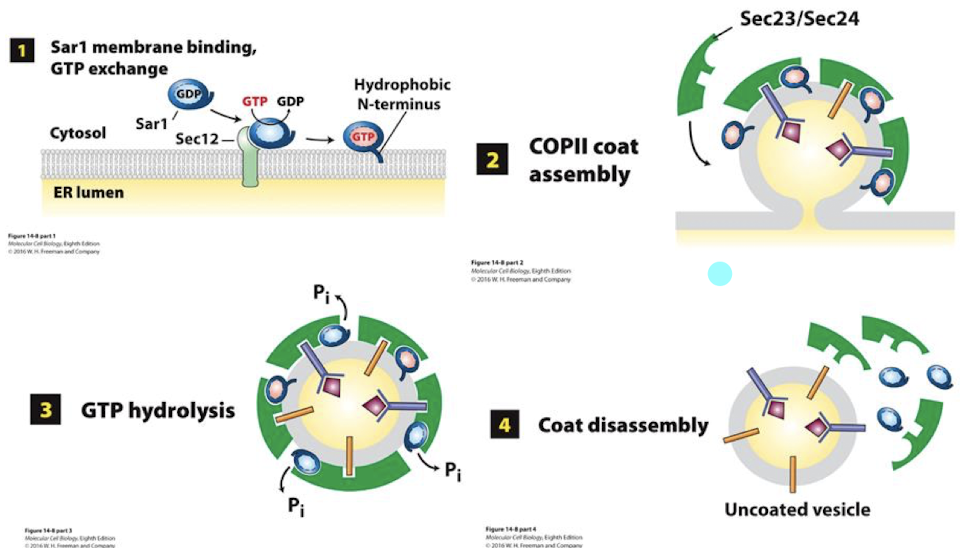
Sec proteins in formation of COPII
On the cytoplasmic surface of the ER, these Sec proteins recognize and bind soluble cargo receptors and ER export signal sequences of TM proteins
Sec 23 and 24 find and bind appropriate cargo receptors and TM cargo proteins, aggregating them into confined space.
The recruitment of Sec 23/24 continues to curve the ER membrane, rounding it up into a spherical shape.
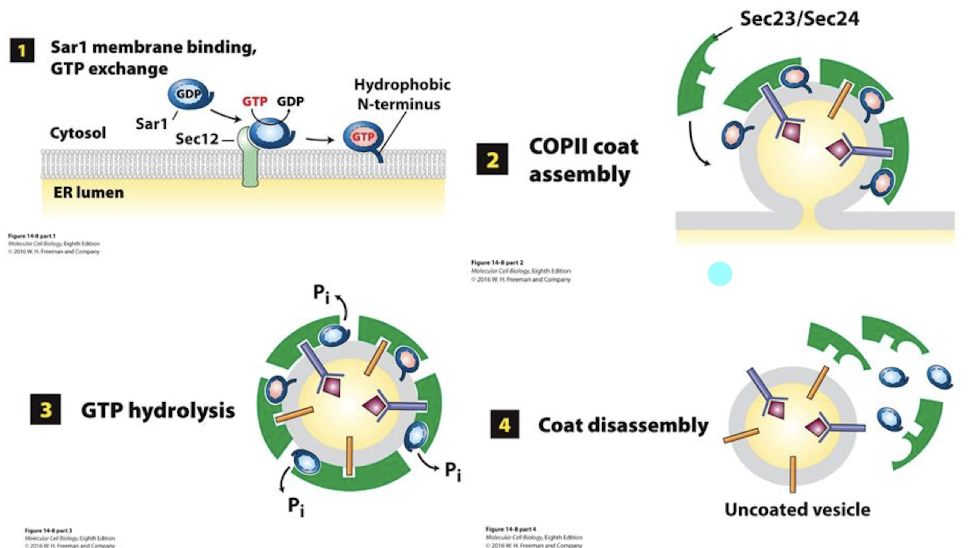
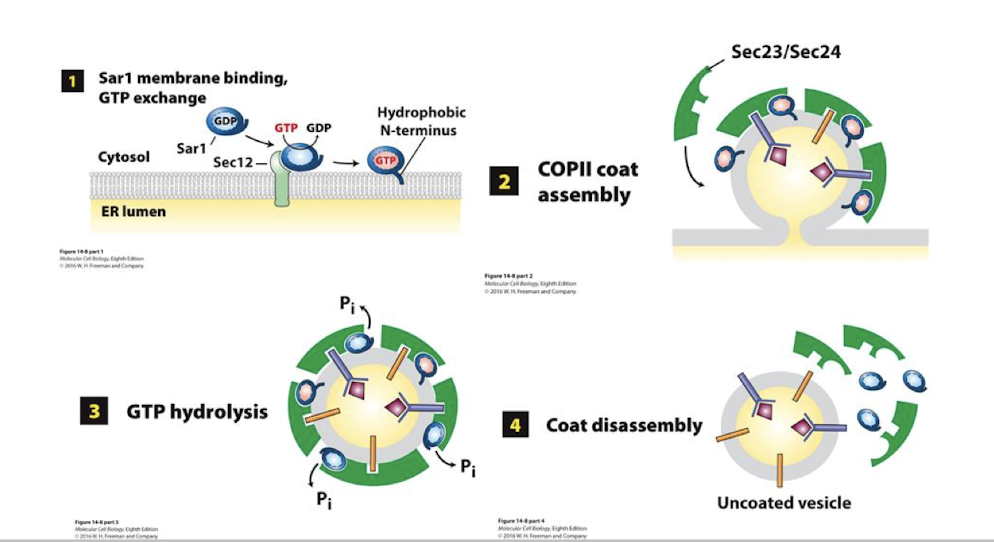
Sec 13 and 31 spontaneously assemble as a caged lattice (lower panel) surrounding the vesicle. This pinches it free of the ER membrane.
Once free, these vesicles must shed the protein coats. This is initiated by Sar-mediated hydrolysis of GTP to GDP, causing a conformational change and dissociation of the COPII protein lattice.
Sorting Signal
A relatively short amino acid sequence within a protein that directs the protein a particular transport vesicles as they bud from a donor membrane in the secretory or endocytic pathway.
Two Types:
The cytosolic surface of membrane cargo proteins possess signal that is directly bound by specific coat proteins
Soluble cargo proteins possess distinct signals that are bound by the lumen portion of transmembrane receptor proteins
KDEL
resident soluble ER protein all possess this amino acid sequence (Lys-Asp-Glu-Leu) which is sufficient to target any protein to the ER
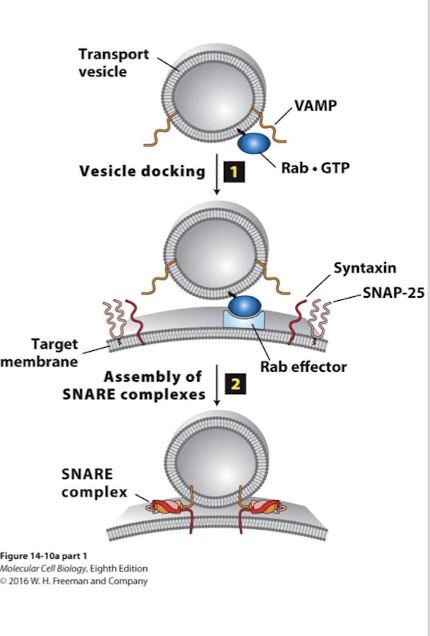
Targeting and fusing vesicles: Docking
Rab mediated: Rabs: another G protein family provide specificity between vesicle and appropriate target membrane
Rabs are lipid anchored G proteins positioned in the cytosolic leaflet of transport vesicles
As Rabs are being assembled in the COPII GEFs activate them
The vesicles are transported by motor proteins into the cell and Rab-GTPs bind to specific Rab effector proteins on appropriate target membrane.
Binding of Rab to its effector docks the cargo vesicle to the appropriate target membrane.
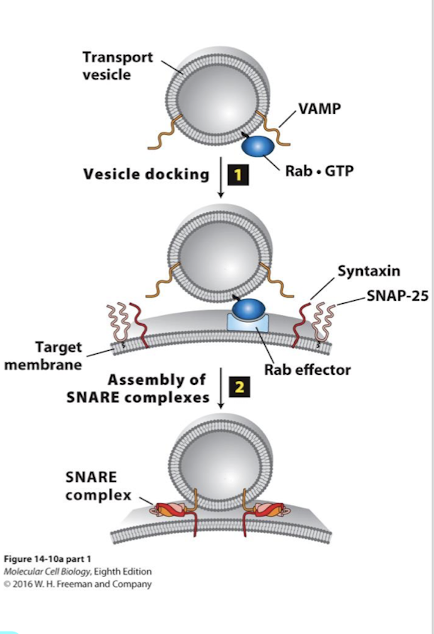
Targeting and fusing vesicles: Fusion
Between vesicle and target membranes.
SNAREs: Proteins that promote fusion between cargo vesicles and target membrane
>35 SNARE family membranes in two categories
v-SNAREs (vesicle-membrane)
t-SNAREs (target membrane)
v-SNAREs and t-SNARES have complementary domains that promote fusion of the two lipid bilayers
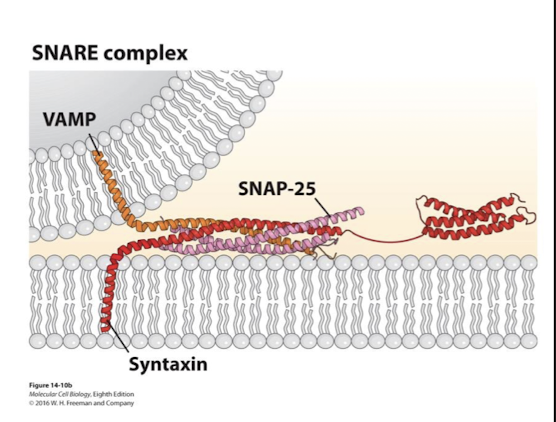
Syntaxin
A transmembrane SNARE
Important in SNARE mediated fusion during neurotransmitter exocytosis
VAMP, SNAP-25, and it’s alpha helixes coil around one another
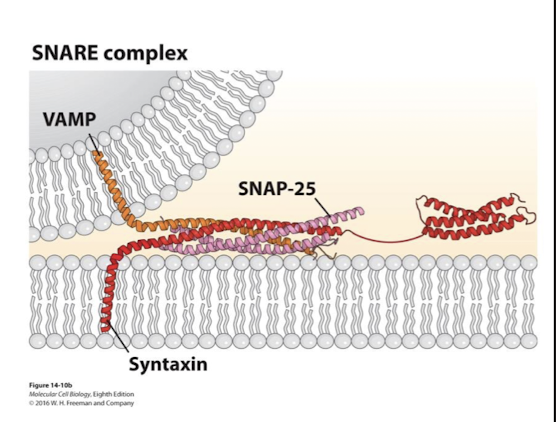
VAMP
Single transmembrane v-SNARE
Important in SNARE mediated fusion during neurotransmitter exocytosis
Syntaxin, SNAP-25, and it’s alpha helixes coil around one another
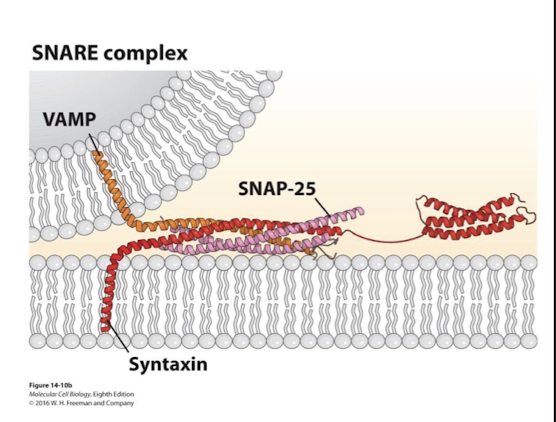
SNAP-25
lipid anchored (2 copies)
Important in SNARE mediated fusion during neurotransmitter exocytosis
VAMP, syntaxin, and it’s alpha helixes coil around one another
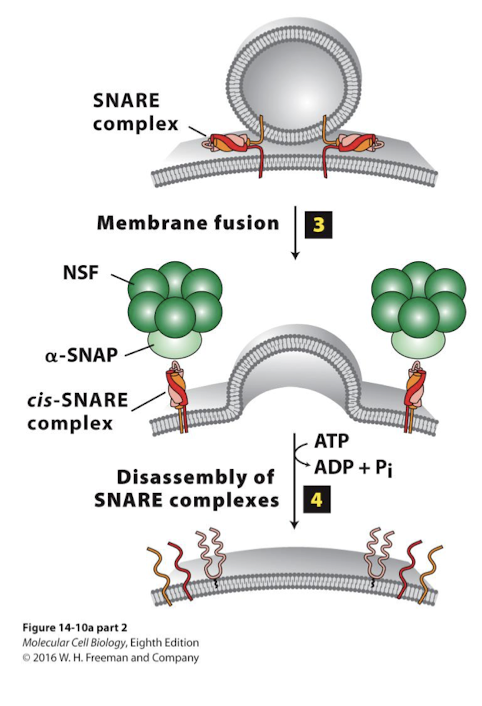
Dissociation of SNARE complexes after fusion
Driven by ATP hydrolysis
Bound SNARE proteins must be freed for use in other fusion events
NSF, a homo-hexamer, with the aid of alpha-SNAP, associates with bundled SNARE complex
Hydrolysis of ATP by NSF drives conformational change to unwind and dissociate the SNARE complex.
How proteins are kept in the ER
Retention of resident molecules that are excluded from transport vesicles
Retrieval of molecules back to the appropriate compartment after escape to or processing by the Golgi
O-linked oligosaccharide
Oligosaccharide chain that is attached to the side-chain hydroxyl group in a serine or threonine residue in a glycoprotein.
Adapter Proteins
Adapter proteins physically link one protein to another protein by binding to both of them. Adapter proteins directly or indirectly (via additional adapters) connect cell-adhesion molecules or adhesion receptors to elements of the cytoskeleton or to intracellular signaling proteins.
Adapter Proteins (AP) complexes
Heterotetrameric protein complexes mediate intracellular membrane trafficking along endocytic and secretory transport pathways. Three different AP complexes are known (AP1, AP2, and AP3), each with four subunits of different, though related, proteins.
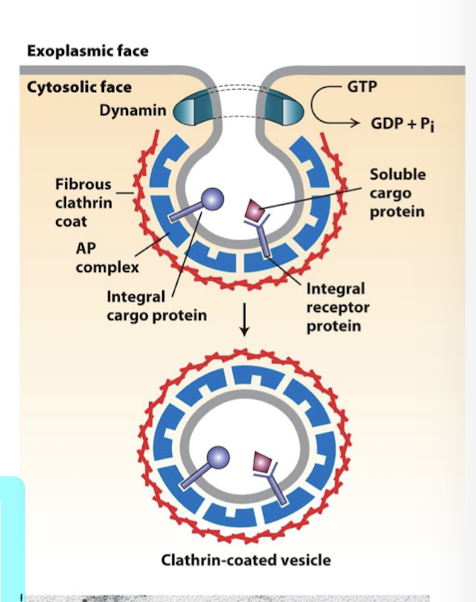
Dynamin
A GTPase that can drive the final step of pinching off. of a clathrin-coated vesicle from a donor membrane.
Another G-protein is required for release of clathrin-coated vesicles
polymerizes around the neck of the budding vesicle
Dynamin GTP hydrolysis causes conformation change that twists the neck, pinching it free of the plasma membrane
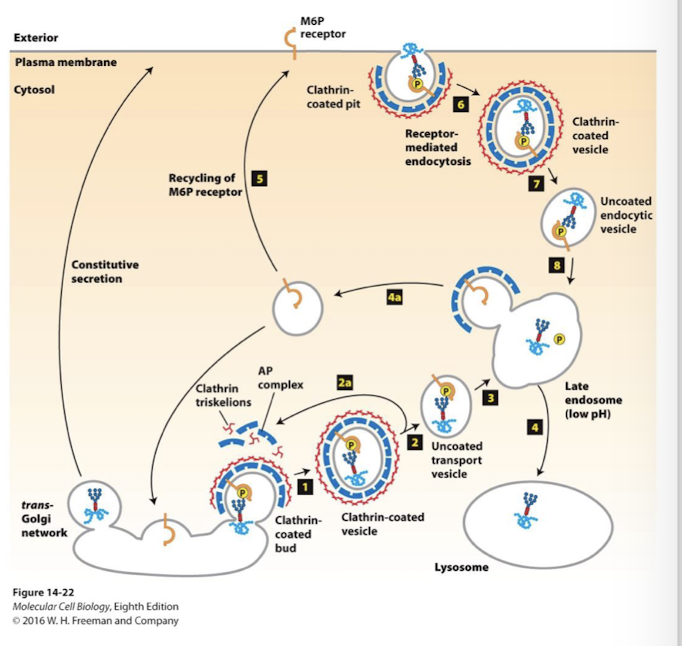
Lysosomal proteins as an example of clathrin-mediated transport
Sorting of lysosomal protein is dictated by modifications to the N-linked glycosylation chain.
In the CGN, soluble lysosomal enzymes are identified by the enzyme GlcNAc phosphotransferase that modifies the core N-linked glycan through addition of one or more mannose sugars phosphorylated on their number 6 carbon, generating mannose-6-phosphate sugars
In the TGN mannose-6-phosphate receptors bind these modified proteins
The receptor is bound by clathrin adapters that concentrate soluble lysosomal proteins in clathrin coated vesicles
Rab/SNARE specificity delivers the vesicles to late endosomes which subsequently fuse with lysosomes
Endosomes, derived by endocytosis at the plasma membrane undergo maturation through the action of V-type pumps causing their pH do drop as they become mature endosomes
pH dictates receptor binds
near neutral pH of the TGN M6P receptors bind cargo tightly
at the lower pH (<6) of late endosomes, the receptor/ cargo complex disassociates
Mannose 6-phosphate (M6P)
A specific modification of N-linked oligosaccharides responsible for sorting of proteins in the trans-Golgi to the lysosome.
Tay-Sachs Disease
Lysosomal storage disorders
inherited neurodegenerative disease
results from deficiency in an enzyme that degrades gangliosides, a major component of neuronal membranes
I-cell disease
Lysosomal storage disorder
defective GlcNAc phosphotransferase leads to nearly non-functional lysosomes.
Endocytosis
General term for uptake of extracellular material by invagination of the plasma membrane; includes receptor-mediated endocytosis, phagocytosis, and pinocytosis.
General term for uptake of extracellular material by invagination of the plasma membrane
Phagocytosis
Process by which relatively large particles (e.g., bacterial cells) are internalized by certain eukaryotic cells in a process that involves extensive remodeling of the actin cytoskeleton; distinct from receptor-mediated endocytosis.
Uptake of large particulate matter- non-specific, does not involve receptors or clathrin vesicles
(phago- eat)
Pinocytosis
aka bulk-phase endocytosis, non-specific uptake of extracellylar fluids that does not involve receptors or clathrin vesicles
(Pino- drink)
Receptor-mediated endocytosis
Uptake of extracellular materials bound to specific cell-surface receptors by invagination of the plasma membrane to form a small membrane-bounded vesicle (early endosome).
Clathrin dependent uptake of specific extracellular ligands following their binding to specific transmembrane receptors
low-density lipoprotein (LDL)
A class of lipoprotein, containing apolipoprotein B-100, that is a primary transporter of cholesterol in the form of cholesteryl esters between tissues, especially to the liver.
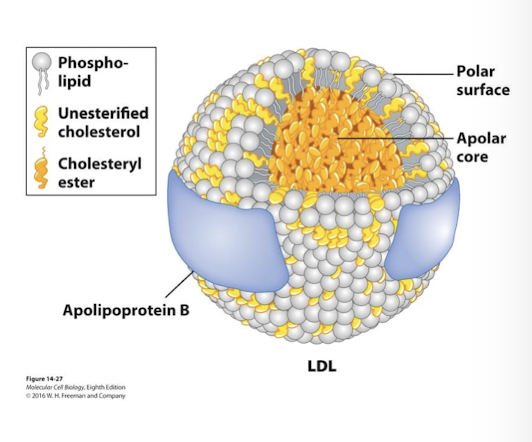
Niemann-Pick disease
Rare recessive disorder cholesterol cannot be transported out of lysosomes leading to neuronal degeneration and early childhood death.
Housekeeping receptors
Responsible for continuous uptake of material necessary for general activities (like LDL receptors, colored red in figure) are recycled back to the membrane for reuse
Transferrin
Protein delivers iron to all growing mammalian cells
Ferrotransferrin- carries fe3+ in blood and has high affinity for the transferrin receptor
The low pH of late endosomes does not promote release of the cargo protein, but does promote release of the bound Fe3+
Empty apotransferrin remains bound to the receptor and is recycled back to cell surface, where at a neutral pH is has low affinity for the receptor released
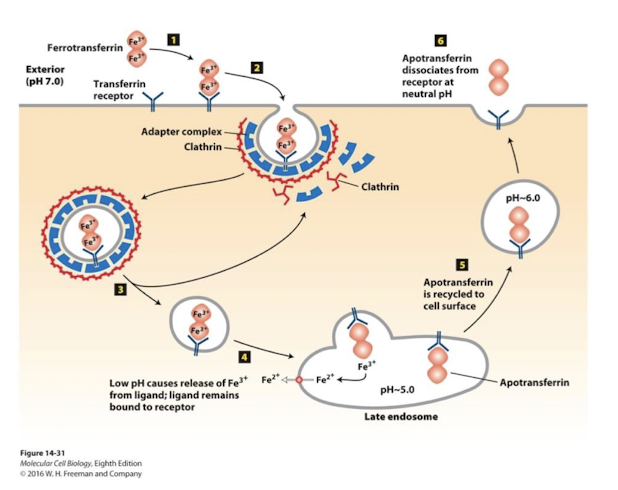
Iron-Response Elelement-Binding proteins (IRE-BP)
4Fe-4S cluster-containing proteins that act as cellular iron sensors. When the cytosolic Fe2+ concentration is too low, the iron in the Fe-S clusters dissociates from the protein and the IRE-BP becomes an active RNA-binding protein that binds to a specific nucleotide sequence within stem-loop secondary structures in the untranslated regions of the ferritin and transferrin receptor mRNAs.
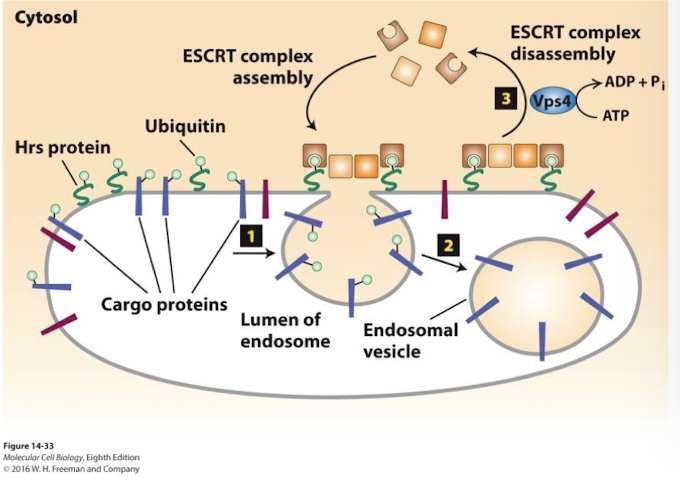
Multivesicular bodies
Such membrane proteins remain associated with the membrane of late endosome as they continue to mature into Multivesicular bodies.
Endosomes that segregate membrane proteins destined for degradation in the lysosome on vesicles that bud into the interior of the endosome.
Hrs
endosome membrane protein directs loading of ubiquitinated membrane cargo proteins into the endosome vesicle buds.
Endosome membrane proteins are protected from degradation because they are not ubiquitinated and incorporated into the internal vesicles.
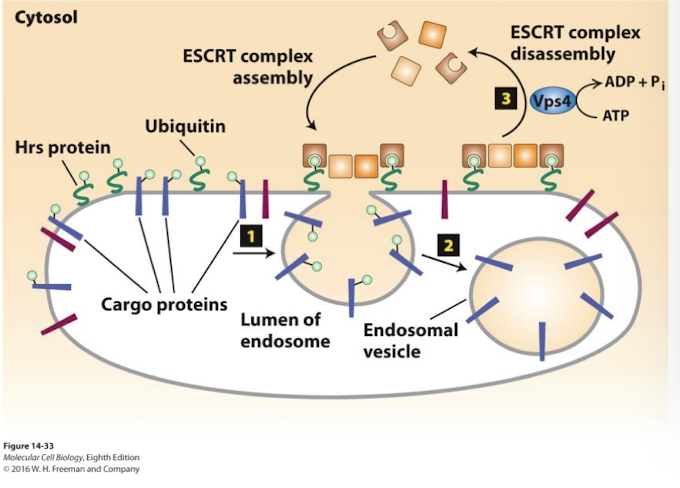
ESCRT
Hrs recruits the cytosolic protein complex ESCRT which drives vesicle budding into the endosome, allowing degradation of these proteins.
Endosome membrane proteins are protected from degradation because they are not ubiquitinated and incorporated into the internal vesicles.
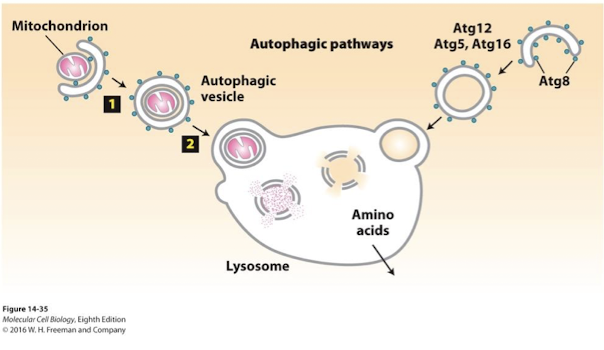
Autophagy
Literally, “eating oneself”; the process by which cytosolic proteins and organelles are delivered to the lysosome, degraded, and recycled. Autophagy involves the formation of a double-membrane vesicle called an autophagosome or autophagic vesicle.