S3.1.4 Properties of metals and non-metals
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
9 Terms
Metals chemical behaviour
Lose electrons to form positive ions
Behave as reducing agents
Form ionic bonds with non-metals
Form basic oxides
Non-metals chemical behaviour
Gain electrons to form negative ions
Behave as oxidizing agent
From covalent bonds with other non-metals
Form acidic oxides
Group 1 metals react..
to form 1+ ions by losing their one valence electron
Group 1 metal properties:
Soft, shiny, can easily be cut
Melting point decreases down group 1, as metallic bonds get weaker
Lithium, sodium, potassium float on water due to low density
How are group 1 metals stored and why?
In oil, to prevent reaction with oxygen in air
What trend do group 1 metals follow for reactivity?
Increases down group
Product of group 1 metal and water reaction
Alkaline solution and hydrogen gas

Product of group 1 metal and halogen reaction
Salts
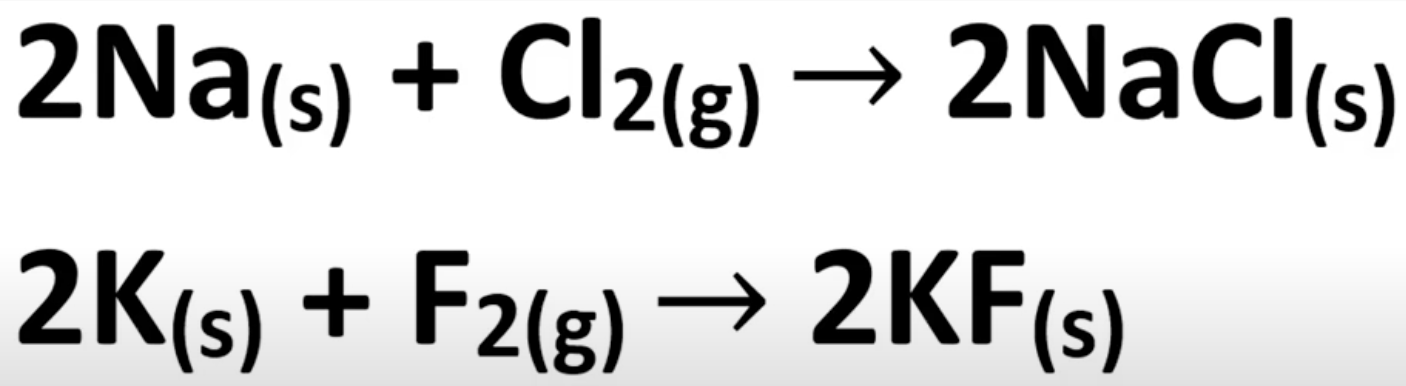
Group 17 halogen properties:
Melting and boiling points increase down group due to higher LDFs