BCEM 393: Nucleic Acids - Structure Quiz 5
1/12
Earn XP
Description and Tags
BCEM 393 UofC
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
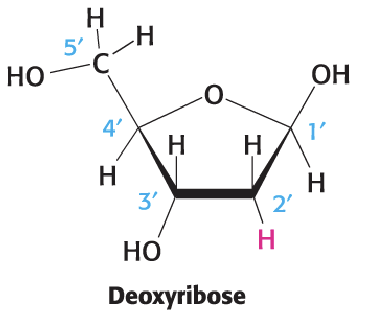
Deoxyribose (4)
Furanose structure
DNA lacks 2’ hydroxyl group; instead has H
In terms of groups going down from 1’: H - H - OH - H
5’ C goes up and has OH on leftside
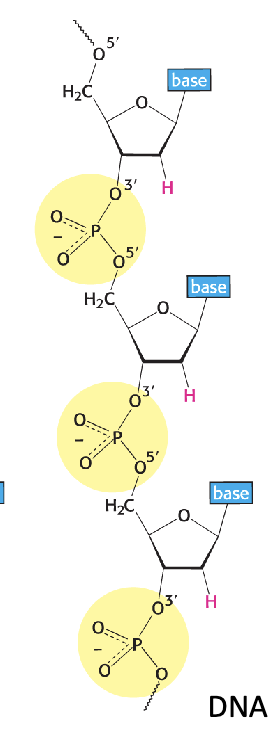
Backbone of DNA (2)
Sugars are linked by phosphodiester bridges between the 3’ OH of one sugar and the 5’ OH of an adjacent sugar
OH groups participate in the bond (become O)
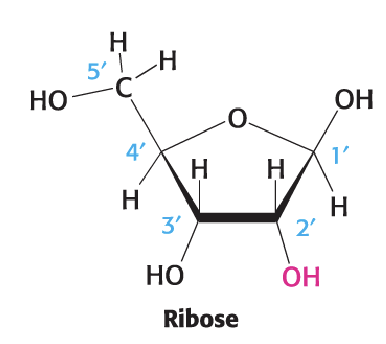
Ribose
Furanose structure
RNA has 2’ hydroxyl group
In terms of groups going down from 1’: H - OH - OH - H
5’ C goes up and has OH on leftside
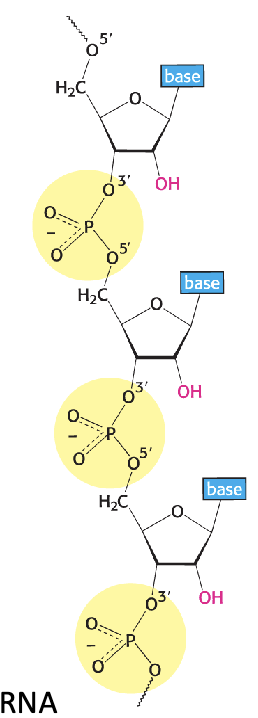
Backbone of RNA (2)
Sugars are linked by phosphodiester bridges between the 3’ OH of one sugar and the 5’ OH of an adjacent sugar
OH groups participate in the bond (become O)
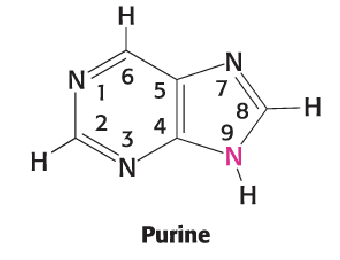
Purine (2)
Common six-membered ring fused to imidazole
Changes to purine bases only occur on the common six-membered ring
4 DBs to draw
Ns at 1, 3, 7, 9
N9 is NH
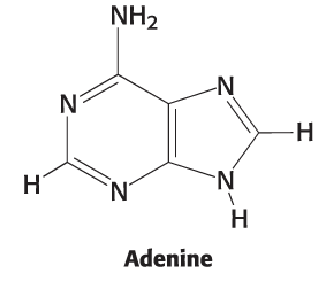
Adenine
NH2 group on position 6
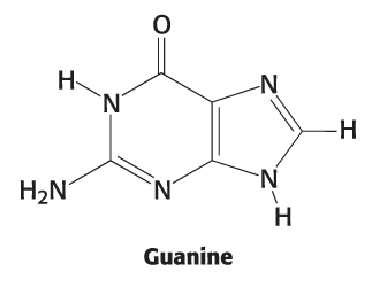
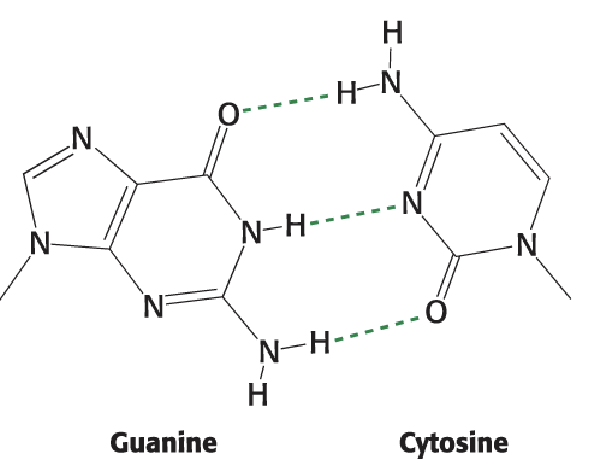
Guanine (3)
Double-bonded O on position 6
Double bond gone between positions 1 and 6 (N becomes NH)
NH2 on position 2
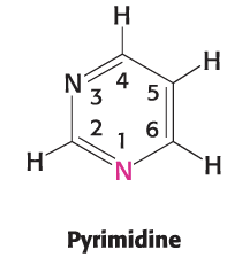
Pyrimidine (2)
Same common six-membered ring
All pyrimidines have NH at position 1 and no double bond between positions 1 and 2
All pyrimidines have double bonded O at position 2
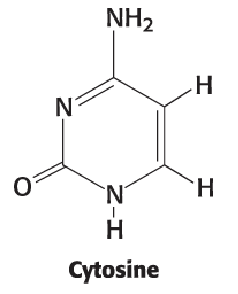
Cytosine (2)
NH2 at position 4
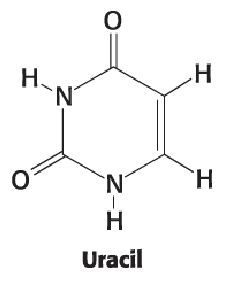
Uracil (3)
No double bond between positions 3 and 4
Double bonded O at position 4
NH2 at position 3
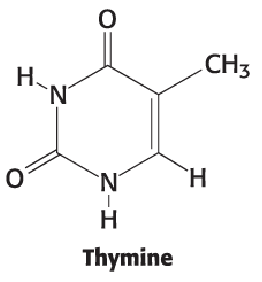
Thymine
Same as uracil but CH3 at position 5
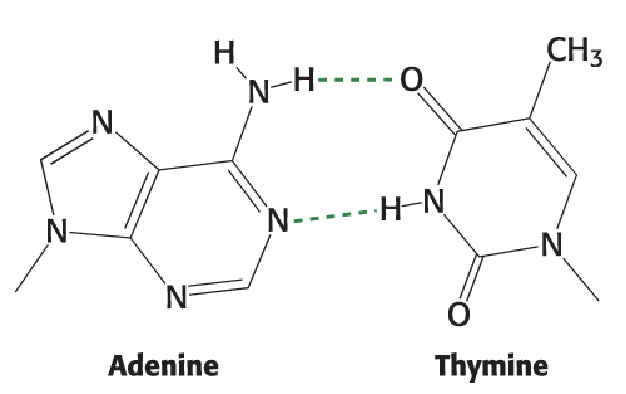
Watson-Crick Base Pairing: GC
Watson-Crick Base Pairing: AT