PreLab Quiz: Experiment 4, Atomic Spectroscopy
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Arrange the colors of the visible spectrum in order from highest to lowest energy.
Highest energy
Lowest energy
violet
blue
green
yellow
orange
red
If an electron transition involves 3.47×10−19 J of energy, what is the wavelength (in nm) of the photon that is emitted?
572 nm
If a photon has a wavelength of 675.0 nm, what is the energy of the photon (in J)?
2.94×10^-19 J
When electricity passes through a gas element in a discharge tube, light is given off. What is the cause of this phenomenon?
Select one:
a. Electricity excites electrons from the element into higher energy states. The electrons then return to the ground state, releasing energy corresponding to the wavelengths of the light seen.
b. The energy from electricity causes an electron to move from the ground state to a lower energy state, emitting a photon of a certain wavelength.
c. Electricity excites electrons from the element into higher energy states. The presence of electrons in these excited states causes the element to emit light.
d. The energy from electricity causes most elements to decompose, emitting light.
a. Electricity excites electrons from the element into higher energy states. The electrons then return to the ground state, releasing energy corresponding to the wavelengths of the light seen.
To use a spectroscope, position the tool to allow light to enter through the
(slit, wavelength scale, diffraction grating)
The light will reflect off the
(diffraction grating, wavelength scale, slit)
which separates it into colors. View the results on the
(wavelength scale, diffraction grating, slit)
slit
diffraction grating
wavelength scale
When an electron moves up to higher energy levels, the atom
(transfers, absorbs, emits)
photon of light whereas the atom
(absorbs, emits, transfers)
photon of light when an electron drops to a lower energy level.
The photons emitted from an atom appear as
(a line spectrum, a continuous spectrum, white light.)
absorbs
emits
a line spectrum
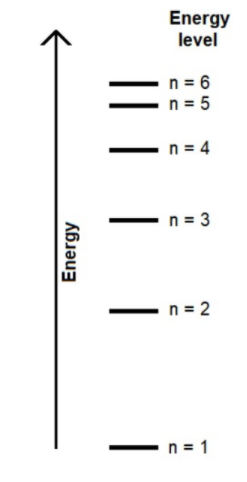
Consider the diagram of the energy levels in a hydrogen atom, determine the most likely electron transition corresponding to each color on a line spectrum.
Each transition will be used once.
Blue line
Red line
Violet line
n=5 to n-2
n=3 to n=2
n=6 to n=2
What needs to be included in the label for each axis of a graph?
Select one or more:
a. Units of quantity displayed on axis
b. Description of how measurement was made
c. Relationship to quantity displayed on other axis
d. Name of quantity displayed on axis
a. Units of quantity displayed on axis
d. Name of quantity displayed on axis