Lecture 2: hormone action
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
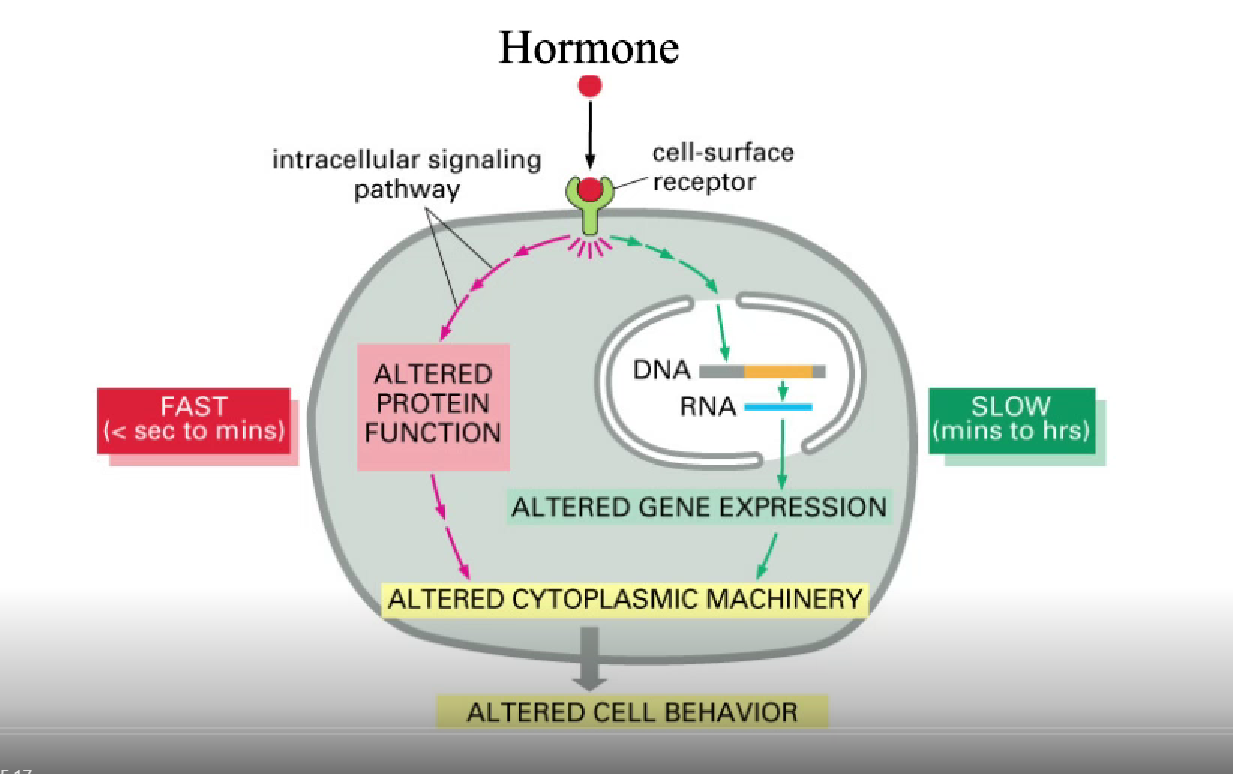
What do hormones do on a molecular level?
Alter protein function or gene transcription
What is the endocrine axis?
cascades in which the target tissue of one hormone is another endocrine gland
Ex: hypothalamo-pituitary-thyroid axis
list the types of hormone receptors
cell surface receptors
intracellular receptors
what causes the creation of a hormone to be slow vs fast?
quick: modification of something (protein) already present in cell
usually phosphorylating it
slow: needs DNA
What are the types of cell surface receptors
receptors with in-built (inherent/intrinsic) tyrosine kinase activity
insulin
EGF1 (growth FACTOR)
receptors that recruit/activate (extrinsic) tyrosine kinase- cytokines
prolactin
leptin
growth HORMONE
g-protein coupled receptors
catecholamines
what is tyrosine kinase?
an enzyme that transfers a phosphate group from ATP into a tyrosine residue in a protein
phosphorylation induces conformational changes
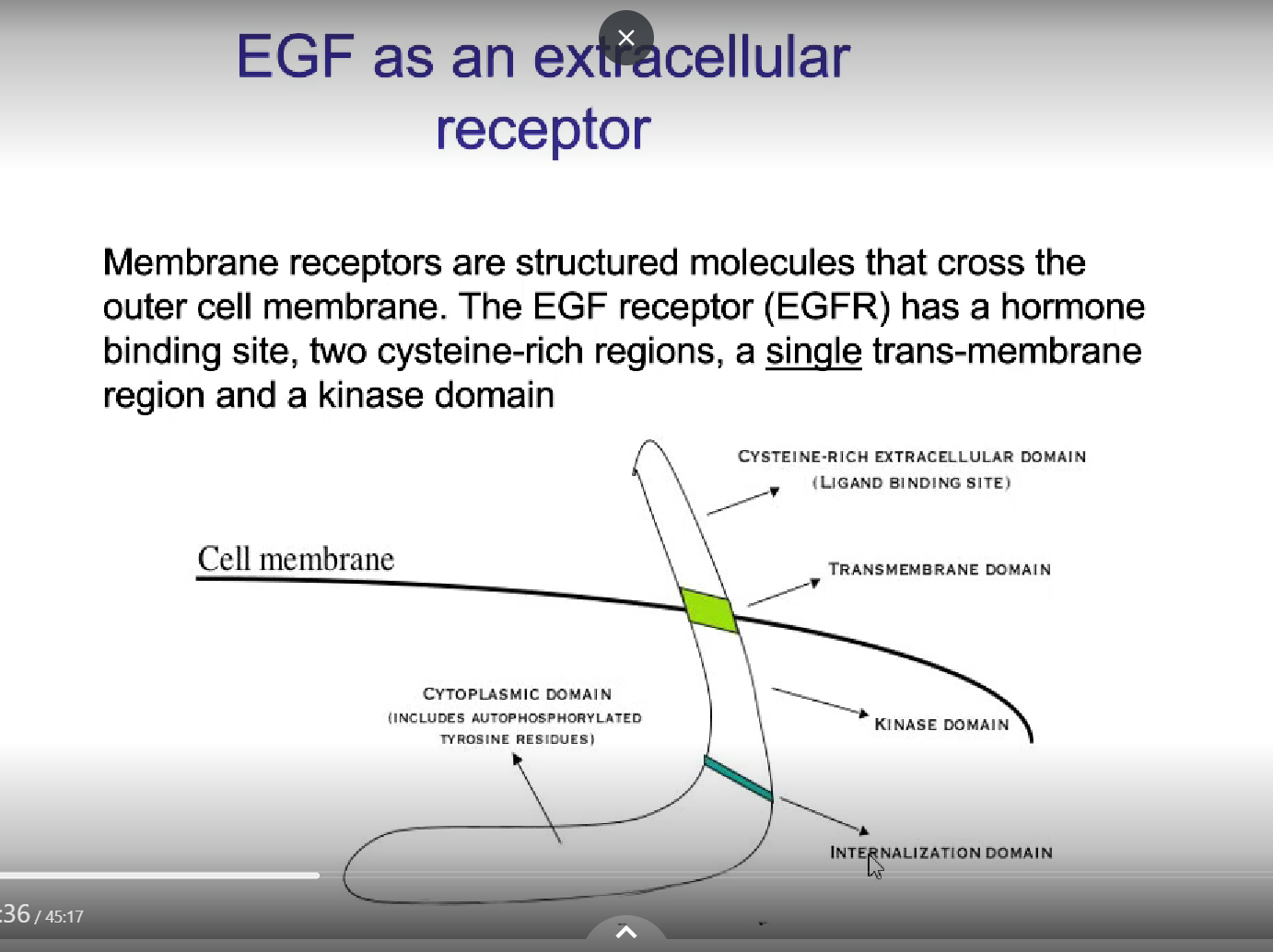
Outline the epidermal growth factor receptor
extracellular receptor w/4 family members (EGF 1-4)
Ligand-induced dimerisation
autocrine, paracrine cell signaling
signal transduction processes: signaling pathway becomes activated by these proteins
Ras
PI 3-kinase
JAK-STAT
HAS INTRINSIC KINASE ACTIVITY
What is autocrine vs paracrine cell signaling
Auto = cell produces a GF to which it also responds
Para = local cell to cell signaling in which a molecule released by one cell acts on a neighbouring target cell
What are membrane receptors
structured molecules that cross the outer cell membrane
outline the structure of the EGF receptor
hormone binding site
2 cysteine-rich regions (for ligand binding)
a SINGLE trans-membrane region
a kinase domain
What is a hormone receptor?
a protein on/in a cell that:
has high affinity (binds well) with the hormone
binds the hormone specifically, identifying it amongst other hormones and molecules
binds the hormone in a reversible manner
is only on specific tissues
is saturable and has a limited number of binding sites (needs a threshold)
does something! mediates a biological response
outline receptor modification: post-translation modification
turning receptor on:
the kinase-linked hormone receptor binds with the hormone
the kinase activity of the receptor phosphorylates the receptor
the signal can enhance itself and go through the cell
turning receptor off:
reverse reaction, phosphatase removes phosphate
INTRINSIC KINASE ACTIVITY
Ras
small GTP-ase. found in two forms:
bound to GDP = inactive
bound to GTP = active
GDP —> GTP needs co-factors
Outline post-translational modification using EGF as an example
hormone binds
EGF receptors dimerise, meaning receptor can become active
conformational change occurs, allowing for kinase activity
kinase activity causes phosphorylation
phosphorylation = activation of the receptor allowing for the…
recruitment of GRB + SOS allows for the exchange of GDP on Ras to GTP
Ras + GTP activates intracellular signaling pathways such as MEK signaling pathway
GRB
adaptor protein
SOS
exchange factor, allows for GDP to change to GTP
how does the post-translational modification of the insulin receptor differ from that of the EGF receptor?
insulin receptors already dimerised
uses PI 3-kinase to get activated
outline recruited tyrosine kinase activity
receptors have no kinase domain built-in
most common pathway = JAK-STAT pathway
same deal: binding to receptor, conformational change
JAK associated w/receptor, phosphorylation allows for…
recruitment of STAT which is also phosphorylated
STAT takes signal to DNA, allowing transcription
only difference is how they get phosphorylated
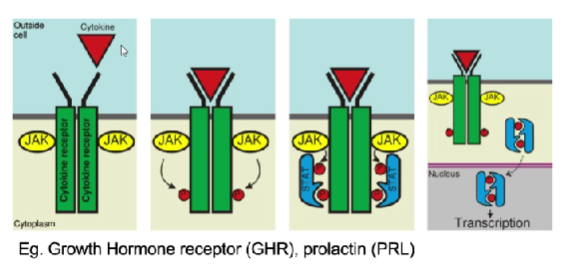
JAK
janus-associated kinase
just a kinase :)
STAT
signal transducer and activator of transcription
a transcription factor
g-protein
a bit like small GTPases
have an inactive and active state
how do g-protein coupled receptors work?
act via SECOND MESSENGER molecules to transfer signal into cell
relies on phosphorylation as well, and calcium flux
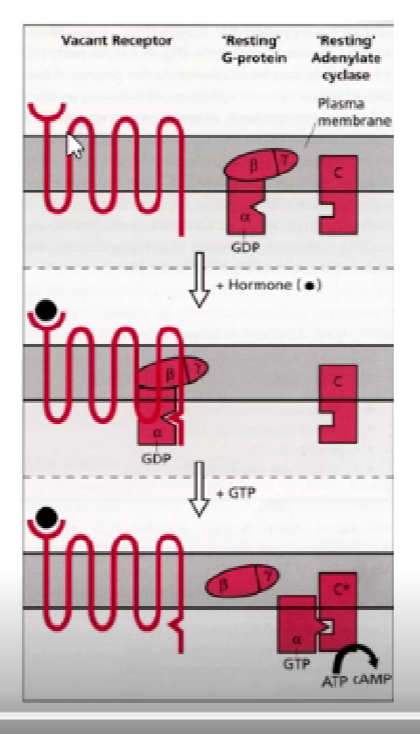
outline the structure of the g-protein
subunits:
1. alpha
beta
gamma
heterotrimeric (made up of 3 subunits)
activation of receptor releases alpha subunit
outline g-coupled receptor signaling
resting g-protein: alpha subunit associated w/GDP (inactive)
hormone binds, conformational change to alpha subunit
change allows for exchange of GDP —> GTP
alpha subunit released and activates 2nd messenger
what do kinases do?
phosphorylate things
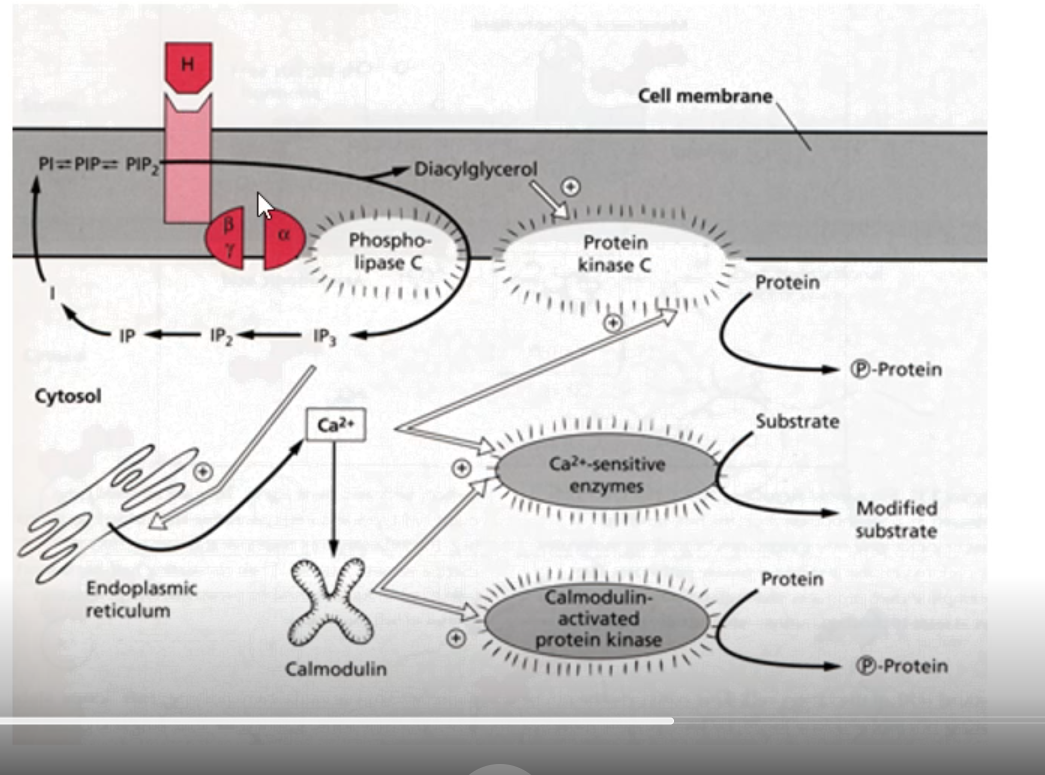
outline g-coupled receptors with reference to diaglycerol (DAG), IP3 and Ca2+
end result = phosphorylated protein or modified substrate
hormone binds, conformational change
alpha subunit released after binding to GTP
DAG as second messenger: activates protein kinase C that phosphorylates a protein
IP3 as a second messenger: works through phospholipase C, enables ER to release calcium stores into the cytoplasm
cytoplasmic conc. of calcium increases
activate calcium-sensitive enzyme (Ca2+ sensitive enzyme) allowing for substrate to be modified to produce a modified substrate
calmodulin = calcium binding protein, sensitive to calcium, calmodulin-activated protein kinase cause protein to be phosphorylated
which type/s of hormones use cell surface receptors?
peptide hormones
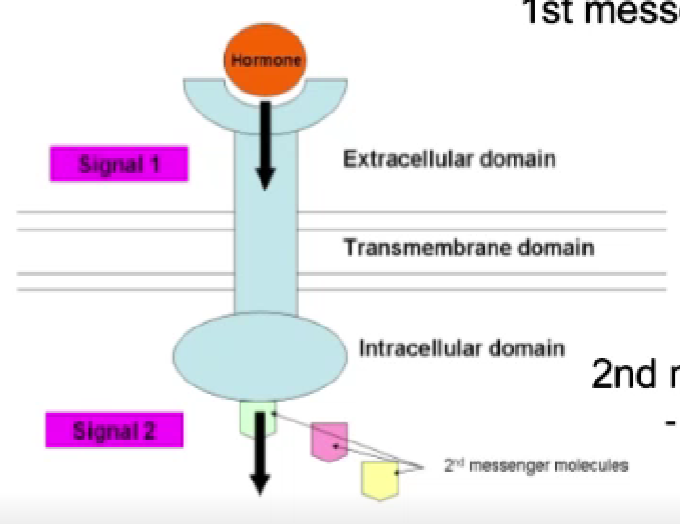
1st vs 2nd messenger
1st: the hormone. signal through cell surface receptor
2nd messenger: interactions between the intracellular domain and other molecules w/in the cell
list the second messengers you should know
PI 3-kinase
JAK-STAT
cAMP
DAG
IP3
give an overview of steroid hormone receptors
ligands = small lipophilic molecules
receptor encoded by a single gene
have an ability to bind to DNA
function as transcription factors
what is the issue with steroid hormones?
lipophilic = hydrophobic
blood = water
cannot move through blood
need protein carriers to move through blood, which makes them insoluble in fat
where are steroid hormone receptors found?
cytoplasm or nucleus
outline the general steroid hormone receptor mechanism
hormone binds
conformational change
receptor goes from cytoplasm to nucleus to bind DNA
when bound to DNA, allows transcription
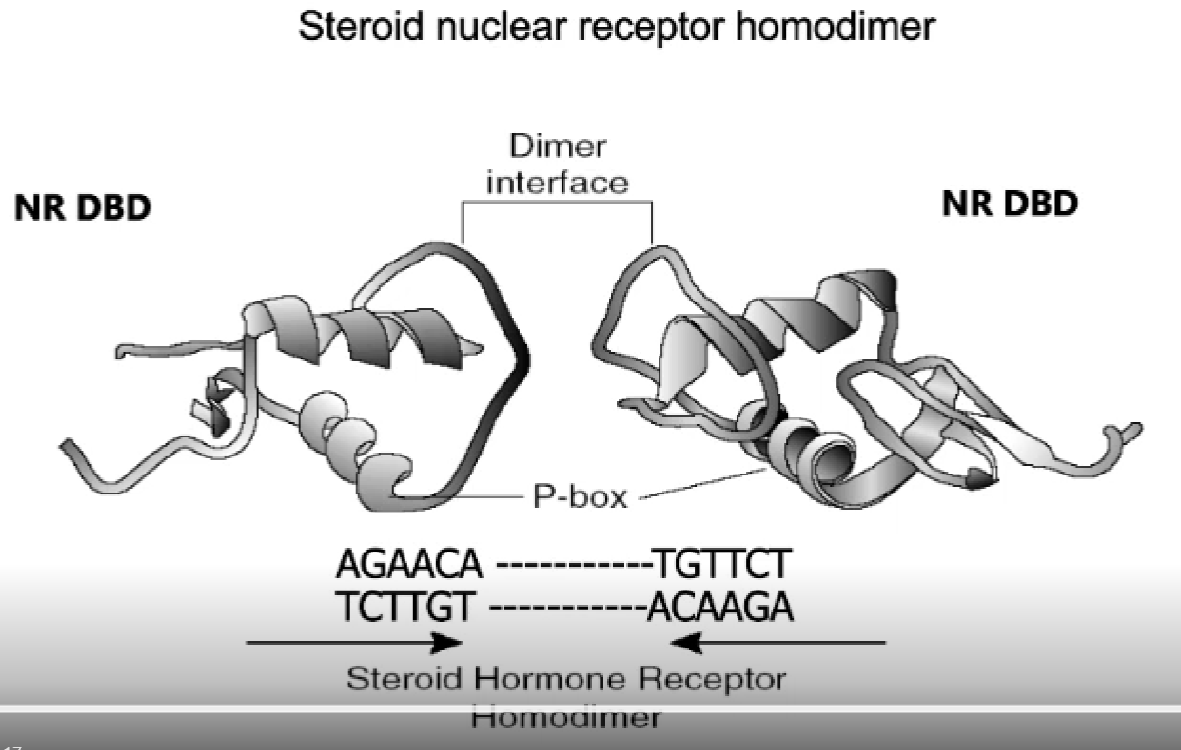
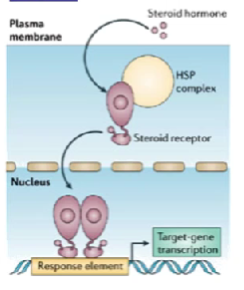
outline type 1 nuclear hormone receptors
homodimers (bind two of the same together, ex 2 glucocorticoid receptors bound together)
found in cytoplasm (usually) and in conjunction with heat-shock proteins (inactive)
usually all steroid receptors
type 1 nuclear hormone receptor mechanism
hormone binds, heat-shock protein drops off and homo-dimerises
protein enters nucleus, binds to DNA
DNA contains hormone response elements (HREs) telling it where to bind
nuclear receptor/DNA complex recruits other proteins to transcribe DNA
new genes expressed, new proteins being made
type 2 nuclear hormone receptors
heterodimers
cytoplasmic or nuclear
all have the same common factor- the retanoid X receptor
usually involve small molecules w/similar properties to steroid hormones
ligand-dependent transcription factors
centrally conserved DNA binding domain, zinc fingers
when hormones bind, AF1+Af2 work together to change cell behaviour
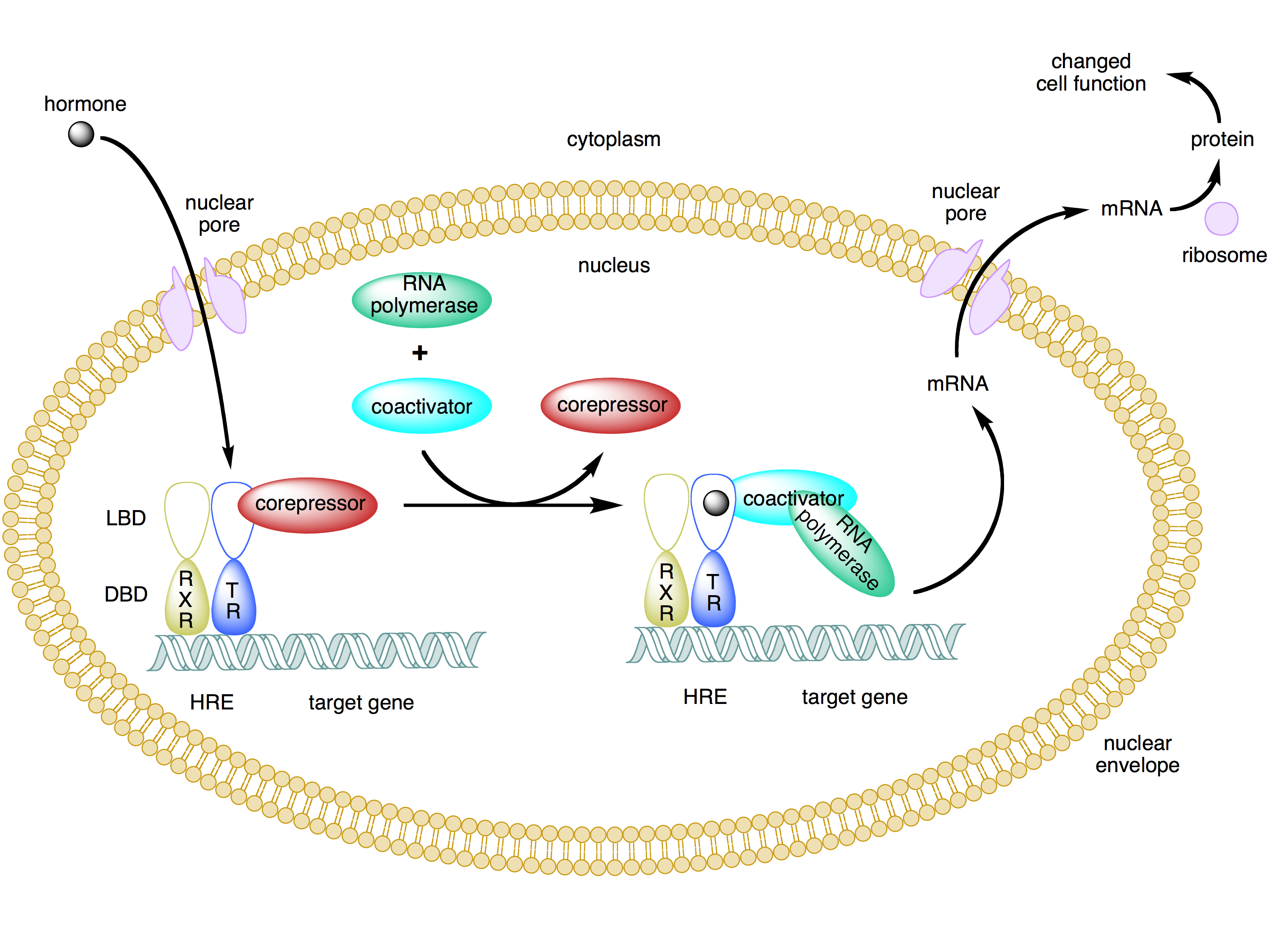
type 2 nuclear hormone receptor mechanism
already hetero-dimerised, already bound to nucleus
inactive: requires repressor proteins to bind
active: hormone binds, co-repressor proteins moved and replaced by co-activators
co-activator complex activates gene, enhances transcription
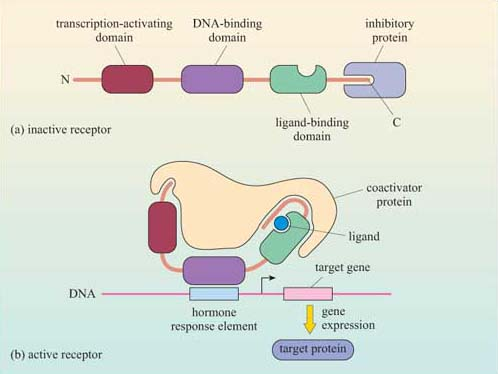
outline the structures of nuclear hormone receptors
3 domains:
transactivation domain
within this domain = AF1
DNA binding domain
localises to nucleus + docks onto DNA
hormone binding domain
within this domain = AF2
transcriptional activator areas- when hormone is at on receptor, always will have a basal level
AFs upregulate transcription/gene expression
outline how hormones change the pattern of gene expression
promoter regions: region of DNA where DNA polymerase attaches + initiates transcription
gene = area of DNA which codes for mRNA
binding via its receptor
hormone response elements
what are hormone response elements
sequences (consensus) different for each of the hormone receptors
located in regulatory regions of target gene
usually 5’, close to core promoter
6bp hexamer core recognition motif
usually 2 half sites- intervening base pairs
what are zinc fingers?
four cysteine residues that hold zinc
what is the structure of HREs?
monomeric
dimeric: palindrome or inverted repeat
direct repeat
inverted palindrome
sequence for each HRE is different for each hormone receptor- glucocorticoid has different HRE to oestrogen, etc
hexamers repeated because you have the two molecules joining onto it (2 half-sites)
how does a receptor recognise a specific HRE?
inside DNA binding area = p-box
inside p-box = zinc fingers + recognition sequences for HREs
p=box is where all the binding happens