IB Chemistry Energetics
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
Which enthalpy change cannot be calculated directly from average bond enthalpy data?
Average bond enthalpies give data for species in the gaseous phase (so cannot be caculated when not gaseous)
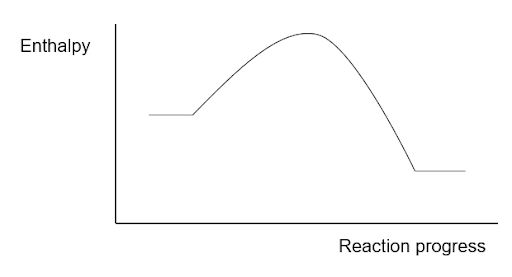
Comment on the average bond enthalpies of the reactants vs the products
The products have a higher total average bond enthalpy than the reactants. The products are at a lower energy level than the reactants meaning that the total average bond enthalpy of formation of bonds in the products is greater than the total average bond enthalpy of breaking of bonds in the reactants.
Define average bond enthalpy
The energy required to break one mole of covalent bonds in the gaseous state under standard conditions to give gaseous atoms
averaged over a range of similar compounds
Define enthalpy change of formation
Enthalpy change when one mole of a compound is formed from its elements in their standard states under standard conditions.
Define standard bond enthalpy
Em