D and F block elements(JEE)
1/87
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
88 Terms
D and F elements belong to ___ group
3-12
outer electronic configuration of D block
(n-1)d¹⁻¹⁰ ns¹⁻²
outer electronic configuration of D block is (n-1)d¹⁻¹⁰ ns¹⁻². what is the exception for this rule?
Pd (4d¹⁰ 5S⁰)
D block has more than one typical metallic structure, what is/are the exception to this?
Zn, Cd, Hg, Mn
why do D block elements have more melting point?
there is more involvement of (n-1)d electrons(covalent bonding) in addition to interatomic metallic bonding
in a graph of MP(melting point) plotted against Z(Atomic number), the curve peaks at dˣ configuration, where x=___. Exceptions for this are?
5, exceptions: Mn, Tc
metal metal bonds are more uncommon in 5d and 4d series compared to 3d series. true or false?
false(more common in 5d and 4d)
for ions of the same charge, radius _______(increases/decreases) with increasing at. no. Why?
decreases, due to increasing nuclear charge
Zr and Hf have similar atomic radius. this is a consequence of ____
lanthanide effect
density significantly _____ from Ti to Cu
increases
ns electrons are lost before (n-1)d electrons. True or false?
true
True or false: differentiating electron enters ns orbital first
false, differentiating electron enters the (n-1)d orbital
factors affecting IE
attraction to nucleus, inter electron repulsion, exchange energy
name the transition metal exhibiting +1 oxidation state
Cu
name the transition metal exhibiting highest oxidation state
Mn(+7)
what are the oxidation states shown by Fe
2, 3, 4, 6
oxidation states exhibited by Scandium
+3
elements showing the largest number of oxidation states have dˣ configuration, where x=_
5
usually lower oxidation states are favored by heaver members of the p block. Is this trend continued in the d block?
no
true or false: Mo(VI) and W(VI) are less stable than Cr(VI)
false(higher oxidation states of d block are more stable in heavier elements)
MoO3(hehe moo) and WO3 are stronger oxidizing agents than CrO3. true or false?
False(since Mo(VI) and W(VI) are more stable than Cr(VI)
higher oxidation states form
fluorides and oxides
why do higher oxidation states form halides?
due to high lattice energy as in the case of CoF3 or high bond enthalpy as in the case of VF5 and CrF6
highest oxidation state of Mn is found in
MnO3F
fluorides are more stable at low oxidation state. true or false?
false
oxygen stabilizes higher oxidation states better than fluorine. true or false?
true
highest Mn fluoride is?
MnF4
highest Mn oxide is?
Mn2O7
magnetic moment is calculated using the formula
√(n(n+2)) BM
catalytic activity of d block is due to?
multiple oxidation states, ability to form complexes
state 4 properties of interstitial compounds
high melting point, hard, retain metallic conductivity, chemically inert
_____ interstitial compounds approach diamond in hardness
boride
alloys are formed when atomic radii are within ___ % of each other
15
lanthanoids have ___ stable oxidation states
1
actinoids have one stable oxidation state. true or false?
false
characteristics of lanthanoids(3)
tarnish rapidly in air, silvery white metals, soft
hardness _____ with increase in atomic number for lanthanoids
increases
the hardest lanthanoid is
lutetium
lanthanoids are coloured in aqueous solutions. the exceptions are:
La, Ce, Gd, Lu
stability of half filled configuration is found in ____ in lanthanoids
Gadolinium
earlier members of the lanthanoid series are similar to ___, higher members are similar to ___
Ca, Al
mischmetal contains ___% lanthanoid
95
TiO is used in manufacture of ___
pigments
MnO2 is used in the manufacture of ____
dry cell batteries
formula for Ziegler catalyst
TiCl₄
Wacker's process uses the catalyst ___
PdCl2
a compound has empty d orbitals yet is coloured, give a reason for this
due to charge transfer
MnO₄¯ has no d electrons, yet is coloured, why?
due to charge transfer phenomenon
how many of the following are interstitial compounds?
TiC, Mn₄N, Fe₃H
3
___ and ___ do not form amalgams
Fe and Pt
Nichrome consists of
Ni, Fe, Cr
german silver consists of
Cu, Ni, Zn
Gun metal consists of
Cu, Sn, Zn
Devarda's alloy consists of
Cu, Al, Zn
Duralumin consists of
Cu, Al, Mn, Mg
how is potassium dichromate prepared
fusion of chromite ore with sodium carbonate or potassium carbonate in excess air to form chromate, which is made to react with conc. H2SO4 to form dichromate
chromite ore
FeO.Cr2O3
colour of dichromate solution
orange red
when alkali is added to dichromate solution, what is the colour change? what happens when it is acidified
orange red to yellow, back to orange red when acidified
how is potassium permanganate prepared
fusion of MnO2 with alkali metal hydroxide and oxidizing agent like KNO3
common oxidation states of actinides
+3 and +4
radioactive lanthanide
Promethium
Actinides form complexes. True or false?
True
highest negative enthalpy of hydration is for
Ni
why doesn't Copper iodide exist
oxidizes I- to I2
shape of Mn2O7
tetrahedral
shape of chromate
tetrahedral
number of metal - oxygen - metal linkages in Mn2O7
1
number of metal - oxygen - metal linkages in dichromate
1
colour of Mn2O7
green
magnetic nature of KMnO4 is _____
diamagnetic with weak temperature dependent paramagnetism
magnetic nature of manganate
paramagnetic
in acidic solution, permanganate converts I- to
I2
in neutral solution, permanganate converts I- to
IO3-
why are permanganate titrations in HCl unsatisfactory
HCl is oxidized to Cl2
magnetic nature of lanthanoid ions
paramagnetic, except for La3+, Ce4+, Yb2+, Lu3+
which contractions is greater? actinoid or lanthanoid
actinoid
________ (actinoid/lanthanoids) take part in bonding more than the other
actinoid(as their 5f orbitals are less burried)
colour of manganate
green
colour of permanganate
purple
difference in structures of permanganate and manganate
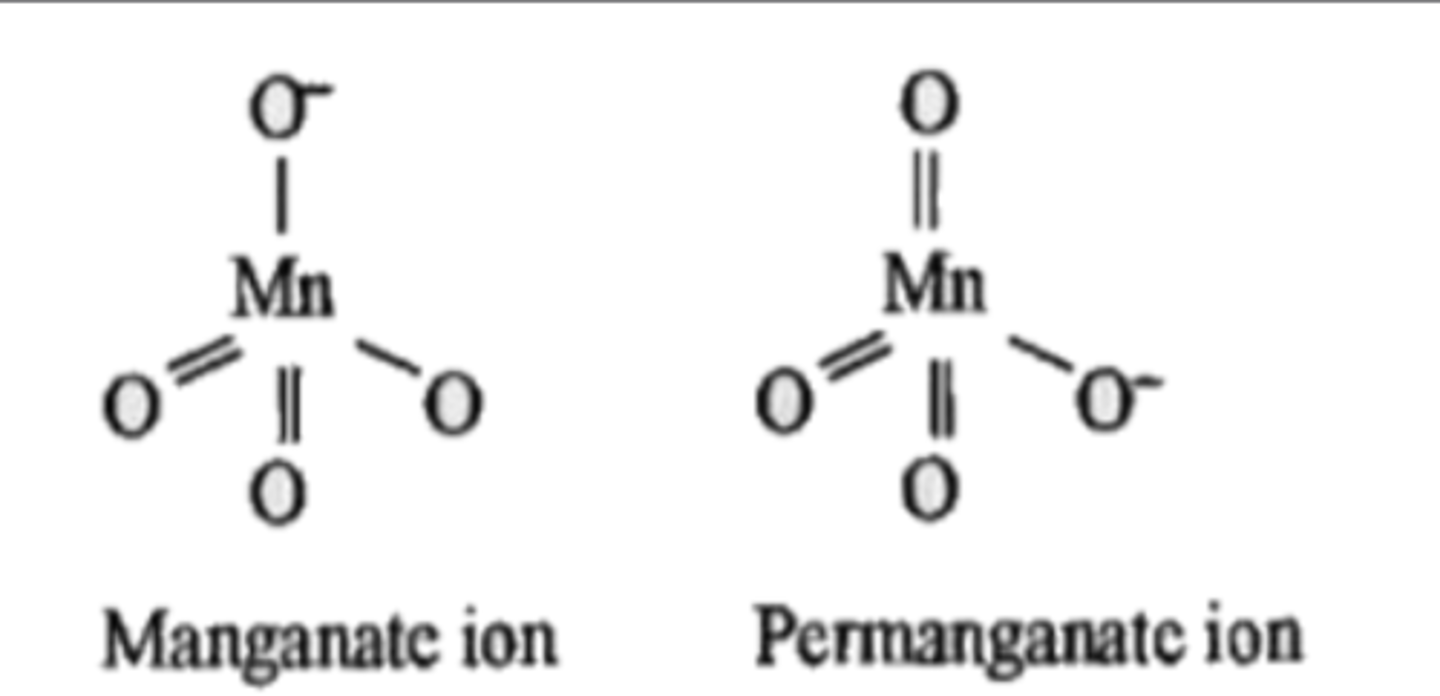
pi bonds in manganate and permanganate are of ___ type
pπ-dπ
FeCr2O4 is fused with Na2CO3 in air, yellow solution is obtained. Formula of this yellow solution is
Na2CrO4
FeCr2O4 is fused with Na2CO3 in air, yellow solution is obtained. On acidification, _____ is formed
Na2Cr2O7
FeCr2O4 is fused with Na2CO3 in air, yellow solution is obtained. On acidification, and on treatment with KCl an orange coloured solution of _______ is obtained
K2Cr2O7
FeCr2O4 is fused with Na2CO3 in air, yellow solution is obtained. On acidification, and on treatment with KCl an orange coloured solution is obtained, which oxidizes Na2SO3 to _______
Na2SO4
heating of KMnO4 gives
K2MnO4
why is La(OH)3 more basic than Lu(OH)3
lanthanide contraction, covalent character increases with decrease in size(Fajan's rule)