Life 207 Pharmacology
1/107
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
108 Terms
Enteral administration
GI-tract
E.g: Oral, rectal, sublingual.
Factors influencing GI absorption
75% drug absorbed in 1-3 hours
- Formulation of drug
- Fed vs fasted, gut pH, gut mobility, gut flora
- Interactions with food
- Drug-drug interactions
When drug is bound to protein vs When it isn't.
Not bound to protein = pharmacologically active = can be absorbed from plasma into tissue/target organ.
Bound to protein = therapeutic drug = stays within plasma compartment.
How does drug protein binding affinity determine the preferred route of administration?
High affinity: IV as high single dose, drug can diffuse out of plasma before binding = good.
Infusion as low continuous dose, most drug bound to plasma = no/poor effect
Why are many drugs designed to be lipophilic/ non-polar?
Help diffusion across membranes.
Means also soluble in other fats - drug can accumulate to toxic levels + become trapped in adipose tissue.
Drugs must be made more polar/hydrophilic for excretion.
Drug metabolism definition
The enzyme-catalysed conversion of a drug into a chemically-distinct product (metabolite).
Main site = Liver.
P450s most important enzymes.
Phase 1 drug metabolism
- Small chemical changes
- Functionalises molecule: adds reactive group/unmasks reactive group
- Small decrease in lipophilicity, small increase in excretion
- Oxidation, reduction, hydrolysis
Phase 2 drug metabolism
- Conjugation reactions: addition of large, often polar/charged, groups.
- Increase water solubility, large decrease lipophilicity, increase excretion, decrease pharmacological effect.
- Glucuronidation, Sulphation, Acetylation, Amino Acid conjugation, Glutathione conjugation
What is an apoprotein?
The site where the drug/substrate binds.
Varies between isoforms.
P450 enzyme cycle
1. Substrate binds
2. Oxygen associates with Fe
3. 1st oxygen forms water with 2H+
4. 2nd oxygen combines with substrate
5. Substrate leaves active site
Flavin monooxygenase (FMO)
- Mediates N + S oxidation reactions
- Requires NADPH
- Generates many metabolites similar to P450: can be difficult to distinguish between the two.
Monoamine Oxidase (MAO)
- MAO-A (Liver, GI tract, placenta)
- MAO-B (Blood platelets)
Both found in neurons + astroglia.
- Vital role in deactivation of endogenous neurotransmitters
- Inhibitors of MAO = antidepressants + caffeine
What is a sign of phase 2 metabolism?
Usually catalysed by a transferase enzyme.
Transfers a polar group from donor/conjugating agent to the phase 1 metabolite.
Parental administration
Non-GI.
E.g: Injection, inhalation, cutaneous (skin), epithelial surfaces (nose + eyes)
Where is the main site of drug absorption?
Small intestine.
Large S.A, high blood flow, bile helps solubilize drugs.
What is Log P?
Indicates lipophilicty of drug.
Want it less than 5.
Are membranes permeable to the ionized form of a drug?
No. Degree of ionization affects concentration available for diffusion.
pKa
pH of an acid/base when it's 50% dissociated.
Weak acids/bases well absorbed, strong acid/bases poorly absorbed.
pH partition theory
Ionisation affects the steady state distribution of drug molecules between two aqueous compartments if a pH difference exists. Weak acids accumulate in compartments of relatively high pH - weak bases in relatively low pH.
3 main proteins that bind to drugs
Albumin (acidic drugs), B-Globulin (some basic drugs) and Acid Glycoprotein (Some basic drugs).
Binding results in reduced excretion, reduced pharmacological effect, and potential displacement of other drugs already bound.
Properties of phase 2 metabolites
More polar, more hydrophillic, more easily excreted.
EXCEPTION = acetylation + methylation, where polar groups are masked.
How does genetic variation effect the therapeutic window?
Ultra-rapid metaboliser = lack of response, doesn't build in system
Poor metaboliser = Adverse effects, drug not cleared quickly
5 types of Adverse Drug Reactions
Type A: Exaggeration of a drug's normal pharmacological actions when given at the usual therapeutic dose
B: Novel responses that are not expected from the known pharmacological actions of the drug
C: Continuing. Persist for a relatively long time after drug withdrawal
D: Delayed. Become apparent some time after the use of a medicine. The timing of these may make them more difficult to detect.
E: End of use. Associated with withdrawal of a medicine.
ST: Chemotherapy
A chemical that binds to & specifically kills microbes or tumour cells.
ST: Antibiotics
Substance produced by a microorganism that in low concentrations inhibits the growth of another microbe.
Bacterial targets include cell wall, altering cell membranes, ribosomes (protein synthesis), biochemical pathways, DNA/RNA synthesis.
ST: Bactericidal vs Bacteriostatic
Bactericidal = Irreversible lethal action on bacteria (e.g. glycopeptides)
Bacteriostatic = Reversibly inhibits growth (e.g. tetracyclines)
ST: What is the MIC?
Minimum Inhibitory Concentration: we want the lowest dilution that inhibits visible growth.
ST: Antimicrobrial susceptibility testing
Determines which antibiotics a pathogen is susceptible to.
ST: What does the ideal antibiotic look like?
- Well tolerated, no toxicity to host
- Low likelihood of developing resistance
- Doesn't induce hypersensitivities
- Relatively long half-life
- Rapid + extensive tissue distribution
- Doesn't interact with other drugs
- Inexpensive, stable, convenient.
ST: Cell wall as bacterial target
Cell wall protects cell from osmotic lysis. Rigidity maintains shape of cell.
β-lactams target cell wall synthesis. Bactericidal.
- Inhibit transpeptidase enzyme (involved synthesis of cell wall)
- Mechanism uses B-lactam ring
- Penicillin acts as steric shield to prevent access of substrate/water to active site.
- Irreversible
- Inhibits bond formation in wall synthesis
ST: β -lactamases
hydrolyse the β-lactam ring, inactivating the β-lactam: creating resistance mechanism which has been rapidly emerging
ST: How can glycopeptides inhibit cell wall synthesis?
They bind directly to the D-Ala-D-Ala terminus of the peptidoglycan pentapeptide chain, preventing transpeptidase from cross-linking.
ONLY ACTIVE against gram-positive bacteria (too large for negative). Narrow spectrum - used for MRSA infections.
ST: What do polymyxins do?
Target the outer bacterial cell membrane.
Interact with phospholipids in cell membrane.
ST: How do aminoglycosides target bacteria?
Induce misreading of genetic code in protein synthesis (codon/anticodon pairing).
Bind to 30S subunit.
Bactericidial, narrow.
ST: How do tetracyclines target bacteria?
Block binding of tRNAs in protein synthesis.
Bind to 30S subunit.
Bacteriostatic, broad.
Avoid in under 8 + pregnancy! Forms salt deposits.
ST: Chloramphenicol / Macrolides/ Ozazolidinoes / Pleuromutilin (bacterial infections)
Bind to 50s ribosomal subunit.
C - Prevents peptide bond formation between AA + peptide chain. Bacteriostatic.
M - Stops polypeptide chain growing. Bacteriostatic.
O - Prevents formation of larger ribosomal complex.
P - Prevents binding of tRNA.
ST: Bacterial Folate Synthesis
Bacteria synthesise their own folic acid (mammals don't)
Sulfamethoxazole + trimethoprim = synergistic effect, respiratory infections.
ST: Fluoroquinolone's
- Inhibit 2 enzymes essential for bacterial DNA synthesis (DNA gyrase + topoisomerase IV)
- Bactericidal
- Examples: Ciprofloxacin, levofloxacin.
ST: Rifamycin's
- Inhibit RNA mechanism
- Halts inhibition of mRNA transcription + translation of polypeptides
- Bactericidal
- Used in tuberculosis
- Induces Cy P450 so can increase metabolism of co-administered drugs
ST: Antimicrobrial resistance
Inability to kill or inhibit the organism with clinically achievable drug concentrations.
ST: What factors accelerate resistance?
- Inadequate levels of antibiotics at site of infection
- Duration of treatment too short
- Overuse/misuse of antibiotics
- Overwhelming no. of organisms
ST: Mechanisms of bacterial resistance
- Inactivation / destruction of antibiotic - Altered binding site
- New binding site
- Bypass metabolic pathways
- Altered permeability
ST: Classifications of bacterial resistance
Intrinsic = e.g. barrier hard to penetrate
Acquired = new mechanisms developed for resistance, new genetic material, Vertical/Horizontal gene transfer
ST: Horizontal vs Vertical gene transfer
Horizontal = Exchange of genes between strains & species.
Vertical = Spontaneous mutations.
ST: Transformation, transduction & conjugation
Transformation = naked DNA uptake
Transduction = bacterial DNA transferred by virus
Conjugation = DNA transferred through cell to cell contact
ST: What qualifies bacteria to be multi-drug resistant?
Resistant to at least one agent in 3 or more classes.
ST: 3 types of TB
1. Drug-sensitive TB:
2. Multidrug-resistant TB
3. Extensively drug-resistant
ST: Mycobacterium tuberculosis
- Lipid rich wall: waxy mycolic acid coating prevents digestion, low permeability prevents drug access = impermeable to many antibiotics
- Slow growth rate compared to most bacteria: this makes them more resistant to antibiotics.
- Spread through air droplets.
ST: Anti TB targets
- Inhibit synthesis of mycolic acid
- Inhibit cell wall synthesis
- Inhibit ATP/RNA synthesis
ST: Drug-sensitive TB treatment
Combo of 4 drugs.
1.) Isoniazid (inhibits mycolic acid), pro-drug, activated by KatG (mutations in this most common, prevents pro-drug conversion)
2.) Rifampicin: inhibits DNA dependent RNA polymerase
3.) Pyrazinamide: pro drug to pyrazinoic acid. highly specific - only active against Mycobacterium tuberculosis. Active only at slightly acidic pH.
4.) Ethambutol. Interrupts cell wall synthesis = increased permeability
ST: MDR-TB treatment
Strains resistant to both isoniazid + rifampicin.
Much longer treatment (20 months)
ST: XDR-TB
Resistant to at least 4 of the core anti-TB drugs.
Resistant to the most powerful - isoniazid + rifampicin + fluroquinolones + at least one of the group 2 parenetals.
ST: How do viruses infect?
Attaches to cells to infect host, genetic material enclosed in capsid.
Viruses will express polypeptide binding sites on envelope or capsid which bind to host surface receptors.
Can be single or double stranded.
DNA viruses tend to replicate in host cell nucleus, whereas RNA viruses tend to replicate in the cytoplasm.
ST: Viral life cycle
Attachment
Viral entry
Uncoating
Replication
Assembly
Release
ST: Targets for anti-viral agents
- Attachment to host cell
- Uncoating
- Synthesis/translation/replication of RNA/DNA
ST: Synthetic nucleoside analogues
- Inhibit DNA/RNA polymerase/ reverse transcriptase
- Or can be incorporated into growing DNA leading to abnormal proteins or breakage
- Pro-drugs, phosphorylated to active form
- E.g. Acyclovir (Herpes)
ST: Interferons
Host cytokines produced by immune system in response to challenge (virus) - good!
Induce intracellular signals, which can inhibit viral penetration/translation/transcription/protein processing/maturation/release.
ST: Retrovirus
RNA virus which synthesises DNA from RNA template.
E.g. HIV
ST: Targets for antiretroviral agents
- Co-receptor antagonists
- Fusion inhibitors: inhibits viral entry
- Reverse transcriptase inhibitors: Either non/ competitively inhibit nucleotide binding to reverse transcriptase + inhibit it. Nucleoside incorporated into DNA, non-nucleoside doesn't.
- Integrase strand transfer inhibitors: inhibits HIV genome integration into host cell chromosome, reserved for resistant cases
- Protease inhibitors: prevents maturation of virions capable of infecting other cells.
ST: Minimising viral resistance
- adherence counselling
- no mono/dual therapy
- ensure optimal serum drug concs
- manage malabsorption
- drug-drug interactions
- stop all drugs at same time
ST: What makes cancer cells?
Activation of proto-oncogenes to oncogenes + Inactivation of tumour suppressor genes.
Genetic changes - proliferate - invade surroundings - blood - differentiate abnormally - display altered cell surface components
ST: types of cancers
Carcinoma: Epithelial cells. 80-90%.
Can be lung, pancreatic, colorectal.
Sarcoma: connective tissue. Bone, muscle, fat, blood vessels, lymph vessels, fibrous tissue.
Haematopoietic: immune system. Leukaemia (WBCS/stem cells) + lymphomas (T or B lymphocytes, hodgkin/non-hodgkin)
ST: Anticancer agents
Cytotoxic
Hormonal
Targeted
Anti-metabolites
Monoclonal antibody
Immuno-oncology (enhance + stimulate immune response)
Cell-cycle specific
ST: Cancer resistance mechanisms
- Increased efflux - reduced drug accumulation
- Insufficient activation of pro-drugs
- Rapid repair of drug-induced lesions
- Overexpression of target enzyme
- Resistant mutations
ST: Pharmacogenomics
Study of genetic basis for the differences between individuals in response to drugs
ST: Pharmacokinetic Variation
Variability in drug concentration at site of action (ADME)
ST: Pharmacodynamic Variation
Variability in individual response to drug (e.g. differences in expression of drug targets)
ST: Are cancers more often Germline or Somatic?
Somatic (90-95%), characteristic tumour cells.
Non-heritable unlike germline.
ST: What are polymorphisms?
Changes to gene sequence.
SNPs most common.
Can affect both pharmacokinetics (changing drug transporters, metabolism, binding) + pharmacodynamics (change drug targets)
ST: 6-Mercaptopurine
- Treat leukaemia
- Metabolised by TPMT, activity varies within population due to germline polymorphisms: Can have 2 functional alleles (normal activity), heterozygous (intermediate activity) or 2 non-functional alleles (Little to no activity)
NL: What is the PNS comprised of?
Autonomic (involuntary) & Somatic (voluntary)
Autonomic = sympathetic & parasympathetic
NL: What are the neurotransmitters in the PNS?
Acetylcholine + Noradrenaline
NL: Somatic nervous system
- Skeletal muscles
- One single axon, no ganglia
- Ach, NICOTINIC Ach receptors
NL: Autonomic nervous system
Pre-ganglionic nerves release Ach to act on nicotinic receptors on post-ganglionic nerve.
Parasympathetic = Post nerves release Ach to act on MUSCARINIC receptors on effector cells.
Sympathetic = Post nerves release Na to act on a + b adrenoreceptors on effector cells
NL: Nicotinic cholinergic synapse (msucle contraction)
Acetyl CoA + Choline = Ach
Ach packaged into vesicles: action potential will release it.
Ach acts on nicotinic receptor, opening ion channel.
Ach terminated by AchEsterase enzyme - choline reclaimed.
NL: Noradrenaline
Tyrosine -> DOPA -> Dopamine -> Noradrenaline
Stored in vesicles. Influx of Calcium ions into nerve ending activates noradrenaline to be released.
Effector cells stimulated by sympathetic nerves have receptors for NA.
NL: Effects of a-adrenoreceptor activation
a1 = contraction of blood vessels, uterus, relaxation of gut, salivary secretion. (noradrenaline)
a2 = inhibition of transmitter release, platelet aggregation, contract vascular smooth muscle, decrease insulin release. (noradrenaline)
NL: effects of b-adrenoreceptors activation
b1 = increase cardiac rate + force
b2= bronchodilation, vasodilation, relax gut (salbutamol)
b3 = lipolysis
antagonist = propranolol
NL: Traditional vs Molecular SAR
Traditional = altering chemical structure of DRUG to change pharmacological response
Molecular = of target macromolecule (protein/receptor/ion/channel/enzyme/DNA)
NL: Myasthenia Gravis
Progressive muscle weakness, antibodies against nicotinic receptors in NMJ.
Treat: Increase Ach (will activate Nic + Mus receptors and cause widespread effects) or mimic its effect at NMJ to strengthen muscle contractions (better, strengthen Nic + weaken Mus ).
NL: Assessing receptor activity + plot magnitude of effect
Compare test compounds to Ach on dose-response curve.
More potent when curve to the left - less substance required to bring about same response.
NL: What is EMR?
A ratio that allows us to compare response of molecules. Less than 1 is more potent than template (e.g. ACh), Greater than 1 is less potent. (So less than 1 is ideal).
EMR = EC50 test compound / EC50 Ach
Anything over 1000 = not active at that receptor. If we remove a group and it causes large increase in EMR - shows its importance / essential to function. Make small gradual changes over time.
NL: What is a rigid analogue?
Addition of a ring structure to lock the molecule in place.
No flexibility within 3D space.
NL: How many molecules of Ach to activate nicotinic receptor?
2
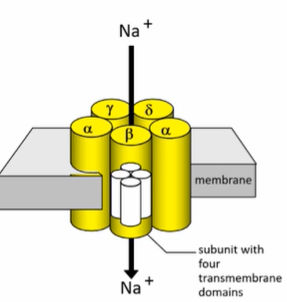
NL: Nicotinic Acetylcholine Receptor Structure
- 5 subunits (2 alpha)
- Gating mechanism for Na+ to travel through
- Ach binds quite far from cell surface: doesn't need to get all the way in.
- Network of H-bonds
- A.A's aren't negatively charged: 2 Tryp, 4 Tyr, 2 Cys
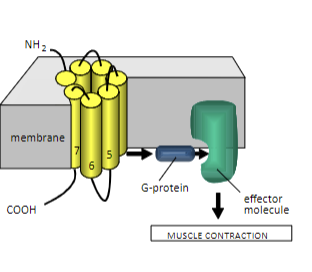
NL: Muscarinic Acetylcholine Receptor
- 7-pass-transmembrane receptor
- Uses intracellular signalling + secondary messengers
- Ach binding site INSIDE cell membrane
- Network of H-bonds
- Ionic bonds: 2 Thre, 4 Tyr, 1 Asp
NL: How does DDF compete with Ach?
Competes for binding site
Under UV light, induces cross-linking + covalent bonds in DDF
NL: Muscarinic Ach receptor pathway
Ach binds to transmembrane domains -> conformational change -> activates G protein -> activates effector molecule for muscle contraction.
NL: What are the 2 types of acetylcholinesterase?
'True version' : Found at nerve endings + skeletal muscle.
'Pseudo version' : Found in plasma, liver + intestine.
Both hydrolyse Ach, but true version has highest affinity for Ach.
NL: Acetylcholinesterase structure
Anionic Site:
-Tryptophan residue
-Substrate recognition
- Helps position the acetyl group of ACh correctly for catalysis at the esteratic site.
- Attracts positive charged molecules
- Not ionic interaction: Cation-pi bond (much stronger!)
Esteratic site:
- Hydrolysis of Ach to acetate + choline
- Terminates neurotransmitter signal
- SERINE residue acts as nucleophile, also histidine + glutamate residues.
- Breaks covalent bond
- Rapid + efficient
NL: 3 different fates at acetylcholinesterase
Reversible: only interacts/blocks anionic site, block Ach from binding to it and breaking down.
Time-dependent irreversible: Molecule degraded so slowly, stays around for years until its broken down.
Irreversible: Organophosphorus compounds. Bind to serine residue on esteratic site irreversibly. Ach can't be broken down. Can result in death. Novichok agents.
ST: Difference between fungi and bacteria
Fungi are:
Eukaryotic (nucelus) + multicellular
Have organelles (mitochondria + ER)
Sterols
Chitin cell wall
Bigger
ST: What is the cell wall of bacteria composed of?
Peptidoglycans
ST: What is the name for fungal infections?
Mycoses
ST: Types of fungus
Candida albicans (most common, white)
Cryptococcus (invasive, infection in those immunocompromised)
Aspergillus fumigatus (airborne spores, inhaled daily but causes problems in those with weakened immune systems)
ST: Are fungi harder or easier to quantify than bacteria?
Harder - they grow more slowly + in multi-cellular forms.
ST: Why is there increased incidence of fungal infections?
Irresponsible use of antibiotics.
Antibiotics can’t treat fungi - instead just upset microbiome + encourage fungi to grow. i.e. HIV patients, due to lowered immune system.
ST: Fungal targets
Inhibit cell wall
Inhibit mitosis
Interfere with DNA synthesis
Disrupt cell membrane
ST: Treating fungi with Azoles
Inhibit ergosterol synthesis
Disrupts cell membrane structure + function, inhibits growth
Inhibits CYP450
Broad spectrum
Example = Fluconazole (fungal meningitis, HIV skin lesions)
St: Treating fungi with Terbinafines
Inhibits ergosterol + lanosterol synthesis
Structural analogue of squalene: accumulation of toxic amounts of squalene = death of fungal cell
Example = Athlete’s foot, fungal nail infections