Chapter 3: Water and Life
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Study the water molecules at the right. On the central molecule, label oxygen (O) and hydrogen (H).
The oxygen is in the center (red), with hydrogen on either side (white).
What is a polar molecule? Why is water considered polar?
Since oxygen is more electronegative than hydrogen, the electrons of the covalent bonds between them are more strongly
attracted to oxygen's than hydrogen's nucleus. This unequal distribution of electrons makes water a polar molecule,
meaning that the two ends of the molecule have opposite charges.
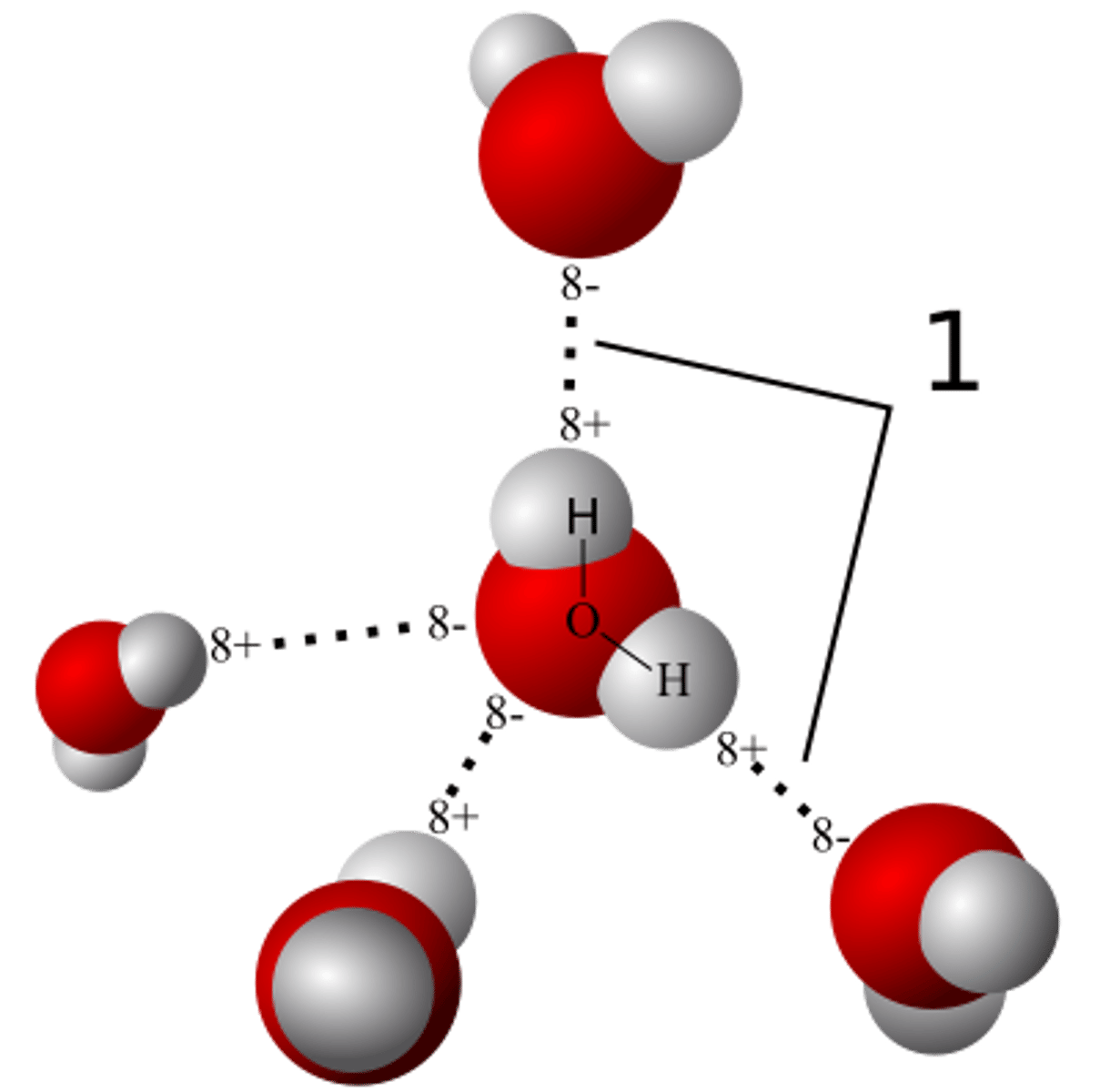
Add + and - signs to indicate the charged regions of each molecule. Indicate the hydrogen bonds.
The oxygen region of each molecule has a partial negative charge (-), and each hydrogen has a partial positive charge (+).
Explain hydrogen bonding. How many hydrogen bonds can a single water molecule form?
Hydrogen bonding occurs when the slightly positive hydrogen of one molecule is attracted to the slightly negative oxygen of a nearby molecule, holding them together. Hydrogen bonds are fragile and at any given moment a substantial percentage of all the water molecules are hydrogen-bonded to their neighbors. Each water molecule can form 4 hydrogen bonds.
Distinguish between cohesion and adhesion.
Cohesion is the phenomenon of the collective hydrogen bonds holding water together. Adhesion, however, is the clinging of one substance to another.
Which is demonstrated when you see beads of water on a waxed car hood?
Adhesion: water molecules attached to other water molecules to form a bead.
Which property explains the ability of a water strider to walk on water?
The point at which the surface of the water interacts with air is an ordered molecular arrangement, causing the water to
behave as though coated with an invisible film. This is attributable to water's high surface tension.
The calorie is a unit of heat. Define Calorie.
amount of heat needed to raise the temperature of 1g of water by 1ºC and also the amount of heat
released by 1g of water when it cools by 1ºC. One calorie is equal to 4.184J.
Water has high specific heat. What does this mean? How does water's specific heat compare to alcohol's specific heat?
The specific heat of a substance is defined as the amount of heat that must be absorbed or lost for 1 g of that substance to change its temperature by 1°C. Water's specific heat is unusually high at 1 cal/g•°C, whereas alcohol's specific heat is is 0.6 cal/g•°C.
Explain how hydrogen bonding contributes to water's high specific heat.
Heat is absorbed and released when hydrogen bonds form; therefore, a calorie of heat causes a relatively small change in the temperature of water because much of the heat is used to disrupt hydrogen bonds before water molecules can begin moving faster. Additionally, when the temperature of water drops slightly, many additional hydrogen bonds form, releasing a considerable amount of energy in the form of heat
Summarize how water's high specific heat contributes to the moderation of temperature. How is this property important to life?
With a high specific heat, a large body of water can absorb and store a huge amount of heat from the sun in the daytime and during summer while warming up only a few degrees. As this water generally cools during
the night and during winter, it warms the air, contributing to milder climates in coastal regions.
Define evaporation. Explain at least three effects of this property on living
organisms.
Evaporation is the transformation of a liquid to a gas. The evaporation of water from leaves of a plant helps keep the tissues in the leaves from becoming too warm in the sunlight. The evaporation of sweat from human skin dissipates body heat and helps prevent overheating on a hot day or as a result of excess heat created by
strenuous activity.
What is heat of vaporization? Explain at least three effects of this property on living
organisms.
quantity of heat a liquid must absorb for 1 g of it to be converted from the liquid to the gaseous state. The heat of vaporization accounts for the severity of steam burns, burns caused by
the heat energy released when steam condenses into liquid on the skin.
Ice floats! So what? Consider what would happen if ponds and other bodies of water accumulated ice at the bottom. Describe why this property of water is important.
If ice sank, eventually ponds, lakes, and oceans would freeze solid and only a few inches at the top would thaw during the summer. Because ice floats, the upper frozen layer insulates the liquid water below, keeping it from freezing and allowing life to exist beneath the surface. Additionally, the frozen layer provides a solid
habitat for some animals, such as polar bears and seals.
Now, explain why ice floats. Why is 4°C the critical temperature?
As a result of hydrogen bonding, water expands as it solidifies. At 4°C water acts like other liquids, expanding as it warms and contracting as it cools. However, below 4°C water begins to freeze as more and more hydrogen molecules are moving to slowly to break the hydrogen bonds. Hydrogen bonds keep the molecules far enough apart to make ice about 10% less dense than water at 4°C; it is this lower density of ice
that allows it to float.
solvent
the dissolving agent of a solution
solution
a liquid that is a completely homogenous mixture of two or more substances
solute
the substance dissolved by the solvent (solid)
Consider coffee to which you have added sugar. Which of these is the solvent? Which is the solute?
Coffee is the solvent and sugar is the solute.
Explain why water is such a fine solvent
traced back to the polarity of the water molecule. Water can dissolve ionic compounds, many compounds made up of nonionic polar molecules, as well as molecules such as proteins if they have polar and ionic regions on their surface.
Distinguish between hydrophobic and hydrophilic substances. Give an example of each.
Any substance that has likes water is said to be hydrophilic, whereas substances that are nonionic and nonpolar and cannot form hydrogen bonds and therefore seem to repel water and are called hydrophobic. An example of a hydrophilic substance is cotton, whereas an example of a hydrophobic substance is vegetable oil.
You already know that some materials, such as olive oil, will not dissolve in water. In fact, oil will float on top of water. Explain this property in terms of hydrogen bonding.
Oil molecules cannot form hydrogen bonds with water and thus behave hydrophobically, because there is a prevalence of relatively nonpolar covalent bonds—in this case, bonds between the carbon and hydrogen,
which share electrons almost equally.
Now, let's do a little work that will enable you to prepare solutions. Read the section on solute concentrations carefully, and show the calculations here for preparing a 1-molar solution of sucrose. Steps to help you do this follow. The first step is done for you. Fill in the rest.
Can you prepare 1 liter of a 0.5-molar glucose solution? Show your work here.
Define molarity.
number of moles of solute per liter of solution—is the unit of concentration most often used by biologists for aqueous solutions.
What two ions form when water dissociates?
hydronium (H3O+) and hydroxide ions (OH-) (we will represent the hydronium ion as H+)
What is the concentration of each ion in pure water at 25°C?
10-7 M (at 25°C)
Water has a pH of 7. pH is defined as the negative log of the hydrogen ion concentration [H+]. Can you
now see how water is assigned a pH of 7?
Although this question is rhetorical, the concentration of [H+] = 10-7
To go a step further, the product of H+ and OH- concentrations is constant at 10-14.
[H+] [OH-] = 10-14
Water, which is neutral with a pH of 7, has an equal number of H+ and OH- ions
Define Acid.
a substance that increases the hydrogen ion concentration of a solution
Define base.
a substance that reduces the hydrogen ion concentration of a solution
Because the pH scale is logarithmic, each numerical change represents a 10X change in ion concentration.
a. How many times more acidic is a pH of 3 compared to a pH of 5?
10^2 = 100
Because the pH scale is logarithmic, each numerical change represents a 10X change in ion concentration.
b. How many times more basic is a pH of 12 compared to a pH of 8?
10^4 = 10,000
Explain the difference between a pH of 8 and a pH of 12 in terms of H+ concentration.
The H+ concentration of a pH 12 is 104 times greater than that of a pH of 8.
On the pH chart, label pH 1-14. Label neutral, acid, and base. Indicate the locations of pure water, urine, gastric juice, and bleach.
Neutral is located at pH 7. Acidity increases from 7 to 0. Baseness increases from 7 to 14. Pure water has a
pH of 7, urine has a pH of 6, gastric juice has a pH of 2, and bleach has a pH of 13.

Even a slight change in pH can be harmful! How do buffers moderate pH change?
A buffer is a substance that minimizes changes in the concentration of H+ and OH- in a solution. It does so by accepting hydrogen ions from the solution when they are in excess and donating hydrogen ions to the solution when they have been depleted. Most buffer solutions contain a weak acid and its corresponding base, which combine reversibly with hydrogen ions.
Exercise will result in the production of CO2, which will acidify the blood. Explain the buffering system that minimizes blood pH changes.
Carbonic acid (H2CO3) is formed when CO2 reacts with water in the blood plasma. Carbonic acid dissociates to yield a bicarbonate ion (HCO3 -) and a hydrogen ion (H+). The chemical equilibrium between carbonic acid and bicarbonate acts as a pH regulator, the reaction shifting from left or right as other processes in the solution add or remove hydrogen ions.
Acid precipitation is increasing. Explain its sources.
Many threats to water quality have been posed ny human activities. The burning of fossil fuels releases huge amounts of CO2, sulfer and nitrous oxides which then chemically reacts with water in the are and alters the delicate balance of conditions for life on Earth- due to affecting water pH and temperature. Electric power plants that burn coal produce more oxides and wind carry the pollutants away giving acid raid the chance to fall more km away from industrial sites.
Discuss how CO2 emissions affect marine life and ecosystems.
About half the CO2 emitted stays in atmosphere, acting like a reflective blanket over the planet that traps heat by preventing it from escaping into outer space. While photosynthesizing organisms absorb some of the gas, approximately 30% is taken up by the oceans, and excessive CO2 uptake might damage the fragile marine ecosystem. CO2 dissolves in sea water creating carbonic acid which then seperates to bicarbonate and carbonate. Sea water acidifies due to the extra protons which lowers the pH of the ocean. Ocean acidification decreases the carbonate concentration, which is required for calcification by many marine organisms. Additionally, CO2 emissions create acid precipitation that damages life in streams and lakes and adversely affects life on land by altering soil chemistry.