Topic 2 - States of Matter and Mixtures
1/179
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
180 Terms
Which state of matter has the strongest forces of attraction between particles?
Solid.
Which state of matter has the weakest forces of attraction between particles?
Gas.
Which state of matter has the highest density?
Solid.
Which state of matter has the lowest density?
Gas.
Describe the arrangement of solid particles.
Regular, very close, fixed positions and lattice structure.
Describe the arrangement of liquid particles.
Irregular, random and quite close.
Describe the arrangement of gas particles.
Irregular, random and far apart.
Describe the movement of solid, liquid and gas particles.
Solids vibrate in fixed positions.
Liquids slide around eachother.
Gases move in random directions at random speeds.
Compare the relative energies of particles in solids, liquids and gases.
Particles in a solid have the least amount of energy and particles in a gas have the most energy.
Which states of matter can be compressed? Why?
Only gas as the particles are in a gas are far apart so can be pushed closer together. However, the particles in a solid and liquid are already close together so cannot be pushed closer together.
Which states of matter can take the shape of the container?
Liquids and gases.
Compare the amount of energy of particles in solids and gases.
Solids have the least amount of energy whereas gases have the most amount of energy.
Describe the difference in movement between particles when they are in a liquid state is compared to a solid state.
In a liquid state, particles are constantly moving but in solid particles they vibrate in fixed positions.
Describe the difference in arrangement between particles when they are in a liquid state is compared to a solid state.
In a solid state, they are regularly arranged, very close together, fixed positions and lattice structure. In a liquid state, they are randomly arranged and further apart.
What does interconversion of state mean?
When matter changes from one state to another due to changes in temperature or pressure.
What type of energy allows particles to move?
Kinetic energy.

D.
Explain why changes of state are called physical changes rather than chemical changes.
Physical changes do not result in the formation of a new substance and are easily reversible.
The forces between particles are changed rather than the chemical properties of the substance.
What is melting?
Solid to Liquid.
What is evaporation/boiling?
Liquid to Gas.
What is freezing?
Liquid to Solid.
What is condensation?
Gas to Liquid.
What is deposition?
Gas to Solid.
What is sublimation?
Solid to Gas.
What is it called when a solid becomes a gas?
Sublimation.
What is it called when a gas becomes a solid?
Deposition.
What is it called when a liquid becomes a solid?
Freezing.
What is it called when a solid becomes a liquid?
Melting.
What is it called when a liquid becomes a gas?
Evaporation.
What is it called when a gas becomes a liquid?
Condensation.
If a substance is below its melting point, what state of matter is it?
Solid.
If a substance is above its melting point and below its boiling point, what state of matter is it?
Liquid.
If a substance is above its boiling point, what state of matter is it?
Gas.
The ___ and ___ points of a substance are the same.
Melting and freezing point.
Are the melting and freezing point of a substance the same or different?
Same.
What happens to the forces of attraction when a solid turns into a liquid?
Overcome or broken.
State what is meant by the term melting point.
The temperature at which a solid turns into a liquid.
State what is meant by the term boiling point.
The temperature at which a liquid turns into a gas.
Suggest what apparatus a scientist could use to measure the melting point of a substance.
Thermometer and water bath.
Describe what happens when a solid is heated and melts into a liquid.
When heated the particles absorb thermal energy which is converted into kinetic energy.
The particles in the solid vibrate more, causing the solid to expand until the structure breaks and becomes a liquid.
The freezing point of water is 0°C. Describe how the movement and arrangement of water particles changes when water is cooled from 10°C to -10°C.
The molecules become close together, lose energy and slow down.
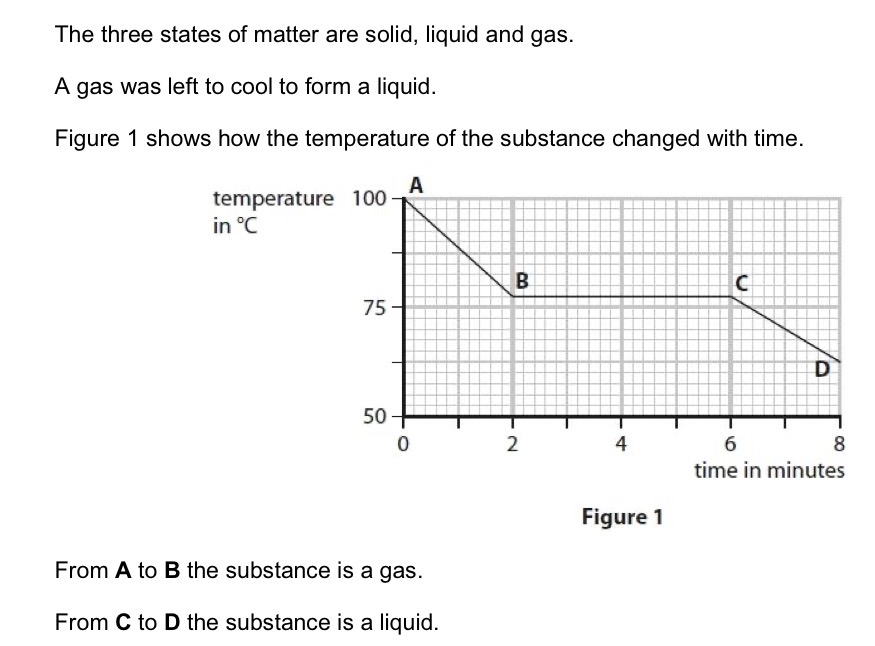
State the time at which the gas first started to form a liquid.
B.
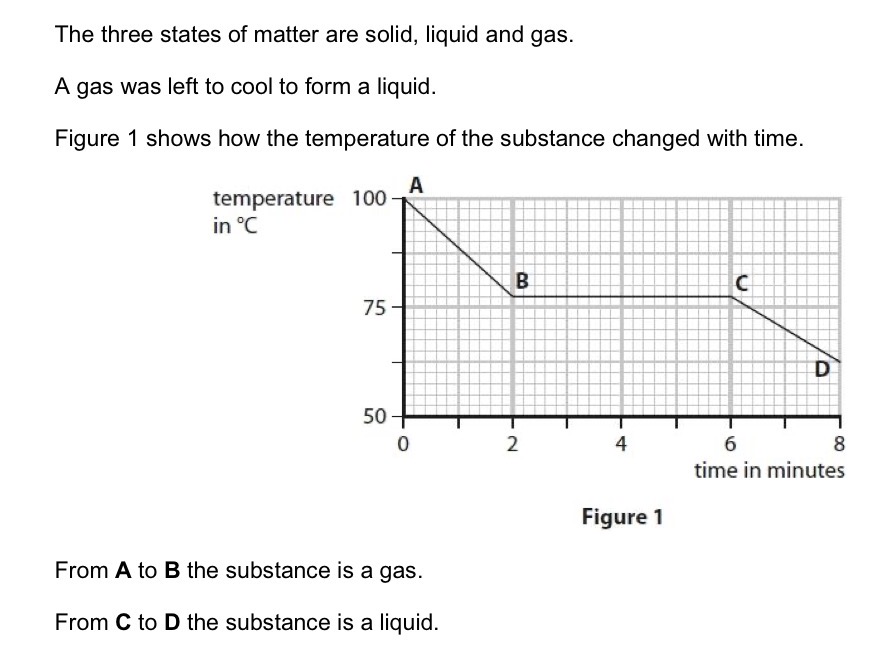
Calculate the number of minutes it took from the gas starting to form a liquid until the substance was completely liquid.
6 - 2 =4 minutes.

- 196°C.
It is lower than all the gas' boiling points.

Describe how to separate only nitrogen from the liquid air.
Heat the liquid to between -196 and below - 186°C.
A piece of dry ice is added inside an empty balloon. The neck of the balloon is sealed. Describe and explain what you would see happening.
The balloon would expand/get bigger.
This is due to the dry ice turning into a gas. Gas particles spread out and fill the balloon.
The student left the beaker of sodium chloride solution out uncovered in the classroom. The following week there was just a white solid at the bottom of the beaker.
Explain what happened.
The water evaporated and sodium chloride crystals are left behind.

B.
What is a cooling curve?
A graph that shows how a substance changes from a gas to a liquid to a solid as temperature decreases.
How does a horizontal line on a cooling curve indicate?
Where a substance is changing state.
What is a mixture?
Contains 2 or more elements/compounds that are not chemically bonded.
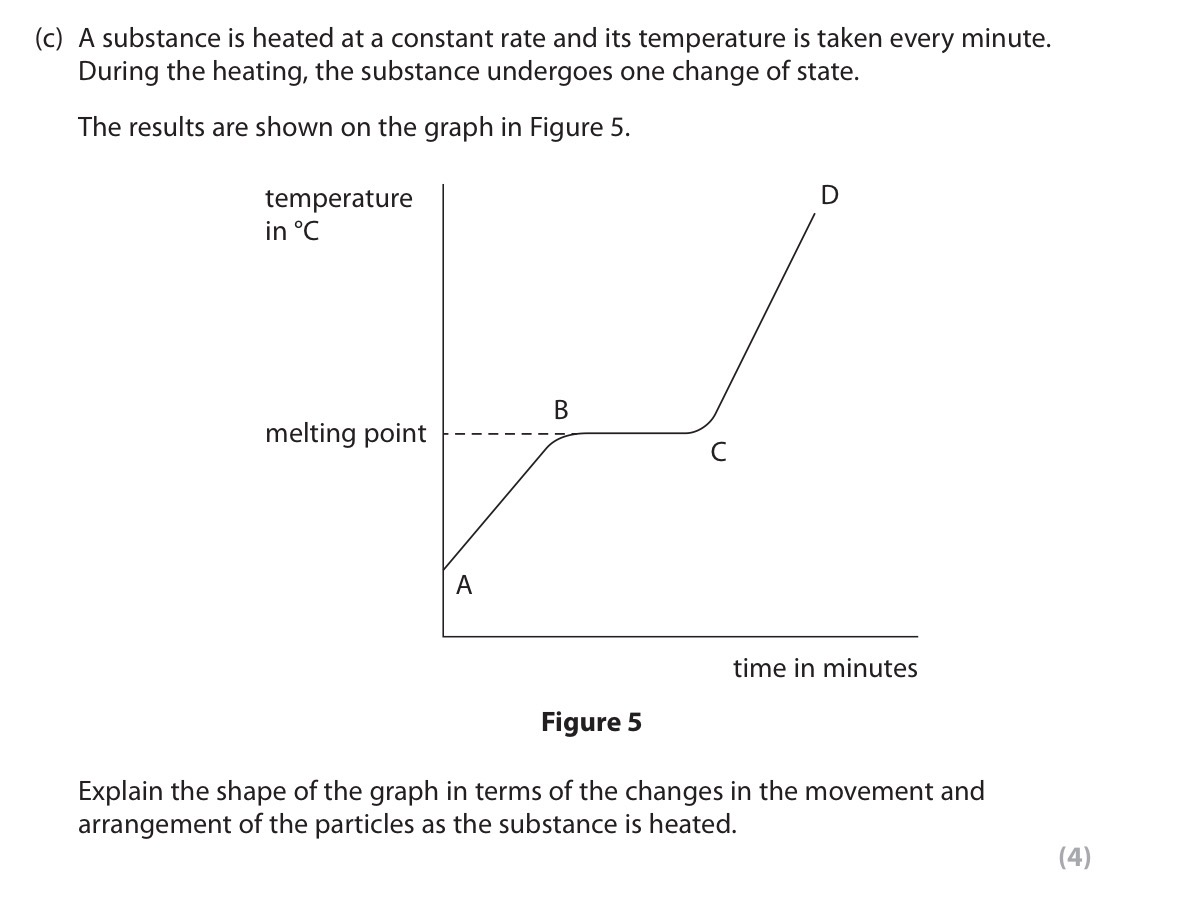
Explain the graph.
A to B: Particles in a solid are in fixed positions but begin to vibrate more rapidly as temperature increases.
B to C: Particles in a solid use energy to break out of lattice/break bonds between particles. Particles becoming randomly arranged as it turns from a solid to liquid.
C to D: Particles in liquid move past one another in a random arrangement. Particles move more rapidly as temperature increases in all directions.
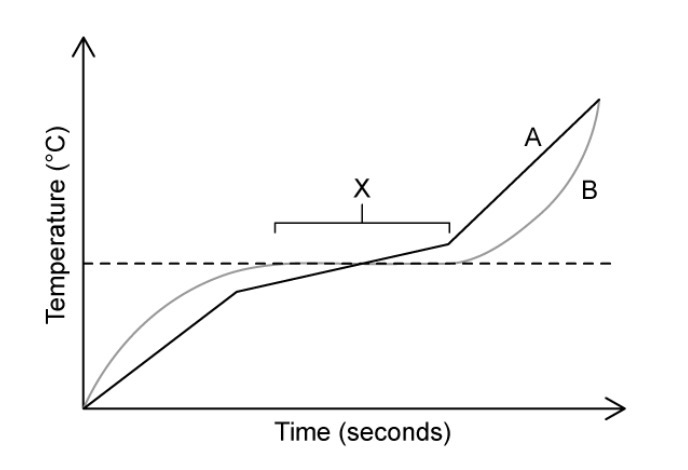
Identify which substance is pure and which one is a mixture.
A is a mixture as it melts over a range of temperatures whilst B is pure as it has a sharp/defined melting point.
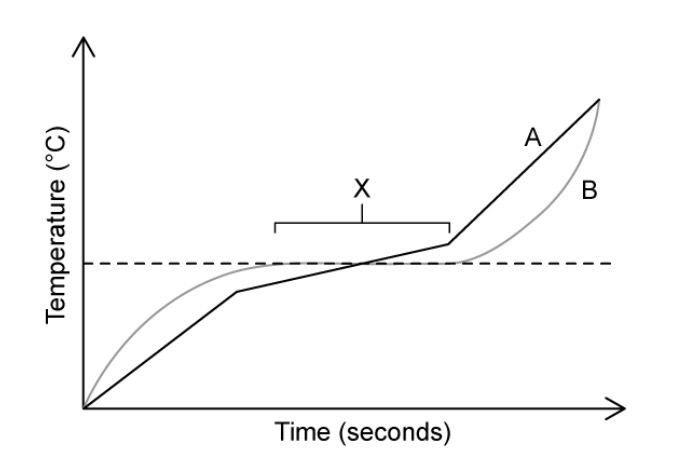
Suggest a reason for the horizontal part of substance B’s heating curve, labelled X.
The temperature doesn’t increase because heat energy is being used to overcome the forces between particles.
Pick A, B, C or D.
C.
What is a pure substance?
Contains a single element or compound and has a fixed position.
Describe the melting and boiling points of a pure substance.
Melt and boil at specific temperatures.
How can you use melting point data to distinguish between pure substances and mixtures?
Pure substances have a specific, exact melting point whereas mixtures melt over a range of temperatures.
What property can be used to distinguish a pure substance from a mixture?
Melting and boiling point.
State the use of melting point analysis.
To assess the purity of a substance.
Does a pure substance or a mixture melt and boil over a range over temperatures?
Mixture.
Does a pure substance or a mixture melt and boil at a specific, sharp temperature?
Pure substance.
What is the difference between a pure substance and mixture?
A pure substance consists of only substance (this can be an element or a compound), while a mixture contains two or more different substances that are not chemically combined.
Salt is dissolved in water. Is this a pure substance or a mixture?
Mixture.
How can melting and boiling points distinguish between pure substances and mixtures?
Pure substances have specific and sharp melting and boiling points, while mixtures have a range of melting and boiling points.
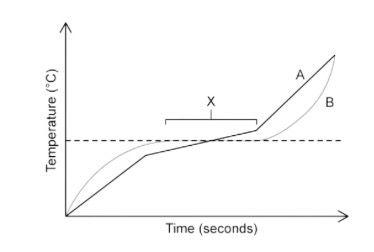
Is substance A pure or impure?
Impure.
What is the purpose of filtration?
A separation technique used to separate mixtures of insoluble/undissolved solids from a solution.
Which separation technique can be used to separate mixtures of insoluble/undissolved solids from a solution?
Filtration.
Which separation technique can be used to separate sand from a mixture of sand and water?
Filtration.
Which separation technique should be used to separate sodium chloride solution and the sand?
Filtration.
Cooper oxide is an insoluble solid. State the separating technique the student should use to remove excess copper oxide.
Filtration.
Explain the process of filtration.
Filter paper placed in a filter funnel above a beaker.
Mixture of insoluble solids and liquid poured into a funnel.
Filter paper only allows small liquids (solution) to pass through. These liquids become known as the filtrate and collect in the conical flask.
Insoluble solid particles are too large to pass through the filter paper so stay there. These solids become known as the residue.
Describe how to separate an insoluble substance from a solution.
Place filter paper in a funnel.
Pour solution containing an insoluble substance through funnel into a conical flask.
The insoluble substance will collect on the filter paper and the solution will collect in the conical flask.
What is the purpose of crystallisation?
A separation technique used to separate a soluble, dissolve solids from a solution.
Which separation technique can be used to separate a dissolved solid from a solution?
Crystallisation.
For crystallisation to happen, the solid must be what?
It must be more soluble in hot solvent than in cold so that it can decompose when heated.
Explain the process of crystallisation.
Solution is heated in an evaporating dish for a solvent to evaporate. Saturated solution left behind.
When crystals begin to form, remove dish from heat and leave to cool.
Wash crystals with pure water.
Filter crystals out of the solution and leave in a warm place to dry and pat dry with filter paper.
Explain how you could separate a soluble solid from a solution.
Crystallisation. Pour solution into an evaporating dish and heat gently.
When crystals begin to form, remove dish from heat and leave to cool.
Wash crystals with pure water.
Filter crystals out of the solution and leave in a warm place to dry (oven) and pat dry with filter paper.
Describe how to obtain a sample of pure, dry sodium chloride crystals can be obtained from the filtrate.
Heat solution.
Leave solution to cool.
Filter off crystals and pat dry between filter papers or leave to dry in an oven.
How would you separate potassium nitrate from potassium nitrate solution?
Crystallisation.
Describe how a sample of pure dry nickel sulfate crystals can obtained from the mixture of nickel solution and excess solid nickel carbonate in the beaker.
Filtration.
Place filter paper in a funnel.
Pour solution containing an insoluble substance through funnel into a conical flask.
The insoluble substance will collect on the filter paper and the solution will collect in the conical flask.
Crystallisation.
Heat solution to leave crystals behind.
Allow to cool. Dry crystals between filter papers and leave in a warm oven.
Solid sodium chloride dissolves in water but not ethanol. Suggest a method of separation that would separate a mixture of sodium chloride and ethanol but not a mixture of sodium chloride and water.
Filtration.
Liquid ethanol would Flow through the filter paper whilst solid sodium chloride would collect as the residue.
However, a mixture of dissolved sodium chloride would pass through the filter paper.
Solid sodium chloride dissolves in water but not ethanol. Suggest a method of separation that would separate a mixture of both sodium chloride and water and sodium chloride and ethanol.
Simple distillation. Water and ethanol would both evaporate and leave behind solid sodium chloride.
What is the purpose of simple distillation?
A separation technique used to separate a solvent from a solution.
Which separation technique can be used to separate a solvent from a solution?
Simple distillation.
Which separation technique can be used to separate a liquid from a solid?
Simple distillation.
How can seawater be purified?
Simple distillation.
Which separation technique could be used to separate methanol and ethanol?
Simple distillation.
Suggest how pure titanium chloride could be separated from dissolved impurities.
Simple distillation.
How can water be separated from sodium chloride solution?
Simple distillation.
What is the liquid collected in the beaker at the end of simple distillation known as?
Distillate.
A condenser is used during distillation. Where does cold water enter and exit the condenser?
Water enters at the bottom and leaves at the top.
During distillation, a bunsen burner is used to heat the flask. Give an alternative piece of apparatus that could be used to heat the flask.
Electric heater or blow torch.
Explain why a condenser is used during simple distillation.
To cool the water vapour back to liquid.
Explain how ethanol be separated from water.
Simple distillation.
Mixture is boiled and since ethanol has a lower boiling point than water, ethanol evaporates first.
Ethanol vapour cools in the condenser, condensing back into a liquid before being collected.
What is the purpose of fractional distillation?
A separation technique used to separate two or more liquids with different boiling points in a mixture.
What is the difference between fractional and simple distillation?
Simple distillation is used to separate ba solvent from a solution whereas fractional distillation is used to separate two or more liquids with different boiling points in a mixture.
Which piece of equipment does fractional have that simple distillation does not?
Fractioning column.
How does fractional distillation separate oil?
Oil is heated until it evaporates into the fractioning column. The vapour rises up the column and condenses at differnet fractions, depending on the relative boiling point of each substance.
A student is using distillation to separate a flammable mixture. Suggest a piece of equipment he could use to heat the mixture.
Electric heater or water bath.