Chemistry: Measuring rates of reactions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is the method of mass loss to measure rate of reaction and when do we use it?
Change in mass/time taken
Used when gas is produced (grams per second g/s)
Measures decrease in mass due to loss of gas
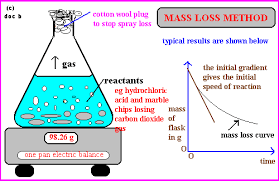
Why do we use a cotton wool plug in mass loss method?
To prevent loss of liquid
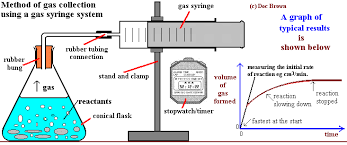
What is the gas syringe system?
Gas pushing syringe
measures volume of gas released
Catches gas to measure volume
Centimeters cubes per second (cm³/s)
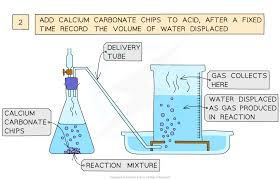
What is the displacement method?
Fill cylinder with water, gas pushes water
Measure volume of water displacement
Measures volume of gas released
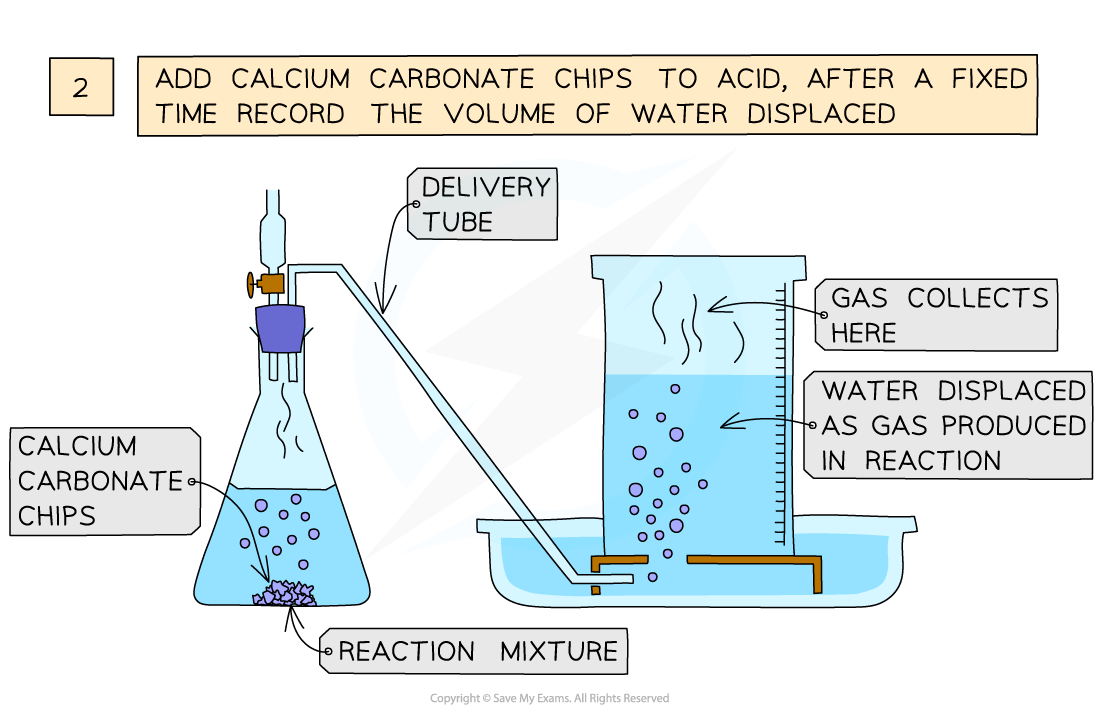
Which method would you use for the effect of surface area of a solid on the rate of reaction?
Displacement:
Add dilute hydrochloric acid to the conical flask
Use a delivery tube to connect this flask to an inverted measuring cylinder upside down in a water trough
Add calcium carbonate chips into the conical flask and close the bung
Measure the volume of gas produced in a fixed time using the measuring cylinder
Repeat with different sizes of calcium carbonate chips
Which method would you use for the effect of concentration of a solution on the rate of reaction?
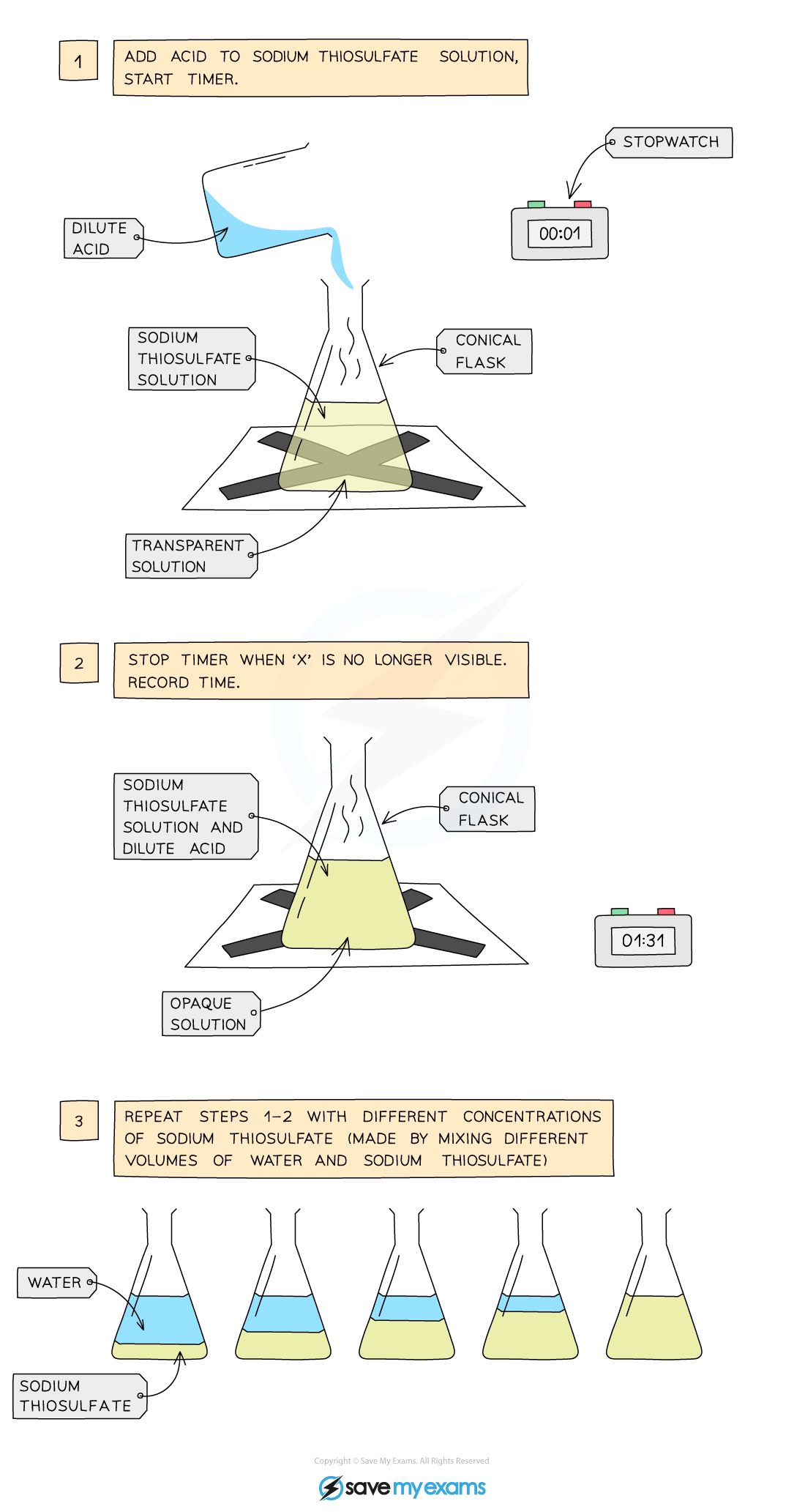
Disappearing cross (precipitation) method:
Measure 50 cm3 of sodium thiosulfate solution into a flask
Measure 5 cm3 of dilute hydrochloric acid into a measuring cylinder
Draw a cross on a piece of paper and put it underneath the flask
Add the acid into the flask and immediately start the stopwatch
Look down at the cross from above and stop the stopwatch when the cross can no longer be seen
Repeat using different concentrations of sodium thiosulfate solution (mix different volumes of sodium thiosulfate solution with water to dilute it)
Result:
With an increase in the concentration of a solution, the rate of reaction will increase
This is because there will be more reactant particles in a given volume, allowing more frequent and successful collisions, increasing the rate of reaction
Which method would you use for the temperature on the rate of reaction?
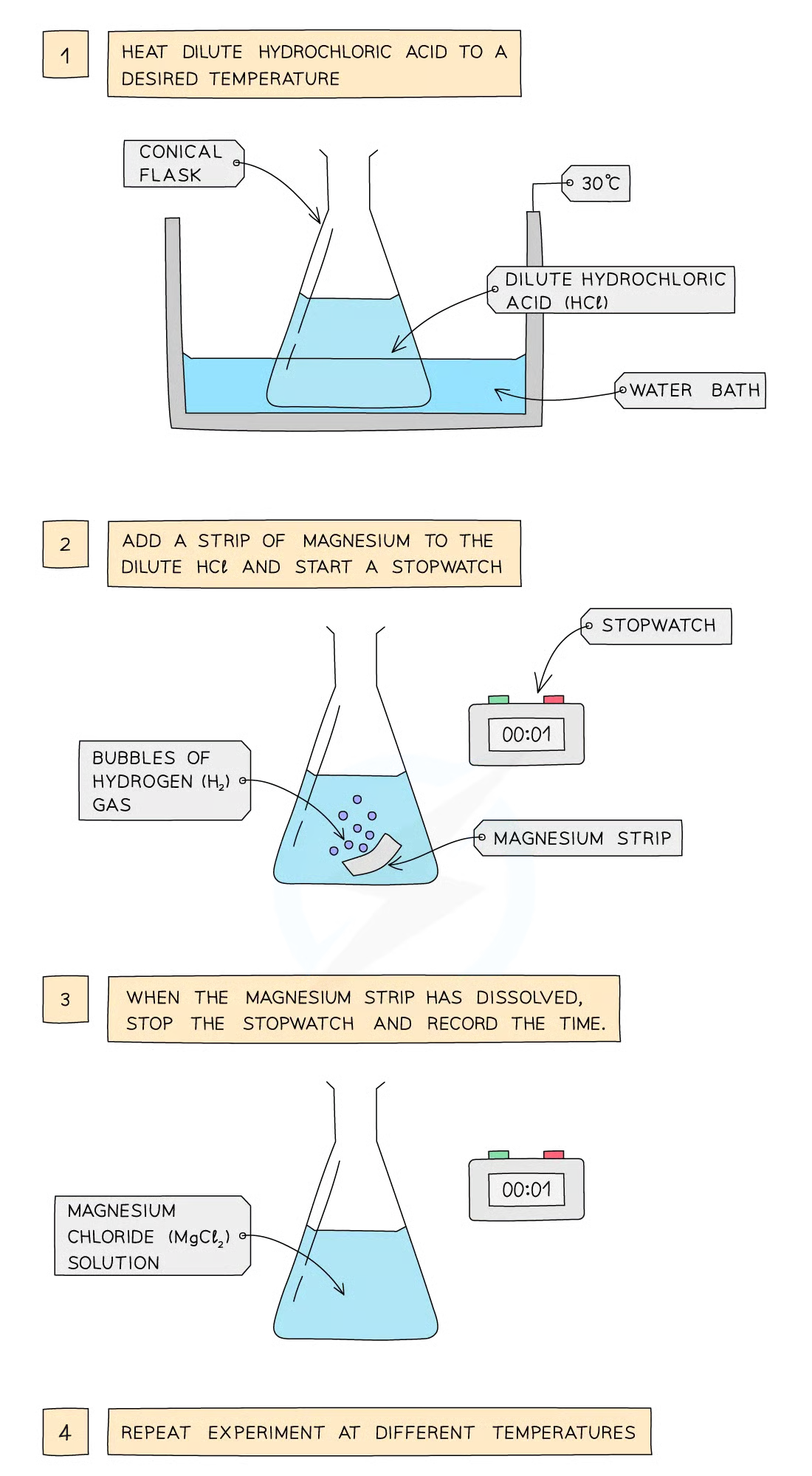
Method:
Dilute hydrochloric acid is heated to a set temperature using a water bath
Add the dilute hydrochloric acid into a conical flask
Add a strip of magnesium and start the stopwatch
Stop the time when the magnesium fully dissolves
Repeat at different temperatures and compare results
Which method would you use for the effect of catalyst on the rate of reaction?
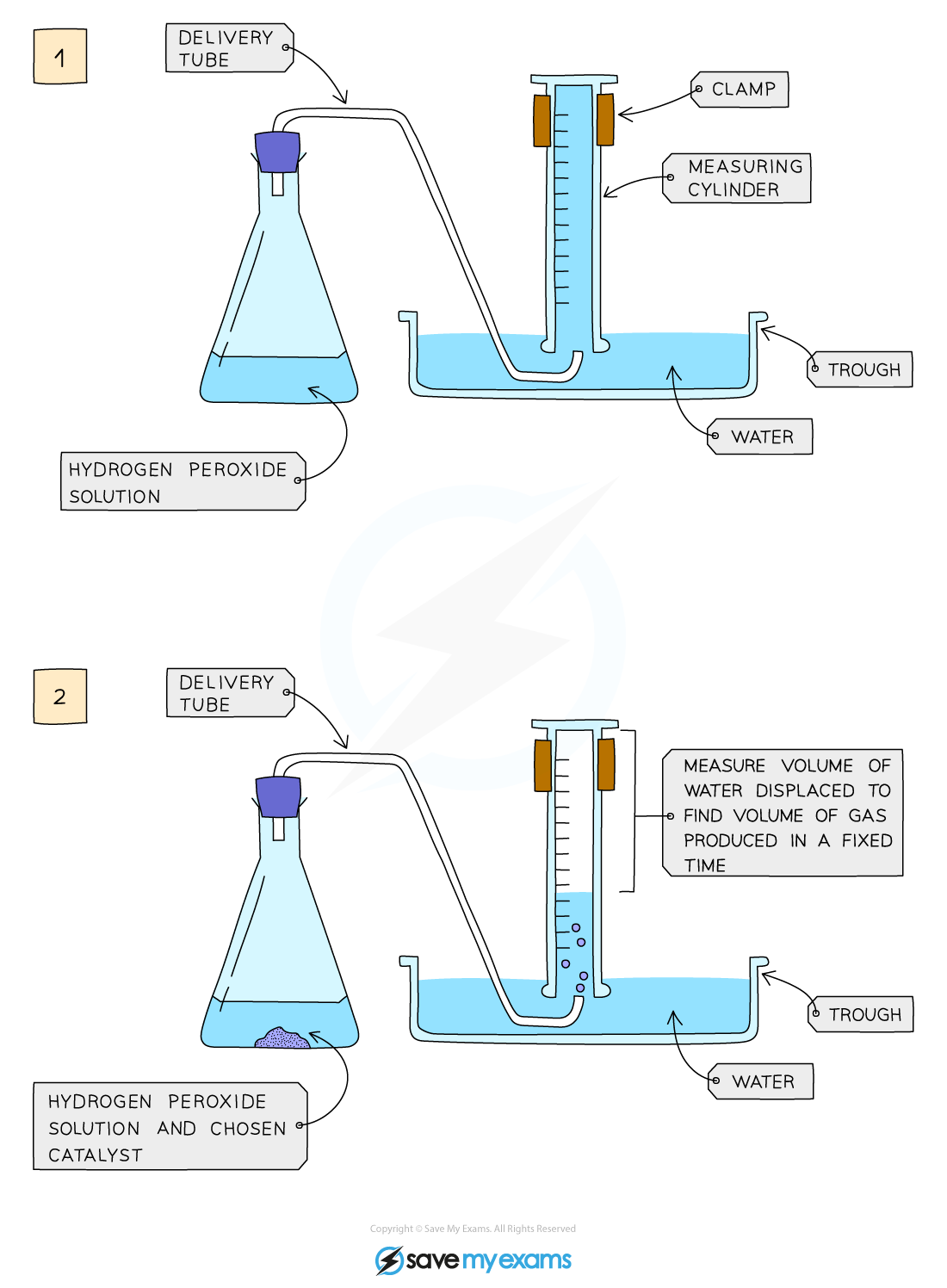
Method:
Add hydrogen peroxide into a conical flask
Use a delivery tube to connect this flask to a measuring cylinder upside down in water trough
Add the catalyst manganese(IV) oxide into the conical flask and close the bung
Measure the volume of gas produced in a fixed time using the measuring cylinder
Repeat experiment without the catalyst of manganese(IV) oxide and compare results
What factors affect the rate of reactions?
The concentration of the reactants in solution or the pressure of reacting gases
The temperature of the reaction
Surface area of solid reactants
The presence of a catalyst
What would the line on a graph look like when the rate of reaction increases in comparison?
Steeper, Becomes horizontal sooner, Forms the same amount of product
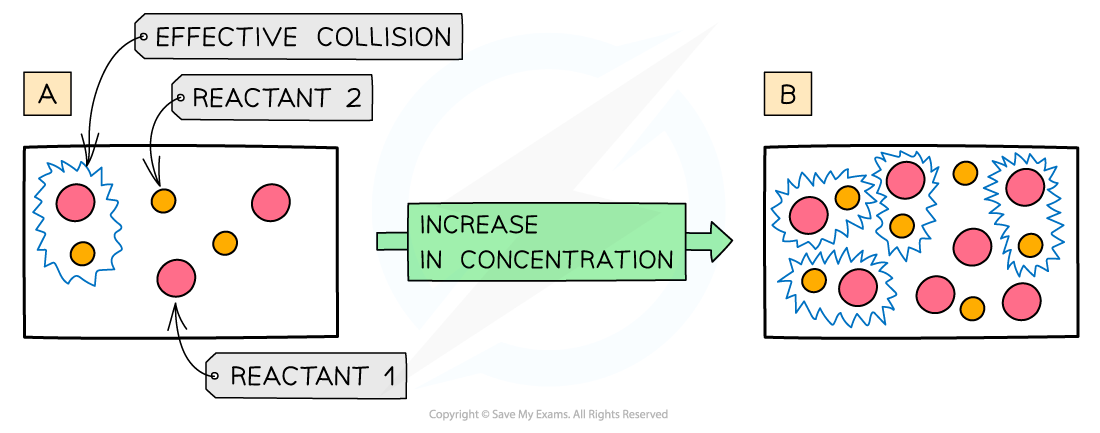
Why does increasing the concentration increase rate of reaction?
Increasing the concentration means that there are more reactant particles in a given volume
This causes more collisions per second
Leading to more frequent and successful collisions per second
Therefore, the rate of reaction increases
number of collisions is proportional to the number of particles present
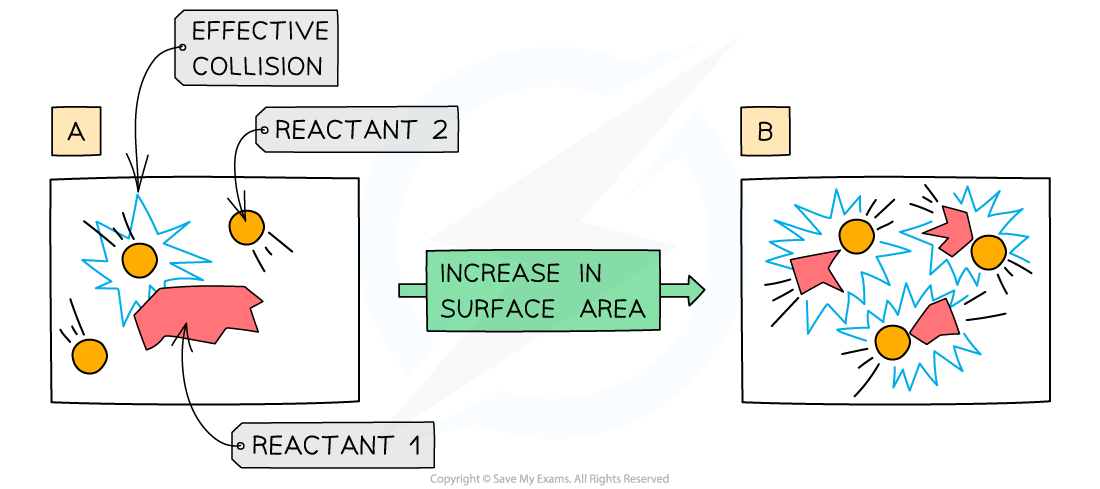
Why does increasing the surface area increase rate of reaction?
Increasing the surface area means that a greater surface area of particles will be exposed to the other reactant
This causes more collisions per second
Leading to more frequent and successful collisions per second
Therefore, the rate of reaction increases
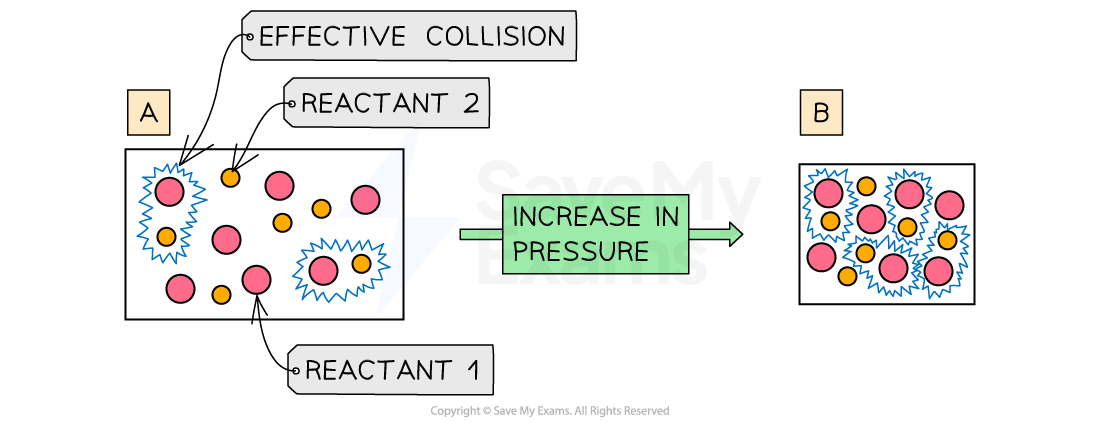
Why does increasing the pressure increase rate of reaction?
Increasing the pressure means that there are the same number of reactant particles in a smaller volume
This causes more collisions per second
Leading to more frequent and successful collisions per second
Therefore, the rate of reaction increases
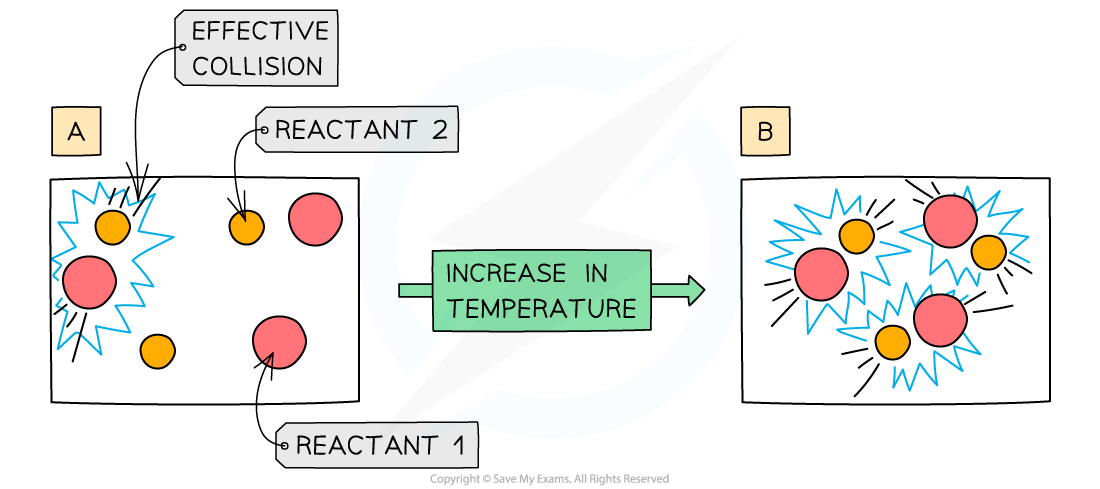
Why does increasing the temperature increase rate of reaction?
Increasing the temperature means that the particles have more kinetic energy
This causes more collisions per second
Leading to more frequent and successful collisions per second
Therefore, the rate of reaction increases
What are catalysts?
substances that speed up rate of reaction but remain unchanged by the end of the reaction.
Why do catalysts speed up rate of reaction?
They provide an alternative pathway for the reaction to occur with a lower activation energy, meaning particle collisions need less energy in order for reaction to occur.