Chem 201 - Chapter 1: Atomic Structures
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
38 Terms
which group contains nonmetals, metalloids, and metals?
15
Which compound contains a Group 14 metal?
A) CCl4
B) SnCl2
C) GeF4
D) ZnCl2
C) GeF4
which metal is an alkali metal?
A) K
B) Mg
C) V
D) Fe
A) K
isotopes
looking for the same number of protons and different numbers of neutrons
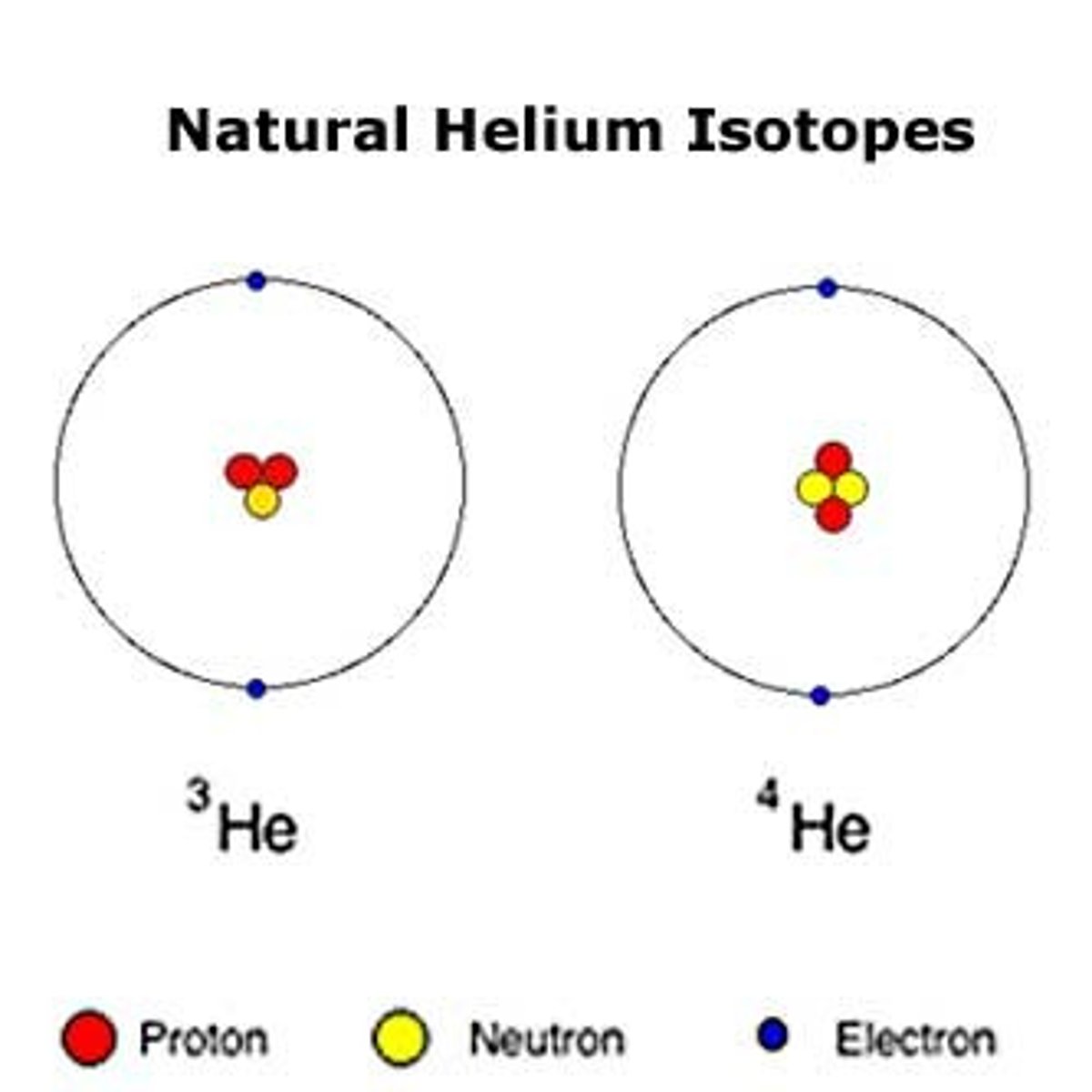
what is the atomic number for an ion that has a charge of +1 and contains 18 electrons?
19
(explanation: the ion has a +1 charge you know that there is one less electron than the proton in the ion. so, 18 + 1 = 19)
how many protons (p+), neutrons (n) and electrons (e-) are present in 47/22 Ti 3+?
22p+, 25n, and 19e-
(explanation: protons = bottom number in the fraction always = 22. neutrons = atomic mass (numerator) - # of protons (denominator) = 25. electrons = # of protons - (3+) = 19)
Chlorine occurs naturally as a mixture of two isotopes with atomic masses of 34.97 amu and 36.97 amu. What are the relative abundances of these isotopes?
76% and 24%
(explanation: [(mass isotope 1) %isotopes1/100%] + [(mass isotopes 2) %isotopes2/100% = average atomic mass.
%^37Cl = (average atomic mass Cl - mass 35Cl / mass 37Cl - mass 35Cl) x 100%
= (35.45 amu - 34.97 amu/ 36.97 amu - 34.97 amu) x 100% = 24%.
since Cl only has 2 natural isotopes 100 - 24= 76% = %^35 Cl)
What is the classification of sulfur?
nonmetal
If the formula of an ionic oxide of element X is X2O3, what is the formula of the chloride of X?
XCl3
(explanation: in X2O3, 1 X has a of + 3 because since oxygen always has a -2 charge in any compound the charge of O3 is -6. since a molecule has to have a balance charge of 0 that means X2 has a charge of +6. since Cl regularly has a -1 charge there needs to be 3Cl to balance 1X)
Which pair represents isotopes?
A) 54/24 Cr and 54/26 Fe
B) 235/92 U and 238/ 92 U
C) 116/48 Cd and 116/50 Sn
D) 239/93 Np and 239/94 Pu
B) 235/92 U and 238/ 92 U
(explanation: same number of protons (92) different masses indicate different neutrons. plus, it's the same element unlike the other options)
how many neutrons are in 14/6 C?
8
in all neutral atoms, there are an equal number of
electrons and protons
which element is represented by 56/24 X?
Chromium
which atoms has the most neutrons?
A) neon - 20
B) chlorine - 35
C) phosphorus - 32
D) sulfur - 35
D) sulfur - 35
Bromine has two naturally occurring isotopes. The most abundant isotope (50.69%) is 79/35 Br of mass 78. 9183 amu. What is the other isotope?
A) 79/36 Br
B) 81/ 36 Br
C) 80/35 Br
D) 81/35 Br
D) 81/35 Br
(explanation: both need the same number of protons (35) and a different number of neutrons than the other isotope (81-35 = 46 while the other is 79-35 = 44)
Two isotopes of hypothetical element X exist with BUNDANCES OF 30.00% 100X and 70.00% 101X. What is the approximate atomic mass of X (in atomic units, amu)?
100.7 amu
Lithium has two isotopes, lithium-6 (atomic mass = 6.015) and lithium-7 (atomic mass = 7.016). what is the relative abundance of each isotope?
Li-6 = 7.49%; Li-7 = 92.51%
which is a main group metal?
A) Pu
B) Mg
C) Sc
D) Fe
B) Mg
Which statement is true?
A) chlorine is a transition metal
B) oxygen is a metal
C) sodium is a metalloid
D) phosphorous is a nonmetal
D) phosphorous is a nonmetal
Which element is actinide?
A) Rb
B) U
C) Mo
D) Ga
B) U
Which compound contains a +1ion?
A) MgO
B) KCl
C) NO
D) AlP
B) KCl
If gallium, atomic number 31, combines with selenium, atomic number 34, what is the most likely formula to be formed?
Ga2Se3
An atom of strontium-90, 90/38 Sr contains
38 electrons, 38 protons, 52 neutrons
(explanation: mass of the atom is 90. The atomic number is 38 so there are 38 protons and electrons. atomic mass - # of protons/electrons = neutrons so 90-38 = 52.
Which group number contains the most diatomic molecular elements?
Group 17
(explanation: the seven diatomic molecular elements are : H2, N2, O2, F2, Cl2, Br2, and I2. four of these are in Group 17 on the periodic table.)
Which term best describes the relation of hydrogen (1H) to deuterium (2H)?
isotopes
which ion has twenty-six electrons?
A) Cr2+
B) Fe2+
C) Ni2+
D) Cu2+
C) Ni2+
which ion has the same number of electrons as an argon atom?
A) Mg2+
B) Cl-
C) Ne
D) Na+
B) Cl-
The element thallium, Tl, has two stable isotopes, 203Tl and 205Tl. Which statement correctly describes the relationship between the relative abundances of these two isotopes?
A) % abundance of 203Tl > % abundance of 205Tl
B) % abundance of 203Tl = % abundance of 205Tl
C) % abundance of 203Tl < % abundance of 205Tl
D) not enough info
C) % abundance of 203Tl < % abundance of 205Tl
(Explanation: The mass of thallium on the periodic table is 204.4 amu. mass of 203Tl is 203amu and the mass of 205Tl is 205amu. 204.4 is closer to 205 than it is to 203.)
which statement is true?
A) the nucleus of an atom contains neutrons and electrons
B) the atomic number of an element is the number of protons in one atom
C) the mass of an atom is the number of protons in the nucleus plus the number of electrons outside
D) the mass of electrons outside the nucleus is the same as the number of neutrons is the nucleus
B) the atomic number of an element is the number of protons in one atom
how many neutrons are in 37/17Cl-?
20
which pair of particles has the same number of electrons?
A) F-, Mg2+
B) Ne, Ar
C) Br-, Se
D) Al3+, P3-
A) F-, Mg2+
A sodium ion differs from a sodium atom in that the sodium ion
has fewer electrons
An enriched sample of carbon contains 20.0% 12C and 80.0% 13C. Which ratio shows that? (white dots = 13C, black dots = 12C)
A) 2 white dots; 8 black dots
B) 5 white dots; 5 black dots
C) 8 white dots; 2 black dots
D) 10 white dots
C) 8 white dots; 2black dots
The halogens are found in group
17
Which compound below contains a transition metal?
A) NaF
B) AlCl3
C) VO2
D) BF3
C) VO2
Magnesium forms an ionic compound with an element with the formula MgX. Which ion could be X?
A) Br-
B) P3-
C) C4-
D) S2-
D) S2-
Alkaline earth metals form
+2 ions
what is the formula for the compound formed when an aluminum ion, Al3+, combines with an oxide ion, O2-
Al2O3