CHEM 2370, Chapter 9, Exam 4
1/33
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
epoxide
oxygen in a three membered ring, unstable + ring strain
phenol
benzene ring with R group attached
benzyl
benzene ring with R group on a methyl group
ether
oxygen with 2 R groups
Boiling point increases with
lower degree alcohols bc steric hindrance allows easier hydrogen bonding
Williamson Ether Synthesis: conditions
SN/SB alkoxide and a primary alkyl halide
Williamson Ether Synthesis: mechanism
oxygen on alkoxide takes R group from alkyl halide as the halogen leaves
Synthesis of unhindered alkoxide for Williamson Ether Synthesis
2° alcohol + NaH → isopropyl attached to ONa + H2 gas

halohydrin
OH and halide on adjacent carbons
Williamson Ether Synthesis: halohydrin form mechanism
acid base reaction where NaH takes H from hydroxyl group on Halohydrin → SN2 reaction as O- forms an epoxide as halogen leaves
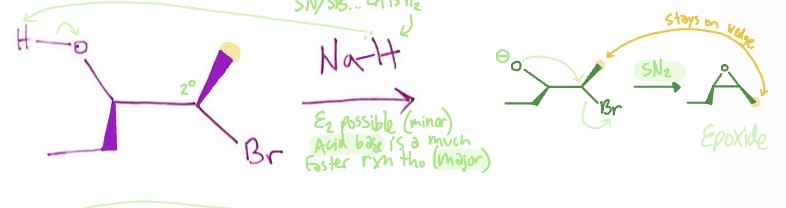
Williamson Ether Synthesis: halohydrin form products
epoxide
Williamson Ether Synthesis: halohydrin form reaction type
acid base → SN2 (inversion)
Williamson Ether Synthesis: halohydrin form conditions
alkane with hydroxyl group and halogen on adjacent carbons (anti) + NaH
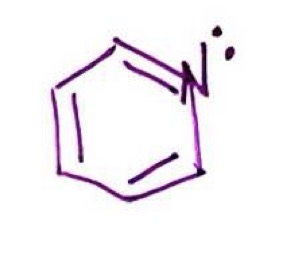
pyridine
benzene ring with Nitrogen as connecting point
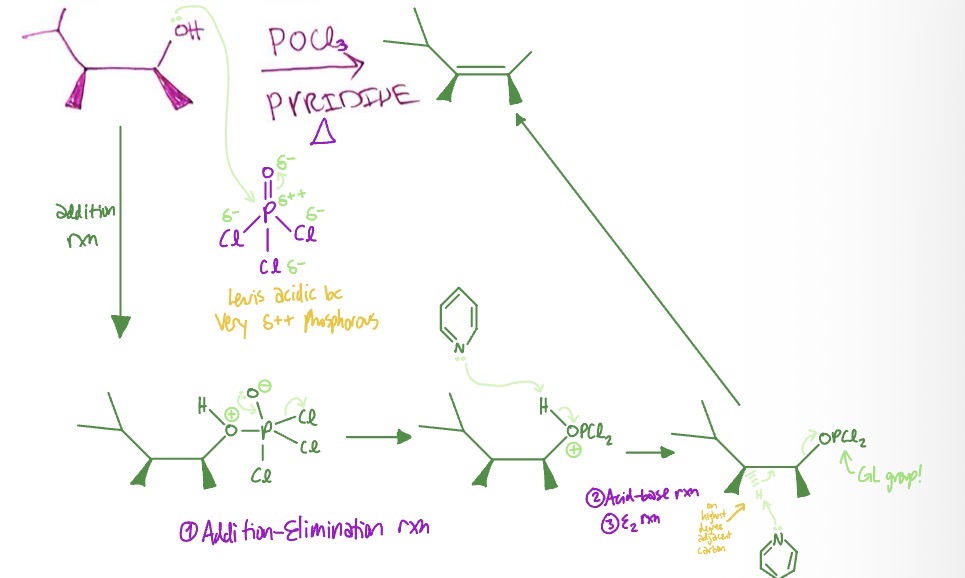
Acid-Catalyzed Dehydration w/o rearrangement: type of reaction
addition-elimination → acid-base → E2
Acid-Catalyzed Dehydration w/o rearrangement: mechanism
hydroxyl group takes POCl3 as pi bond on partially negative O breaks → lone pair on O- on P turns into pi bond again as Cl leaves, O from hydroxyl is positive → LP on pyridine takes H from O+ → LP on pyridine takes H from highest degree adjacent carbon as OPCl2 leaves → alkene forms from absent H
Alcohols and HX: mechanism
ROH + HX → ROH2+ → RX
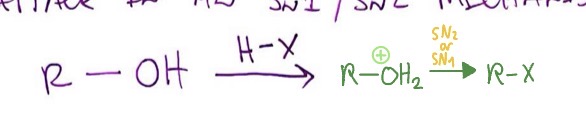
Alcohols and HX: conditions
alcohol and HBr, HI, or HCl (AND ZnCl2)
Alcohols and HX: type of reaction
SN2 if methyl or 1°, SN1 if 2° or 3° w/ rearrangement
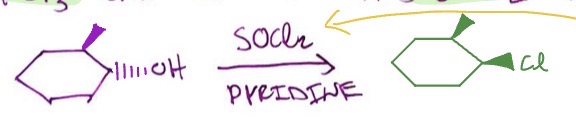
Other OH → alkyl halide conversions: SOCl2 mechanism
1°/2° OH + SOCl2 + pyridine → SN2 rxn, alkyl chloride
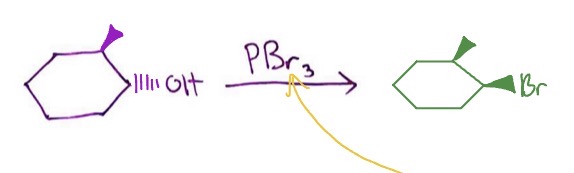
Other OH → alkyl halide conversions: PBr3 mechanism
1°/2° OH + PBr3 → alkyl bromide
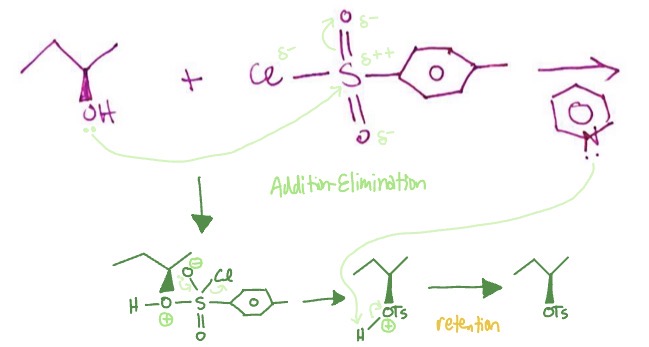
Alcohols into Tosylate Group: conditions
alcohol + TSCl (p-Toluene Sulfonyl Chloride)/MsCl (mesyl chloride)/TfCl (triflic anhydride) + pyridine
Alcohols into Tosylate Group: mechanism
O from hydroxyl attaches to S as a pi bond in attached O breaks and makes it O- → O- reforms pi bond with LP as Cl leaves, O from OH group is positive → pyridine takes H from positive hydroxyl → left with retained OTs group on alkane
Alcohols into Tosylate Group: sulfonyl groups possible
TsCl, MsCl, TfCl → large molecule with central sulfide
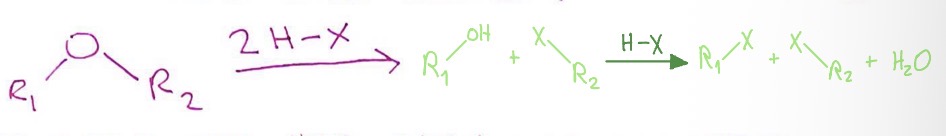
Ether to Alkyl Halide: conditions
ether + strong acid + it’s conjugate base (ether + 2HX)
Ether to Alkyl Halide: mechanism
ether + 2HX → ROH + RX + HX → RX + RX + H2O
Ether to Alkyl Halide: 3° mechanism type
SN1 mechanism
Ether to Alkyl Halide: 1° mechanism type
SN2 mechanism
Ether to Alkyl Halide: outlier form
ether attached to a benzene ring + 2HX → OH on benzene ring + X on alkane from opposite side of ether + HX
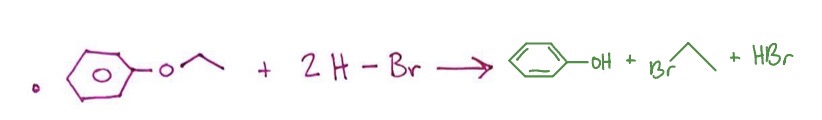
BLAM
Base will attack Least substituted carbon under basic conditions, Acid will attacked Most substituted carbon under acidic conditions
Epoxide in Acid: mechanism
epoxide takes H from acid and becomes positive → CB of acid attacks most substituted carbon as ring breaks → H from positive H2O is taken by CB
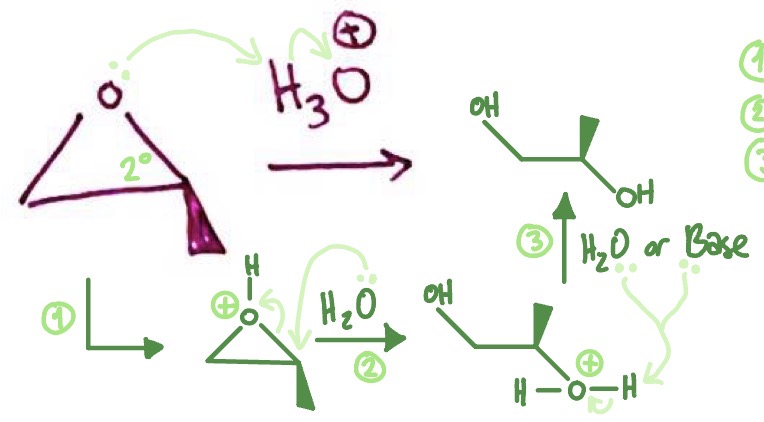
Epoxide in Acid: reaction type
acid base → SN2 rxn→ sometimes acid base again
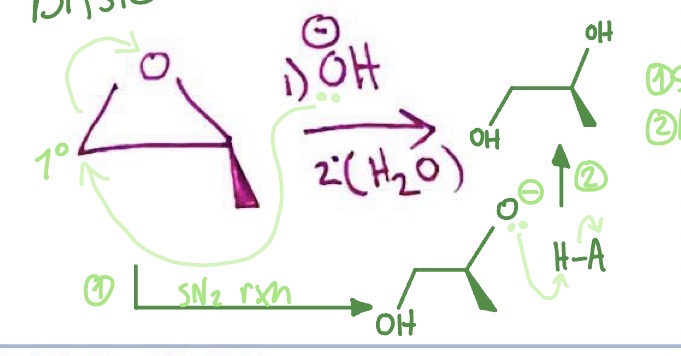
Epoxide in Base: mechanism
base attacks least substituted carbon of epoxide as ring breaks and epoxide O becomes negative → O- takes H from CA of base
Synthesis of Epoxides from Alkenes and Peroxy Acids: mechanism
alkene + peroxy acid (mCPBA) → epoxide on wedges + EN