Specific heat capacity: Energy: Physics: GCSE (9:1)
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
14 Terms
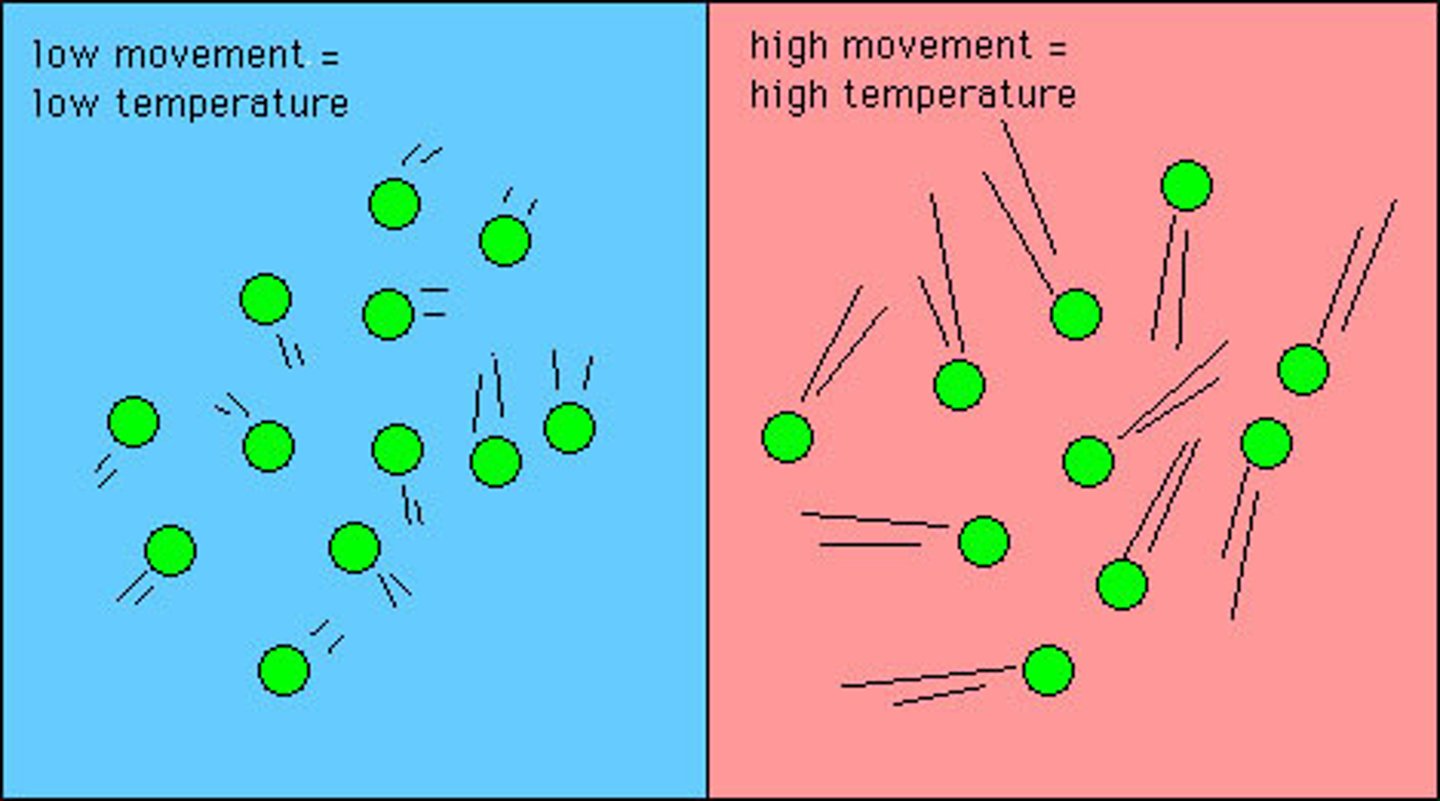
Temperature
A measure of the average kinetic energy of the particles in a substance
Specific heat capacity (SHC)
The energy required to raise the temperature of one kilogram (1 kg) of a substance by one degree Celsius (1 ºC)
ΔE = mcΔθ
The equation linking the change in thermal energy, mass, specific heat capacity and temperature change
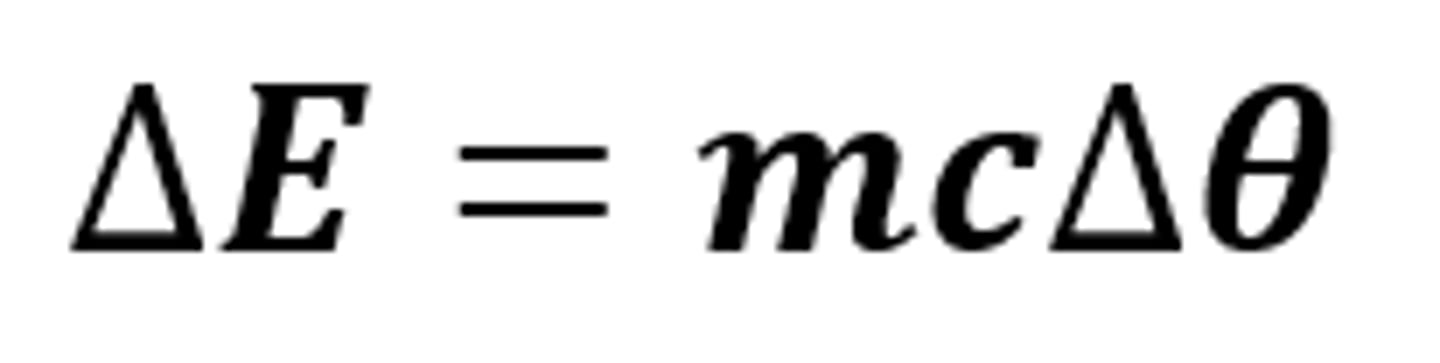
ΔE
The symbol for the change in thermal energy
m
The symbol for mass
c
The symbol for specific heat capacity
Δθ
The symbol for change in temperature
Joules (J)
The SI unit for energy and work

Joules per kilogram-degrees Celsius (J/kgºC)
The SI unit for specific heat capacity
Kilograms (kg)
The SI unit for mass

Degrees Celsius (ºC)
The SI unit for temperature

4200 J/kgºC
The specific heat capacity of water

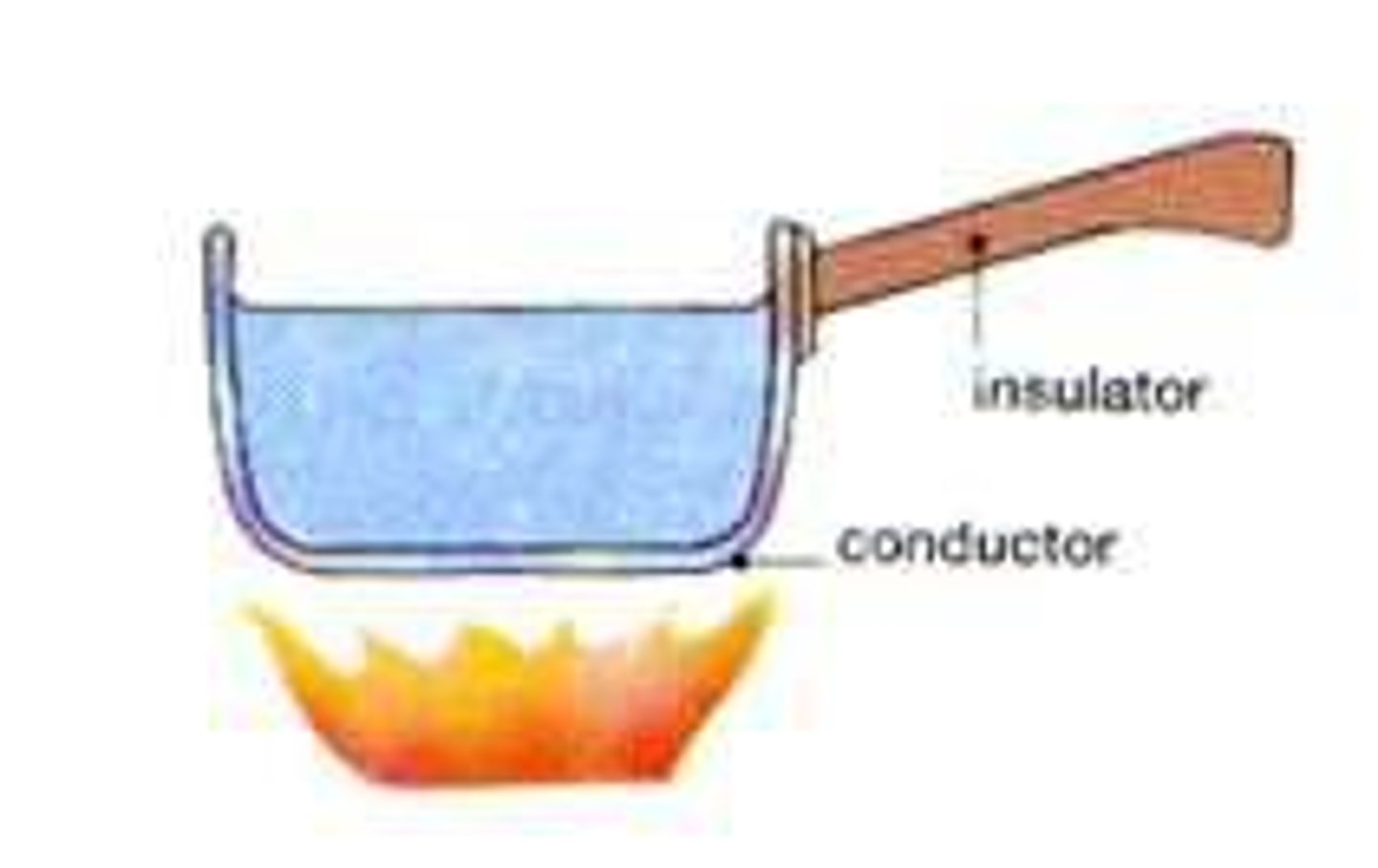
Thermal conductors
Materials with a low specific heat capacity so heat up and cool down quickly
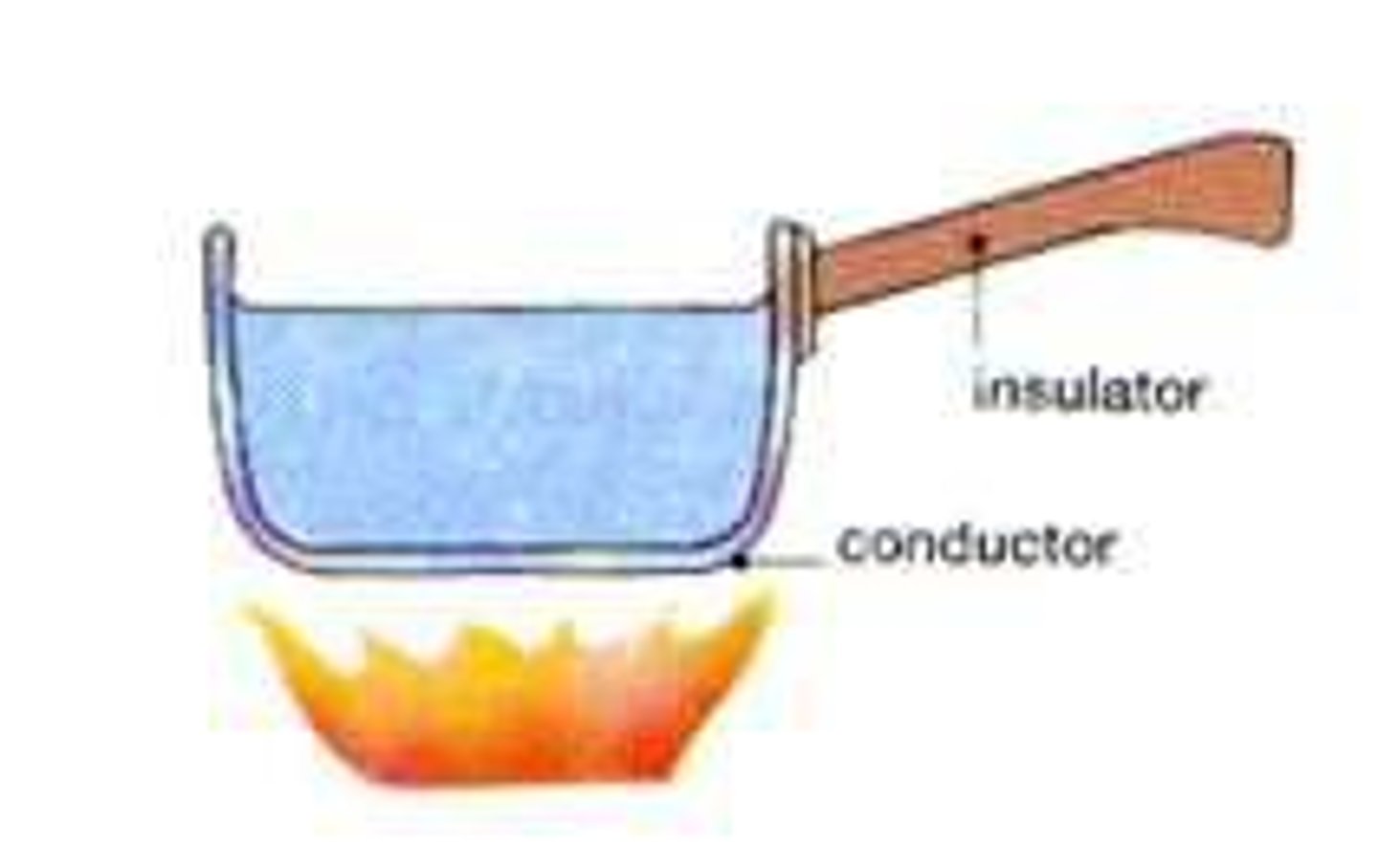
Thermal insulators
Materials with a high specific heat capacity so heat up and cool down slowly