Dienes, Cnojugated Dienes, Pericyclic Reactions.
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
34 Terms
What is an Isolated Diene
RCH2=CHCH2CH2CH=CH2R. Always has 4 carbons in between the double bonds.
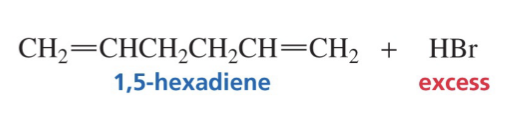
Give the product of this Isolated Diene RXN and Show the reaction mechanism.
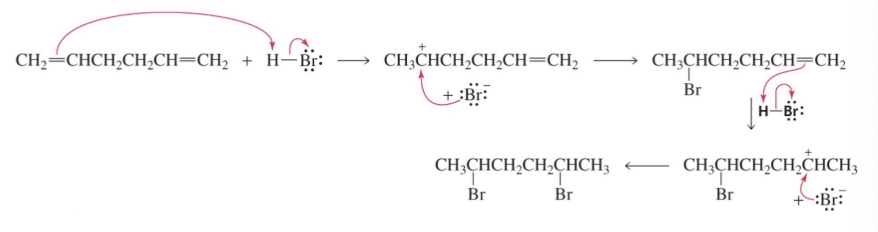
What does hydrohalogenation with a diene prefer to do?
Create a more subsitueted carbocation then attack it with a halogen.
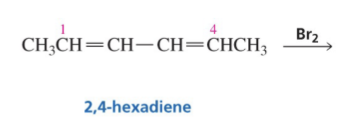
Show the the 1,2 products and 1,4 products of this reaction.
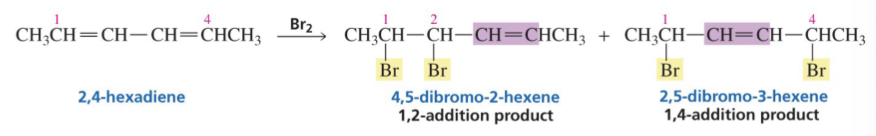
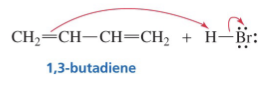
Show reaction mechanism of conjugated Diene
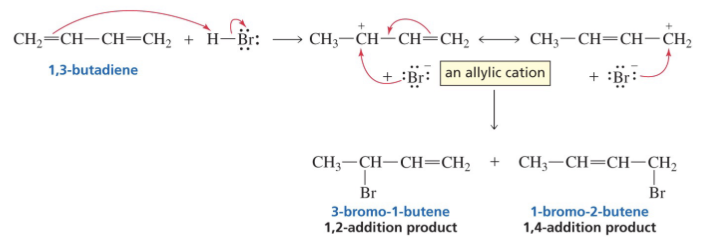
If reaction is irreversible, what product dominates?
Kinetic Product
If reaction is reversible, what product dominates?
Thermodynamic product
What is always a 1,2 product?
A kinetic product.
What is the proximity effect?
When the H and Cl- stay close to each other makeing the 1,2 product form faster.
Kinetic Reaction characteristics
Both reactions are irreversible, major product is whats formed more rapidly.
Thermodynamic Reaction Characteristics
One or both reactions are reversible. Major product is one that is more stable.
What is needed for a Diels - Alder Reaction? What does it form?
Forms a six membered ring
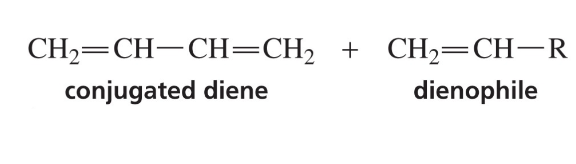
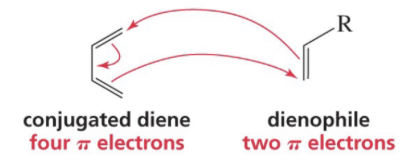
Show reaction mechanism of Diels Alder RXN?
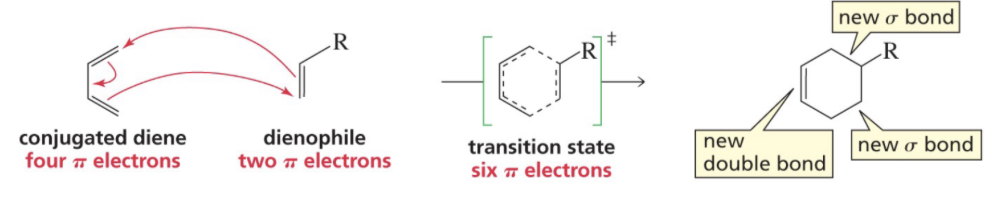
What is happening to pi and sigma bonds in Diels Alder rxn?
2 pi bonds form 2 sigma bonds
What does the Electron Withdrawing Group do?
Makes Dienophiles better Dienophiles
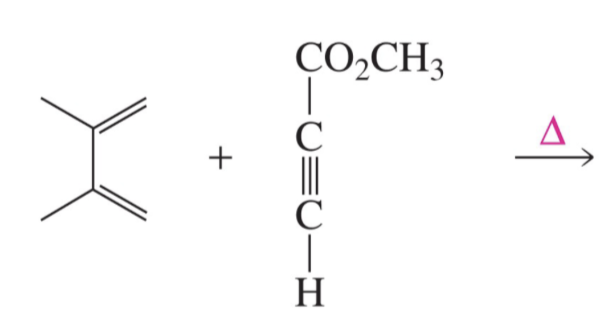
Can alkynes be Dienophiles? If so, draw product of this reaction.
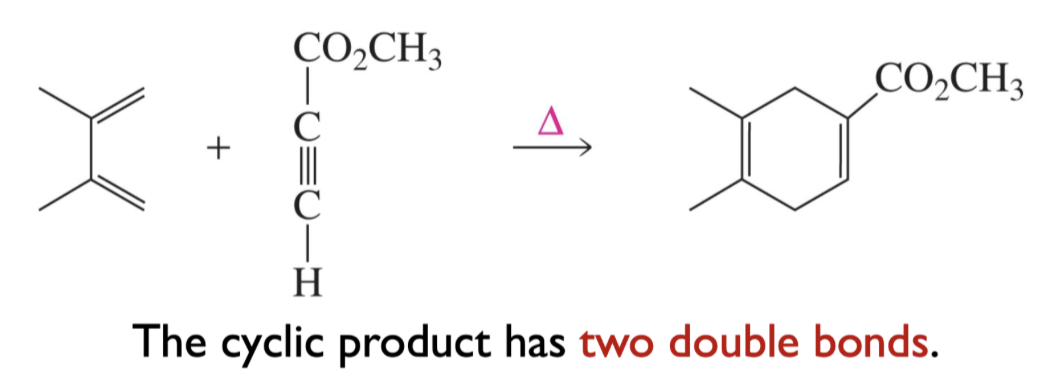
How many products are available if both reactants are not symmetric?
Two products are possible.
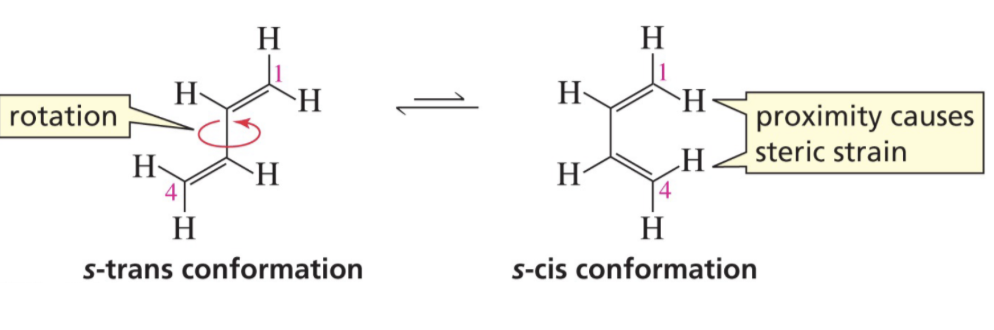
What conformation does Dienes need to react?
s-Cis Conformation
What kind of center does Diels Alder create? An Asymmetric Center?
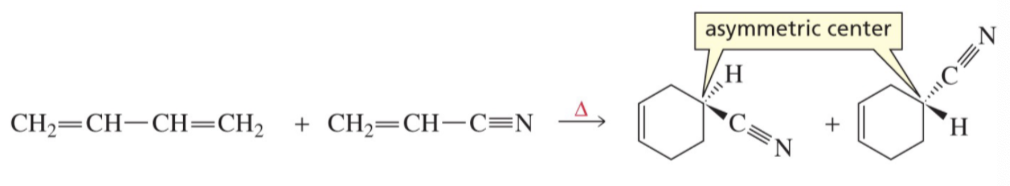
Cis forms Cis and Trans forms Trans
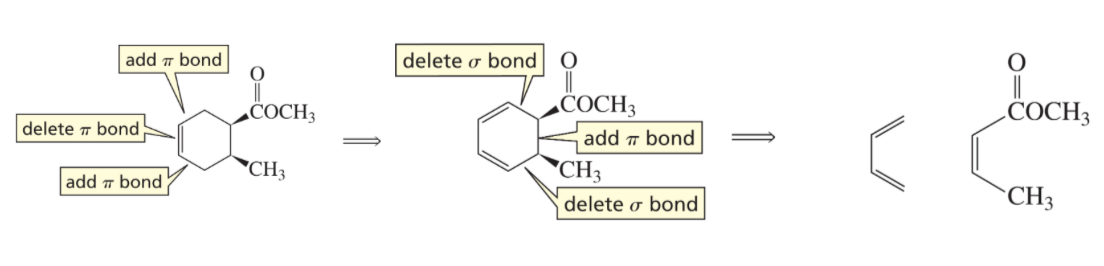
Use this to determine Reactants of Diels- Alder Reaction
What is a Electrocyclic Reaction?
Intramolecular reaction in which a sigma bond is formed between ends of conjugated pi bonds
Reaction mechanism of Electrocyclic reaction
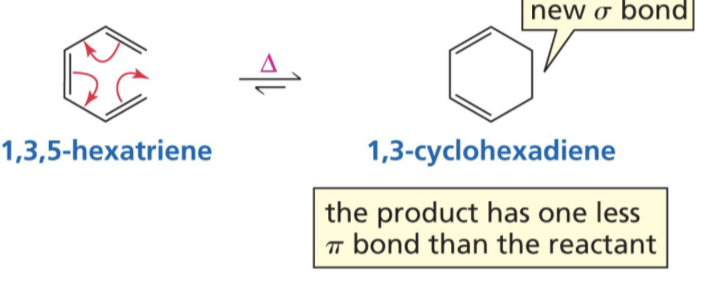
Show reaction mechanism for reversible Electrocyclic reactions
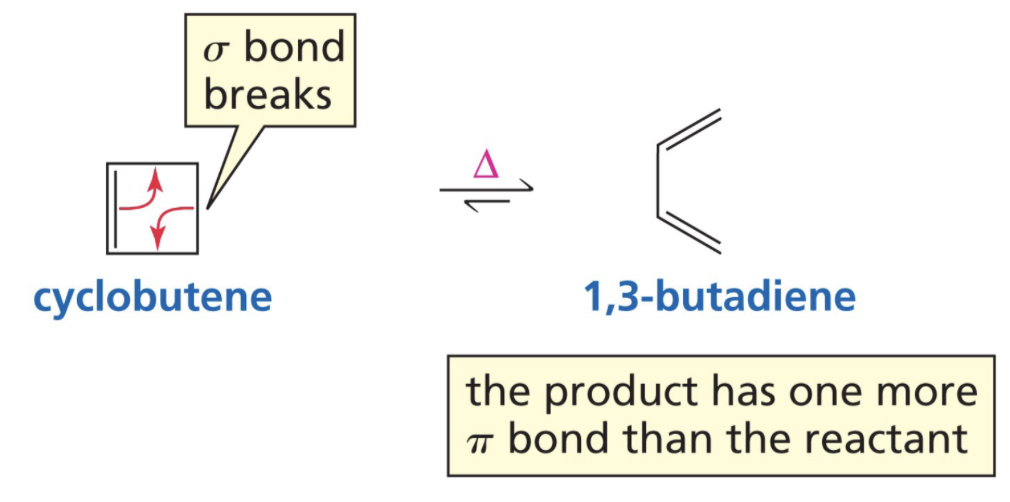
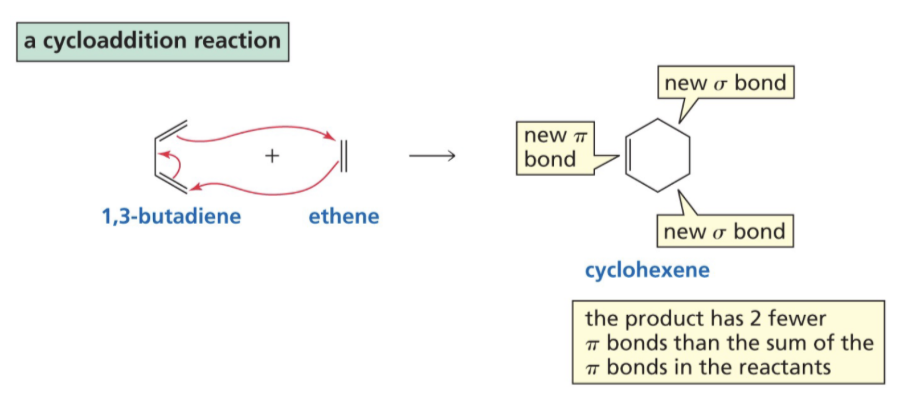
What is Cycloaddition?
Two different pi bond containing molecules react to form a cyclic compound.
Mechanism for Cycloaddition
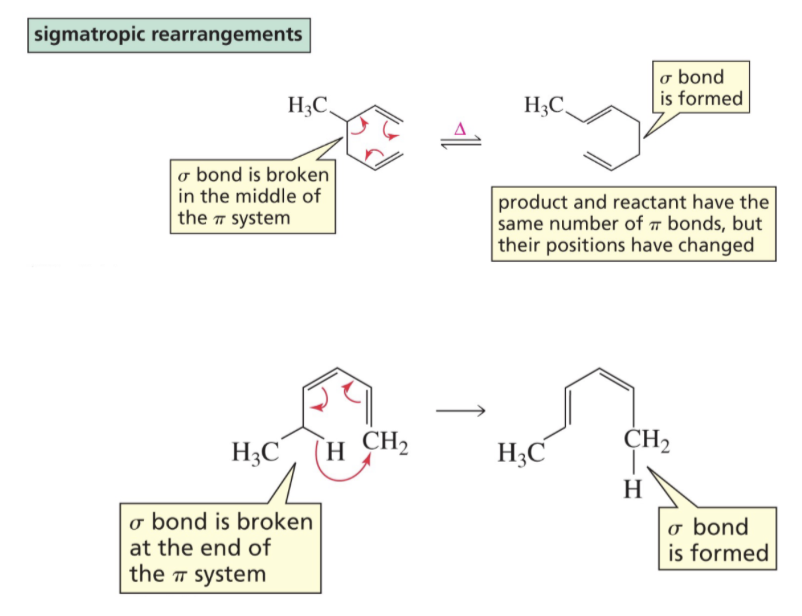
What is a sigmatropic rearrangement?
Sigma bond is broken in the reactant and a new sigma bond is formed in the product and pi bonds rearrange.
Show reaction mechanism of sigmatropic rearrangements?
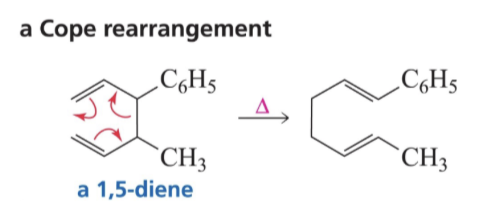
What are the two [3,3] Sigmatropic Reactions?
Cope Rearrangement
Claisen Rearrangement
What do you need for a Cope Rearrangement?
a 1,5 diene
Show reaction mechanism of Cope rearrangement?
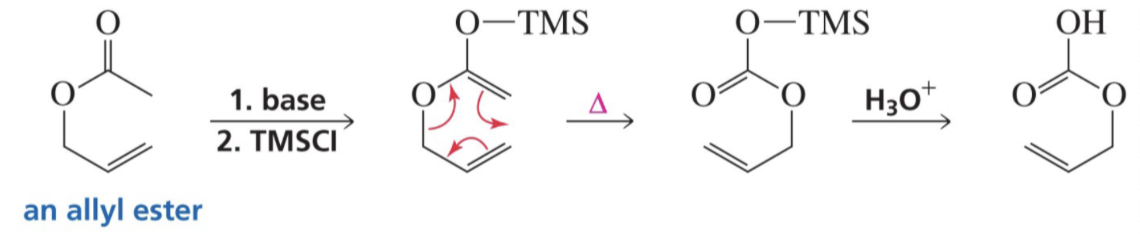
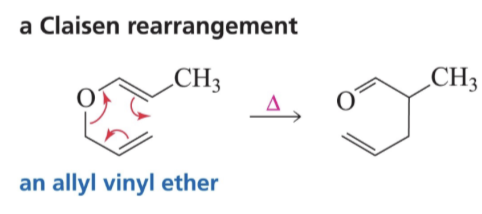
What do you need for a Claisen Rearrangement?
Allyl Vinyl Ether
Show reaction mechanism of Claisen Rearrangement?
Show reaction of the Ireland- Claisen Rearrangement