h - Synthetic polymers
1/6
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Addition polymer
Polymers are made by joining up lots of small repeating units called monomers
The monomers that make up addition polymers have double covalent bondLots of unsaturated monomer molecules can open up C=C double bonds + join to form polymer chains, called addition polymerisation
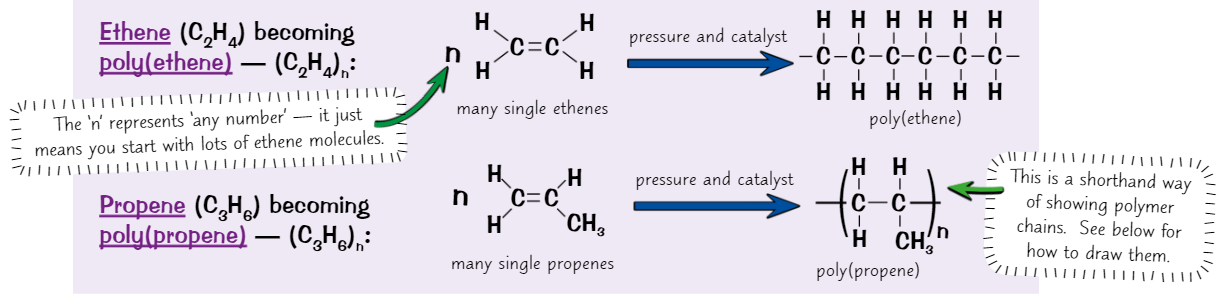
Naming + formula of polymers
Name of from type of monomer it’s made from - put brackets around it + put ‘poly’ in front
e.g. propene → poly(propene)To get formula of polymer, put formula of monomer in brackets + put a small ‘n’ after
e.g. C₃H₆ → (C₃H₆)ₙ
How to draw repeat unit of polymer
Join carbons together in a row with no double bonds, put pair of brackets around repeating unit, and put an ‘n’ after
Draw a bond from each of 2 carbons in chain that pass through brackets - shows that chain continues
To get displayed formula of monomer from displayed formula of polymer, do the reverse
Draw out the repeating unit of polymer, get rid of 2 bonds going out through brackets + put double bond between carbons
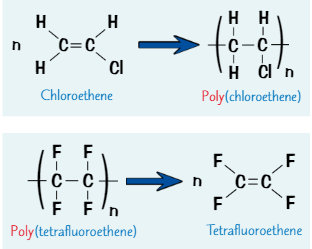
Problems with disposal of addition polymers
Inert - don’t react easily because C-C bonds in polymer chain are very strong + not easily broken
so it takes long time for addition polymers to biodegrade (be broken down by bacteria/other organisms)
Burning plastics can release toxic gases
so it’s difficult to dispose of polymers → best thing is to reuse then recycle
Condensation polymerisation
Involved 2 different types of monomer
Monomers react together + bonds form between them, making polymer chains
Each monomer must contain at least 2 functional groups, one on each end of molecule
Each functional group can react with functional group of another monomer, creating long chains of alternating monomers
For each new bond formed, a small molecule (e.g. water) is lost
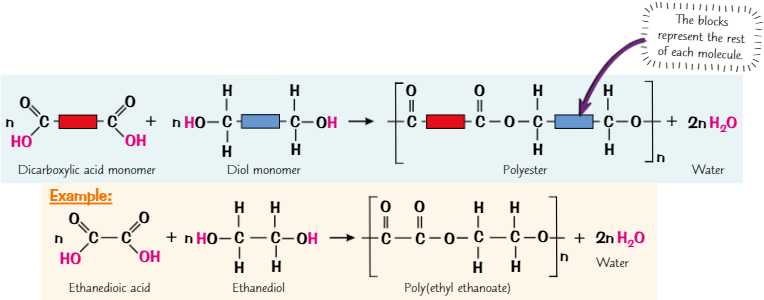
Polyesters
Formed when dicarboxylic acid monomers + diol monomers react together
Dicarboxylic acid monomers contain 2 carboxylic acid (-COOH) groups + diol monomers contain 2 alcohol (-OH) groups
When carboxylic acid group reacts with alcohol group, forms ester link
Polyesters are condensation polymers - each time ester link forms, a molecule of water is lost
Biopolyesters
Biodegradable polyesters, known as biopolyesters, can be broken down by bacteria + other living organisms in environment over time
They decompose + don’t stay in landfill forever, reducing polymers’ pollutant effect