MCAT General Chemistry - Solutions
1/56
Earn XP
Description and Tags
521
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
Solutions
homogeneous mixtures of two or more substances that combine to form a single phase, usually the liquid phase
ex. salt water, soda, cocktails, alloys
homogeneous
the same throughout
mixtures
combinations of distinct compounds
gas molecules do not interact all that much chemically, thus are not exactly solutions
solute
the substance being dissolved; move about freely in the solvent and interact with it by way of intermolecular forces; also relatively free to interact with other dissolved molecules of different chemical identities
solvent
component of the solution that remains in the same phase or is present in greater quantity or is more commonly used to dissolve things after mixing
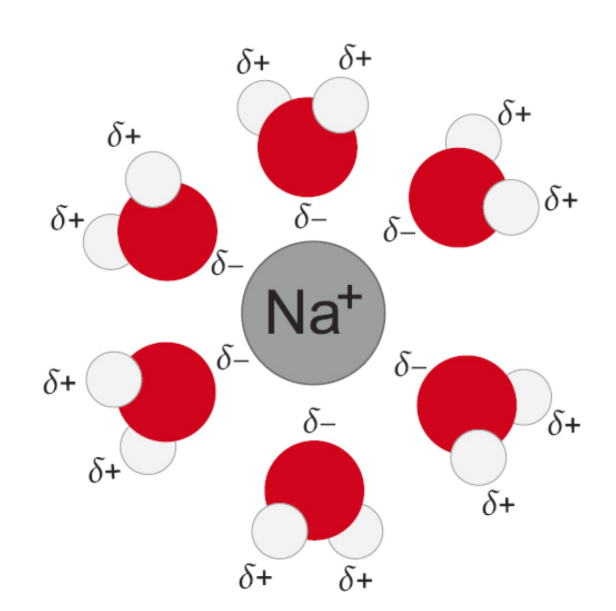
Solvation / dissolution
electrostatic interaction between solute and solvent molecules; maximises new intermolecular interactions
hydration
solvation in water
exothermic solvation
new interactions in solution are stronger than the original ones; favored at low temperatures
ex. gases into liquids
endothermic solvation
new interactions in solution are weaker than the original ones; favored at high temperatures
ex. sugar into water
ideal solution
overall strength of the new interactions in solution is equal to the overall strength of the original interactions; overall enthalpy change for the dissolution is close to / equals zero
entropy of dissolution
At constant temperature and pressure, entropy always increases upon dissolution
spontaneity of dissolution
depends on the change in Gibbs free energy
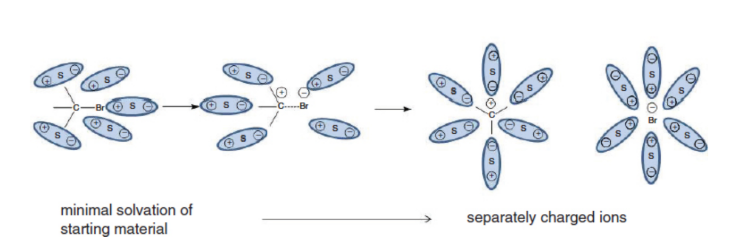
Solvation of NaCl Ions in Aqueous Solutions
component ions dissociate from each other and become surrounded by water molecules → endothermic
+3.87 kJ/mol
solubility
maximum amount of that substance that can be dissolved in a particular solvent at a given temperature
at 25°C, a maximum of 90.9 g glucose will dissolve in 100 mL H2O = 909 g/L
saturated
dissolved solute is in equilibrium with its undissolved state; at maximum equilibrium concentration; calculated IP is equal to the known Ksp
precipitating
solid form that removes itself from solution
dilute
solution in which the proportion of solute to solvent is small
concentrated
solution in which the proportion of solute to solvent is large
sparingly soluble salts
solutes that dissolve minimally in the solvent; molar solubility under 0.1 M
aqueous solution
solvent is water
hydronium ion (H3O+)
protonated water; facilitated by the transfer of a hydrogen ion (H+) from a molecule in solution to a water molecule (H2O)
solubility rules
ammonium (NH4+) & alkali metal (Group 1) = water-soluble
nitrate (NO3−) and acetate (CH3COO−) = water-soluble
Halides (Cl−, Br−, I−), excluding fluorides = water-soluble, EXCEPT with Ag+, Pb2+, Hg22+
sulfate = water-soluble, EXCEPT Ca2+, Sr2+, Ba2+, Pb2+
metal oxides = insoluble, EXCEPT alkali metals, ammonium, CaO, SrO, and BaO
hydroxides = insoluble, EXCEPT alkali metals, ammonium, Ca2+, Sr2+, Ba2+
carbonates (CO32-), phosphates (PO43-), sulfides (S2–), and sulfites (SO32-) = insoluble, EXCEPT alkali metals and ammonium
complex ion / coordination compound
a molecule in which a cation is bonded to at least one ligand
ex. tetraaquadiox-ouranyl cation, haemoglobin
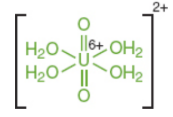
ligand
electron donor molecules attached to cations, can include water
coordinate covalent bonds
form very stable Lewis acid–base adducts
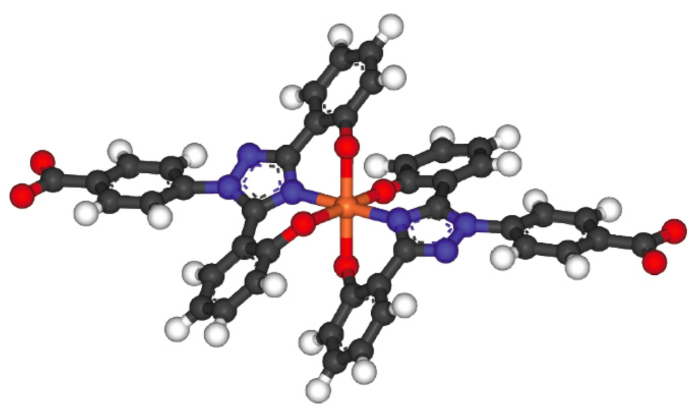
chelation
central cation bonded to the same ligand in multiple places; generally requires large organic ligands that can double back
ex. Iron with Two Molecules of Deferasirox
Concentration
the amount of solute dissolved in a solvent
volume percent
volume of solute divided by volume of solution times 100 percent
alcoholic proof
twice volume percent
degrees Brix (°Bx)
mass of glucose divided by mass of solution times 100 percent
percent composition by mass
msolute/msolution × 100%
used for: aqueous solutions, metal alloys, solid-in-solid solutions
mole fraction (X)
XA = molA/moltotal
sum of all compounds = 1
used for: partial pressure, vapor pressure depression
molarity (M)
M = molsolute/Vsolution in mol/L or molar (M)
used for: rate laws, the law of mass action, osmotic pressure, pH and pOH, and the Nernst equation
molality (m)
M = molsolute/msolution in mol/kg
used for: boiling point elevation and freezing point depression
normality (N)
equal to the number of equivalents of interest per liter of solution
equivalent
measure of the reactive capacity of a molecule; equal to a mole of the species of interest (protons, hydroxide ions, electrons, or ions)
DIlution
when solvent is added to a solution of higher concentration to produce a solution of lower concentration
dilution equation
MiVi = MfVf
where M is molarity, V is volume, and the subscripts i and f refer to the initial and final values
saturation point
the solute concentration is at its maximum value for the given temperature and pressure
solubility product constant (Ksp)
equilibrium constant for its solubility in aqueous solution; temperature dependent.
Ksp = [An+]m[Bm–]n
where the concentrations of the ionic constituents are equilibrium (saturation) concentrations
ion product (IP)
analogous to the reaction quotient, Q, for other chemical reactions
IP = [An+]m[Bm–]n
concentrations used in the ion product equation are the concentrations of the ionic constituents at that given moment in time
unsaturated
salt’s IP is less than the salt’s Ksp
supersaturated
salt’s IP is greater than the salt’s Ksp
molar solubility
molarity of a solute in a saturated solution
formation or stability constant (Kf)
formation of the complex ion in solution to become more soluble in solution
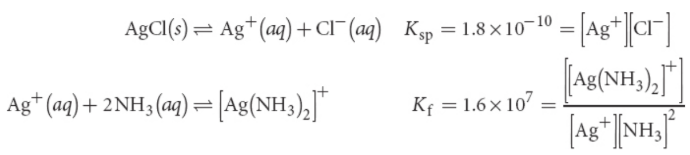
common ion effect
solubility of a salt is considerably reduced when it is dissolved in a solution that already contains one of its constituent ions as compared to its solubility in a pure solvent; no effect on the value of the solubility product constant
colligative properties
physical properties of solutions that are dependent on the concentration of dissolved particles but not on the chemical identity of the dissolved particles
list of colligative properties
vapor pressure depression
boiling point elevation
freezing point depression
osmotic pressure
Raoult’s law
accounts for vapor pressure depression
PA = XAPA°
where PA is the vapor pressure of solvent A when solutes are present, XA is the mole fraction of the solvent A in the solution, and PA° is the vapor pressure of solvent A in its pure state.
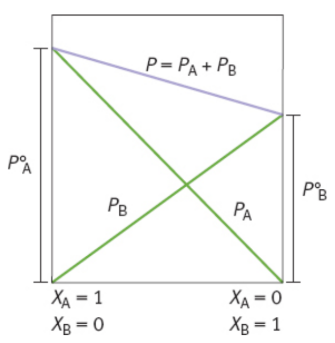
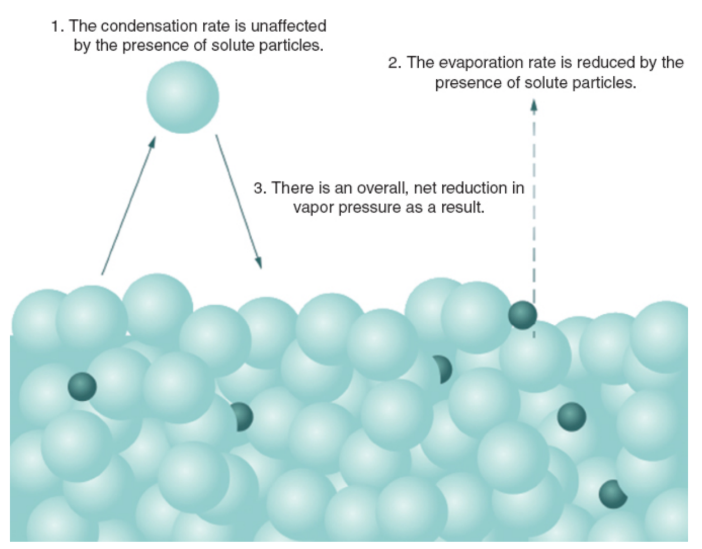
vapor pressure depression
As solute is added to a solvent, the vapor pressure of the solvent decreases proportionately
Boiling Point Elevation
When a nonvolatile solute is dissolved into a solvent to create a solution, the boiling point of the solution will be greater than that of the pure solvent
ex. pasta water
boiling point
temperature at which the vapor pressure of the liquid equals the ambient (incident) pressure
boiling point elevation equation
ΔTb = iKbm
where ΔTb is the increase in boiling point, i is the van’t Hoff factor, Kb is a proportionality constant characteristic of a particular solvent and m is the molality of the solution
van’t Hoff factor
corresponds to the number of particles into which a compound dissociates in solution
Freezing Point Depression
The presence of solute particles in a solution interferes with the formation of the lattice arrangement of solvent molecules associated with the solid state
ex. road salt
Freezing Point Depression equation
ΔTf = iKfm
where ΔTf is the freezing point depression, i is the van’t Hoff factor, Kf is the proportionality constant characteristic of a particular solvent, and m is the molality of the solution
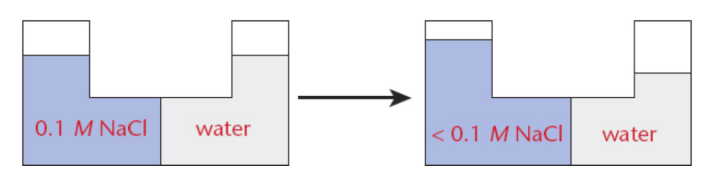
Osmotic pressure
“sucking” pressure generated by solutions in which water is drawn into a solution; formally, the amount of pressure that must be applied to counteract this attraction of water molecules for the solution
∏ = iMRT
where ∏ is the osmotic pressure, i is the van’t Hoff factor, M is the molarity of the solution, R is the ideal gas constant, and T is the temperature